US Pharm. 2016;41(11):HS2-HS6.
ABSTRACT: Opioid use—encompassing prescription opioids received via the legitimate healthcare system and illegal substances (e.g., heroin) obtained through illicit distribution—in the United States has increased dramatically since 2002. Abrupt discontinuation of opioids can result in acute opioid withdrawal. Symptoms of opioid withdrawal are often managed in the inpatient setting. Methadone and buprenorphine are the two medications that are FDA-approved for use in treating opioid withdrawal. Morphine is typically used to treat infants with neonatal abstinence syndrome. Several different scoring systems have been used to quantify opioid-withdrawal symptoms and to guide therapy. Pharmacists can play an essential role in identifying the appropriate use of pharmacotherapy in acute opioid withdrawal.
Opioid abuse is an epidemic on two fronts. The first encompasses prescription opioids that are abused by patients who receive them via the legitimate healthcare system. The second front comprises illegal substances, such as heroin, that are obtained through illicit distribution. Heroin use increased dramatically from 2002 to 2013, especially in the age group of 18 to 25 years. Drug abuse may involve multiple agents, which may complicate presentations of withdrawal.1
In 2013, more people died of a drug overdose than in any other year on record, and most of these deaths involved an opioid. Opioid-related deaths have quadrupled from 1999 to 2014, with an apparent disconnect between pain and the prescription of opioids: During this time period, the amount of opioids prescribed nearly quadrupled; however, patients’ overall pain ratings remained the same.2 Given the abundance of prescription opioid-abuse problems, national attention has overwhelmingly focused on the appropriate prescribing of opioids, as well as patient and prescriber education. The CDC recently established guidelines for prescribing opioids for chronic pain, and all healthcare practitioners should be familiar with these guidelines in order to avoid inadvertently fostering abuse.3
Among acute opioid-abuse issues, opioid overdose is readily discussed; however, the problem of acute opioid-withdrawal syndrome, especially in the acute-care treatment setting, deserves equal attention. Treatment of opioid withdrawal depends on the healthcare setting, the type(s) of substances abused, and individual patient characteristics.
Healthcare Setting
Patients who abuse opioids are at increased risk for severe infections and other conditions requiring acute-care treatment in a hospitalized setting. Hospitalization may interrupt the patient’s typical abuse of opioids, resulting in withdrawal symptoms. Patients in this setting may or may not have an indication for treatment of pain. For example, a patient with no past medical history (other than IV drug abuse) who develops infective endocarditis may not have a pain indication for opioids or may not have pain requiring a dosage sufficient to avert opioid-withdrawal symptoms. Likewise, when typical starting doses of pain medications are used to treat acute pain in opioid-tolerant patients, a patient with a long-standing history of severe, chronic pain may exhibit signs or symptoms of opioid withdrawal.4
Individual Patient Characteristics
It is important to consider the method of exposure when a treatment protocol for withdrawing a patient is being determined. Patients may be prescribed opioids for legitimate indications (e.g., chronic pain), but then may develop opioid tolerance over time, leading them to experience withdrawal if opioids are withheld. Some patients with no true pain indication are addicted to illicit (e.g., heroin) or illicitly used substances (e.g., oxycodone). Newborns who were exposed to opioids in utero experience withdrawal within hours to days following birth, a situation referred to as neonatal abstinence syndrome (NAS).5 Opioids used in hospital settings for pain relief and sedation in ventilated patients may lead to withdrawal when they are discontinued after as few as 5 days of exposure to these infusions.6
Presentation of Withdrawal in the Acute Setting
Opioid-withdrawal syndrome may be classified into two phases: an initial, acute phase and a second, chronic phase with signs of protracted opioid withdrawal. Typically, it is the initial withdrawal symptoms, such as agitation, anxiety, dysphoria, insomnia, and temperature instability, that are evaluated and quantified in the acute-care setting.7
The type(s) of substances being used by the patient must be considered. Some data suggest that opiate-withdrawal symptoms are worse in patients who are codependent on opioids and benzodiazepines. On August 31, 2016, the FDA issued a black box warning for hundreds of drug products that contain either an opioid or a benzodiazepine, based on the risk of fatality when these drug classes are used concurrently.8 In a study conducted by Reed and colleagues, opiate-withdrawal symptoms were less severe in patients treated with buprenorphine than in those treated with methadone.9
Opioid withdrawal should be distinguished from other types of withdrawal—mainly alcohol, benzodiazepine, and barbiturate withdrawal—because management and treatment goals differ. It should be noted that acute-care patients who are admitted for other serious medical issues may choose to leave against medical advice or may otherwise not comply with necessary medical treatment. For this reason, acute opioid withdrawal should be seen as an urgent need and treated appropriately.
