US Pharm. 2014;39(8):56-60.
ABSTRACT: Given the aging of the population, chronic kidney disease (CKD) is increasing in prevalence owing to the rise in incidence of hypertension and diabetes mellitus. CKD is a progressive disease, and early detection and comprehensive management are key to preventing further complications. This is especially important in the inpatient setting, where management of fluid and electrolyte abnormalities, anemia, and mineral metabolism imbalances is essential to optimizing therapy. Pharmacists should be conversant with therapy goals for and management of hospitalized patients with CKD, the framework for classification, and the management and treatment of abnormal parameters associated with CKD. The knowledgeable pharmacist can have an impact on the management of this disease state.
Chronic kidney disease (CKD) is defined as abnormalities of kidney structure or function that are present for more than 3 months, resulting in a progressive loss of renal function that can occur over years. It is classified based on cause, glomerular filtration rate (GFR), and albuminuria. In the United States, about one in nine adults has CKD.1 In 2012, the prevalence was as high as 1,800 cases per million.2 The increase in CKD correlates with the increased prevalence of diabetes mellitus (DM) and hypertension, as these disease states are the two most common causes of CKD. The rising number of CKD cases and associated comorbidities is accompanied by an inherent increase in hospitalizations necessitated by complications of this disease state.
This patient population presents a unique challenge to healthcare professionals. Accordingly, this article discusses therapy goals and management of hospitalized patients with CKD. It also reviews key CKD concepts: the framework for classification, management, and treatment of abnormal parameters associated with CKD, and the impact of the pharmacist in comprehensive management of this disease state. Treatment approaches follow the National Kidney Foundation Kidney Disease Outcomes Quality Initiative’s CKD guidelines and the 2012 Kidney Disease Improving Global Outcomes clinical practice guideline for CKD and apply to adult patients not currently on dialysis.
Stages of CKD
The stages of CKD are stratified by measuring or calculating GFR, which is the gold standard for the quantitative index of renal function (TABLE 1).3,4 There is a gradual, age-related decline in GFR that often is asymptomatic and goes unnoticed; stress on the nephrons accumulates over time, especially in the presence of comorbidities such as hypertension and DM.5 The rate at which GFR declines can be a useful predictor of progression to end-stage renal disease (ESRD). GFR, usually expressed as mL/min, represents the volume of plasma ultrafiltrate presented to the nephrons per unit time during the process of urine formation.6

Creatinine Clearance as a Marker of Renal Function
Various estimations of GFR have been used in practice for recognition and risk stratification of CKD. Some commonly used equations are derived from the Modification of Diet in Renal Disease study.4 In these equations, GFR is measured by renal clearance of 125I-iothalamate. However, this form of measurement cannot be generalized to patients without CKD, as can the Cockcroft-Gault (CG) equation.
GFR cannot be directly measured, and calculation of creatinine clearance (CrCl) is the most widely used method of estimating GFR.6 A limitation of using CrCl as a marker for filtration is that tubular secretion of serum creatinine (SCr) overestimates GFR. The CG equation, the most commonly used method of calculating CrCl, identifies age and body mass as factors that affect CrCl: CrCl (mL/min) = [(140 – age in years) (weight in kg) (0.85 if female)]/[(72) (SCr in mg/dL)].
There is controversy regarding which body weight should be used, however. Actual, ideal, or adjusted body weight may be used, depending upon the institution. A limitation of the CG equation is that it cannot be used in patients with unstable renal function because SCr values are constantly changing and steady state cannot be assumed.4 However, because CrCl estimation using the CG equation remains the most commonly used method in clinical trials for drug development and FDA-approved package inserts, the use of other methods may result in dosing errors.
Acute Kidney Injury Versus Acute-on-Chronic Kidney Disease
Acute kidney injury (AKI) is defined as a rapid decline in renal function.7 AKI is not considered renal failure, but rather is seen as a functional impairment in relation to the physiological demands on the kidney. The stages of AKI, which are defined by changes in SCr or urine output (UOP), appear in TABLE 2.
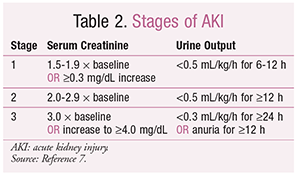
The RIFLE criteria (risk, injury, failure, loss of function, and ESRD) use SCr, UOP, and GFR to define AKI. There are a number of known causes of nonspecific AKI, including sepsis, critical illness, circulatory shock, burns, trauma, cardiac surgery, nephrotoxic drugs, radiocontrast agents, dehydration or volume depletion, advanced age, CKD, DM, cancer, and anemia.7 Targeting and treating the causes may reverse AKI progression.
Acute-on-chronic kidney disease is defined as a rapid deterioration and loss of kidney function. It is determined by an evaluation for potential urinary tract obstruction, volume assessment, and medication review.7
Treatment and Monitoring
Fluid Status: Hospitalized patients with CKD present a unique challenge owing to complications of fluid status. Regardless of the primary reason for hospital admission, CKD patients are at risk for fluid-status abnormalities such as venous congestion, sympathetic nervous system dysfunction, renin-angiotensin-aldosterone system imbalances, and disruption of the hypothalamic-pituitary axis. Volume overload develops early during the course of the disease and plays a significant role in the development of complications, especially those related to cardiovascular disease. Keeping the patient euvolemic can reduce the risk of complications secondary to volume overload.8
In CKD patients at high risk for AKI, it is important to maintain adequate renal perfusion and hemodynamic stability. When clinical assessment of volume status cannot be determined, central venous pressure can be measured.8 If intravascular volume is depleted, fluid resuscitation with isotonic solutions (i.e., normal saline) is preferred. If the patient is still hypotensive, vasopressors can be used to achieve a mean arterial pressure >65 mmHg.
Anemia of CKD: Anemia is a common complication in patients with CKD. Anemia of CKD develops progressively owing to blood loss or interference with red cell production.9 Anemia is diagnosed in patients aged >15 years when hemoglobin (Hgb) concentrations are <13.0 g/dL in males and <12.0 g/dL in females.9 Other tests for initial evaluation of anemia include CBC, RBC indices, platelet count, absolute reticulocyte count, serum ferritin levels, serum transferrin saturation (TSAT), serum vitamin B12, and folate levels.9
Currently, the most effective treatment options for anemia of CKD are iron-replacement agents and erythropoiesis-stimulating agents (ESAs) (TABLE 3).10-13 With regard to iron-replacement therapies, oral and parenteral agents are available; however, IV administration is superior.14 Patients whose levels fall below the goal iron parameters need a minimum dose of 1,000 mg of elemental iron to raise Hgb concentrations, followed by maintenance doses given once weekly to once monthly in order to maintain iron and Hgb levels.15 Doses, regimens, and side-effect profiles differ among available IV iron preparations. IV options for nondialysis inpatient treatment are given in TABLE 3. With IV iron therapy, the patient should be monitored for 60 minutes after infusion for potentially severe acute reactions, including hypotension, dyspnea, and anaphylaxis. Iron TSAT and ferritin levels should be evaluated every 3 months to determine whether iron therapy needs to be continued.14
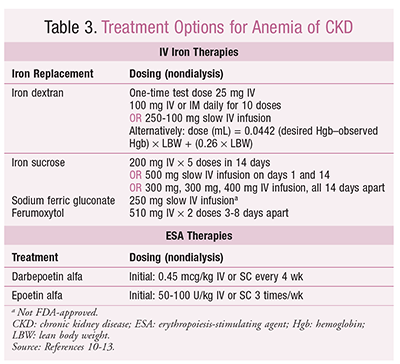
CKD-associated anemia also develops as a result of insufficient production of endogenous erythropoietin. When the Hgb level falls <10 g/dL, other causes of anemia must be ruled out prior to initiation of ESA therapy. Patients with Hgb levels <9 g/dL must be treated, whereas those with concentrations >10 g/dL should not be treated.9 The available ESA options are listed in TABLE 3. ESA therapy should not to be used to intentionally increase the patient’s Hgb concentration >13 g/dL because of the increased risk for venous thromboembolism.9 Once proper Hgb level is achieved, it should be monitored every 2 weeks until the patient has stabilized.16 If the Hgb level has risen too quickly, the ESA dosage should be reduced by 25% or withheld, if clinically necessary. Adverse reactions to monitor include pure red cell aplasia (rare but potentially dangerous), infusion reactions, hypertension, headache, tachycardia, nausea and vomiting, shortness of breath, hyperkalemia, and hypersensitivity reactions.15