Pharmacologic Treatments in the Inpatient Setting
Only two agents—buprenorphine and methadone—are FDA-approved for the treatment of opioid-agonist withdrawal.10 The use of these agents depends on the situation and the setting. In the acute-care setting, a patient admitted for reasons other than opioid addiction or withdrawal treatment may be at risk for withdrawal symptoms if, prior to admission, he or she abused opioids; home chronic opioid treatment was abruptly stopped; or there was another situation in which large doses of opioids were abruptly discontinued. A thorough medication and social history of both prescription and illicit substances should be an integral part of the admission process. Understanding the physiological timing of withdrawal symptoms delineates possible withdrawal from other presentations. See TABLE 1 for typical timing of opioid withdrawal.
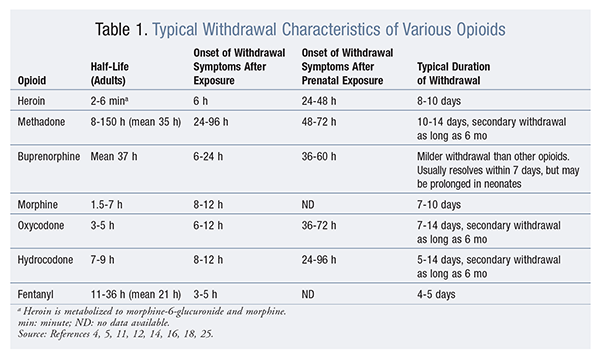
Patients who have a clear indication for opioid use may be started on an appropriate dose. However, the use of opioids for acute withdrawal is complicated when a patient lacks a clear diagnosis, as when he or she has no pain indication but uses illicit substances such as heroin. For these patients, opioids may be used as a bridge to prevent withdrawal during admission and possibly until entering a treatment program.11
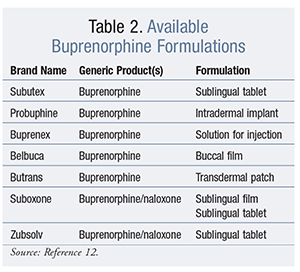
Buprenorphine: Buprenorphine is a mixed mu-receptor agonist-antagonist that demonstrates preferential mu-agonist properties at lower concentrations; however, there is a threshold at which it no longer produces mu-agonist effects. Buprenorphine has a more favorable side-effect and safety profile than methadone, and it is available in a naloxone combination product and alternative dosage forms, such as sublingual films, to reduce abuse potential.4,12,13 Buprenorphine and the naloxone combination are FDA-approved to treat opioid dependence and withdrawal.12,14 See TABLE 2 for available buprenorphine formulations.
Daniulaityte and colleagues conducted a Web-based study regarding lay attitudes toward traditional dosing of buprenorphine. Collected comments indicated that usual buprenorphine doses prescribed by physicians, such as 16 to 32 mg, were too high. Posts from lay drug users recommended conserving buprenorphine prescriptions by splitting the medication or taking it less often. Other anecdotal comments indicated that a dose much lower than those routinely prescribed is adequate for preventing withdrawal symptoms, possibly indicating the need to start buprenorphine at doses of 2 mg or 4 mg to prevent withdrawal and reduce abuse potential.15
Methadone: This agent, a mu agonist and N-methyl-d-aspartate (NMDA) antagonist, is a synthetic opioid analgesic FDA-approved for analgesia and detoxification or maintenance in opioid addiction. Methadone is used for opioid detoxification mainly because of its pharmacokinetic and pharmacodynamic parameters in withdrawal syndromes; compared with morphine, it has a slower onset, a more prolonged course, and milder symptoms, all of which favor a slower detoxification taper with a decreased risk of severe withdrawal. Methadone’s drawbacks include QTc prolongation and potential CYP3A4 and CYP2D6 interactions. Additionally, the analgesic effect of methadone is shorter than the pharmacologic half-life, leading to potential additive respiratory depression if the medication is dosed solely based on pain.12,16
Morphine: Morphine is a mu agonist with a shorter half-life and duration of action than methadone. Morphine is typically used to treat infants with NAS. Gradual weaning decreases the incidence of seizures and other severe symptoms of withdrawal. Morphine is typically given every 3 to 4 hours.5
Clonidine: Clonidine is an alpha2 agonist that is often used in conjunction with methadone. A recent Cochrane review described the usefulness of alpha2 agonists to relieve withdrawal symptoms in adult opiate-withdrawing patients. Alpha2 agonists were found to be more effective than placebo in lessening the likelihood of severe withdrawal.17 The main side effect of clonidine is hypotension. Abrupt discontinuation may result in increased heart rate and blood pressure.5
Nonpharmacologic Adjunctive-Treatment Options