Electrolyte Abnormalities: Common electrolyte imbalances in CKD patients include hyperkalemia, hyperphosphatemia, hypermagnesemia, hypernatremia or hyponatremia, and metabolic acidosis. Patients with renal dysfunction are more sensitive to sodium fluctuations with changes in fluid status. Most patients with stable CKD retain more sodium and water, leading to extracellular fluid volume expansion, but most patients remain asymptomatic. Alternatively, severe gastrointestinal (GI) loss and overdiuresis can compromise kidney function owing to hypoperfusion.8
Potassium levels must be carefully monitored because of potential deleterious effects on cardiac conduction. Additionally, ACE inhibitors and angiotensin receptor blockers can contribute to hyperkalemia, so extra caution is warranted. Potassium levels ≥6.5 mEq/L, or <6.5 mEq/L with ECG changes, constitute severe hyperkalemia. In patients without ECG changes, regular insulin, dextrose, or Kayexalate should be considered to lower potassium. If ECG changes are present, calcium gluconate can be used.8 Dialysis may be required in emergent situations.
Mineral Metabolism Abnormalities: Hyperphosphatemia is typically observed in CKD, as the amount of phosphate filtered decreases over time. It is recommended that serum phosphorus be maintained within the normal range (3.0-4.5 mg/dL) in stage 3-5 CKD patients to decrease morbidity and mortality. Baseline serum phosphate levels may be measured in adults with GFR <45 mL/min or in patients at risk for phosphate accumulation.17 Treatment generally involves phosphate-binding agents (TABLE 4), which bind dietary phosphorus in the GI tract and form insoluble phosphate compounds that are excreted in the feces.4
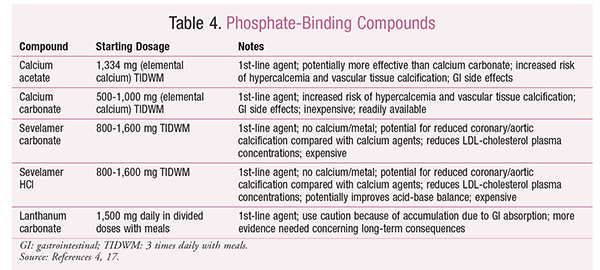
Because calcium-containing agents can increase the risk of hypercalcemia and vascular-tissue calcification, individualized therapy must be considered. Sevelamer-based agents are not absorbed and confer a lower incidence of hypercalcemia. However, because of increased costs and little evidence of a cause-and-effect relationship with reduced mortality, they are preferred in patients with normal serum calcium or in those at risk for hypercalcemia.17
In addition, serum calcium levels should be maintained in the normal range (8.5-10.5 mg/dL) in stage 3-4 patients and slightly lower than the normal range in stage 5 patients.18 Controlling phosphorus levels best controls serum calcium. Also, it is recommended that total elemental calcium intake (including dietary and calcium-based phosphate binders) not exceed 2,000 mg/day.4
Serum calcium-phosphate product (Ca + P) should be maintained <55 mg2/dL2 to prevent precipitation. When calculating Ca + P, corrected serum calcium should be used and Ca + P should be monitored regularly.18 The formula is: corrected serum Ca (mg/dL) = measured Ca (mg/dL) + [0.8 (normal albumin (g/dL) – measured albumin (g/dL))].4
CKD patients are at risk for vitamin D deficiency. However, in the absence of suspected or documented deficiency, it is not recommended to administer vitamin D supplements or analogues to suppress elevated parathyroid hormone (PTH) concentrations. If serum 25-hydroxyvitamin D <30 ng/mL and PTH levels are above the target range, supplementation with ergocalciferol (oral only) may be initiated (TABLE 5).18 The dose and duration of treatment depend upon the severity of the deficiency. To prevent vitamin D insufficiency, doses of 600 to 800 U/day are recommended.4 Calcitriol, the active form of ergocalciferol, also may be used, especially in patients with severe kidney disease, because it does not require conversion by the kidney to its biologically active form. Calcitriol (IV and oral) may be administered via conventional dosing (0.25-0.5 mcg/day) or pulse dosing (0.5-2 mcg 2-3 times per week).4 Baseline serum calcium, phosphorus, and Ca ´ P should be monitored regularly.

Pharmacist’s Role
The number of patients with CKD or at risk for it is increasing, and a substantial rise in stage 5 disease is expected in the next decade.4 Although it is crucial to delay the progression of CKD, it is equally important to detect and manage secondary complications. Common complications of CKD include fluid and electrolyte abnormalities, anemia, calcium and phosphorus abnormalities, and vitamin D deficiency. These complications can ultimately lead to cardiovascular and neural complications if not treated early.
Pharmacist intervention, appropriate monitoring, and patient education play significant roles in the comprehensive management of CKD. Pharmacists, as a vital part of the multidisciplinary healthcare team, can ensure appropriate and cost-effective medication therapy. Their expertise can help in preventing medication-induced injury and progression of disease, as well as providing a smoother transition of care upon discharge and ensuring that patients are better able to manage their disease state.
REFERENCES
1. St Peter WL. Introduction: chronic kidney disease: a burgeoning health epidemic. J Manag Care Pharm. 2007;13(9 suppl D):S2-S5.
2. Levey AS, Coresh J. Chronic kidney disease. Lancet. 2012;379:165-180
3. National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 suppl 1):S1-S266.
4. Dowling TC. Clinical assessment of kidney function. In: DiPiro JT, Talbot RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill; 2011:719-739
5. Kidney Disease: Improving Global Outcomes CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl. 2013;3:1-150.
6. Schwartz GJ, Furth SL. Glomerular filtration rate measurement and estimation in chronic kidney disease. Pediatr Nephrol. 2007;22:1839-1848
7. Kidney Disease: Improving Global Outcomes Acute Kidney Injury Work Group. KDIGO clinical practice guideline for acute kidney injury. Kidney Int Suppl. 2012;2:1-138.
8. Rahman MR, Shad F, Smith MC. Acute kidney injury: a guide to diagnosis and management. Am Fam Physician. 2012;86:631-639
9. Kidney Disease: Improving Global Outcomes Anemia Work Group. KDIGO clinical practice guideline for anemia in chronic kidney disease. Kidney Int Suppl. 2012;2:279-335.
10. Levin A, Hemmelgarn B, Culleton B, et al. Guidelines for the management of chronic kidney disease. CMAJ. 2008;179:1154-1162
11. National Kidney Foundation. KDOQI clinical practice guidelines and clinical practice recommendations for anemia in chronic kidney disease. Am J Kidney Dis. 2006;47(suppl 3):S1-S132.
12. Thomas R, Kanso A, Sedor JR. Chronic kidney disease and its complications. Prim Care. 2008;35:329-344
13. Irwin C, Moriarity-Suggs C. Anemia management in chronic and end-stage kidney disease. US Pharm. 2010;35(3)(Oncology/Hematology suppl):8-13.
14. INFeD (iron dextran) product information. Morristown, NJ: Watson Pharma, Inc; September 2009
15. Aranesp (darbepoetin alfa) product information. Thousand Oaks, CA: Amgen Inc; December 2013.
16. Epogen (epoetin alfa) product information. Thousand Oaks, CA: Amgen Inc; December 2013
17. Kidney Disease: Improving Global Outcomes CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder (CKD-MBD). Kidney Int. 2009;76(suppl 113):S1-S130.
18. National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42(4 suppl 3):S1-S201.
To comment on this article, contact rdavidson@uspharmacist.com.