Nonpharmacologic treatment options may be used alone in mild cases of neonatal withdrawal or in conjunction with medications in cases of more severe withdrawal. Gentle rocking and swaddling of the infant may decrease crying and promote sleep. Music therapy and massage therapy may soothe some infants. Mothers who room-in and breastfeed (when appropriate) can also help reduce the severity of withdrawal.5 Counseling and cognitive-behavioral therapy on the consequences of opioid addiction have moderate supporting evidence and can help address anxiety related to opioid withdrawal in adults.18
Opioid-Withdrawal Scoring Systems
Adults: Several opiate-withdrawal scales have been studied to quantify the amount of withdrawal patients are experiencing. The withdrawal scale used depends on the age of the patient. The Clinical Opiate Withdrawal Scale (COWS), which is useful in adults, is an 11-item scale consisting of observed, clinician-rated, and patient-rated items. Symptoms assessed include resting pulse rate, tremor, restlessness, yawning, pupil size, gooseflesh, and anxiety. Higher COWS scores reflect a greater number of withdrawal symptoms; scores exceeding 36 are indicative of severe withdrawal.19
Two additional scales used to evaluate opiate withdrawal in adults are the 16-item Subjective Opiate Withdrawal Scale (SOWS)20 and the 10-item Short Opioid Withdrawal Scale–Gossop (SOWS-Gossop).7 The SOWS assesses 16 different symptoms, each rated on a scale of 0 to 4 (0 = “not at all” to 4 = “extremely”). The total score can range from 0 to 64, with scores higher than 21 denoting severe withdrawal.20 The SOWS-Gossop was derived from the 32-item Opiate Withdrawal Scale and shortened to remove redundancies in reporting symptoms while maintaining an equal measure of the intensity of opiate-withdrawal symptoms.7
Children: Pediatric patients who are experiencing iatrogenic withdrawal from opioids and/or benzodiazepines may be evaluated with the Withdrawal Assessment Tool–Version 1 (WAT-1). The WAT-1 is an 11-item scale for monitoring autonomic dysregulation, gastrointestinal symptoms, hyperirritability, and motor abnormalities. Nursing staff evaluate a patient at risk for withdrawal for 2 minutes, stimulate the patient, and monitor for symptoms such as tremor or exaggerated startle for up to 5 additional minutes. Additionally, the patient’s medical records are examined to determine whether the patient has had fevers, loose stools, or vomiting in the previous 12 hours. The sum of the 11 numbers can range from 0 to 12, with increasing numbers indicating increasing withdrawal. WAT-1 is performed only twice daily, in contrast to other symptom-assessment scales, which are performed more often.21
Newborns: In the majority of cases, withdrawal in newborns is due to maternal substance exposure, whether licit or illicit. Infants who were exposed to opiates prenatally are at risk for NAS, which can cause increased fussiness and decreased growth and development. The most common scoring system used to evaluate the extent of withdrawal from NAS is the Finnegan scoring tool. Scoring in this age group is typically performed every 3 to 4 hours while the infant is awake. Symptoms such as excessive high-pitched crying, tremors, diarrhea, temperature instability, mottling, sneezing, and nasal stuffiness are assessed, and scores are evaluated.3 A higher numeric value on the Finnegan score indicates more severe withdrawal, and treatment with opioids generally is initiated when the scores average 8 or greater.22
Legal Implications
Methadone: Prescriptions for outpatient treatment of withdrawal and opioid dependence are restricted under Title 21 of the Code of Federal Regulations. Methadone is a Schedule II narcotic that may be prescribed only for use in opioid dependence and weaning by practitioners who have obtained a separate Drug Enforcement Administration (DEA) registration as a Narcotic Treatment Program in accordance with the Narcotic Addict Treatment Act (NATA). Practitioners must also register with the Center for Substance Abuse Treatment within the Substance Abuse and Mental Health Services Administration and any applicable state methadone authority.23
Adult inpatient admissions present a potential point of confusion concerning the NATA because narcotic-addicted patients, without access to their usual substances, may withdraw during their inpatient admission. Patients who are admitted to the hospital for reasons other than addiction may be continued on their maintenance opioid medications during the inpatient stay.23
The prescribing of methadone for children and infants who are withdrawing from opiates is another exception to the NATA. Infants who were exposed to opiates in utero are generally weaned by an inpatient facility for the course of the wean, which can take from 7 to 50 days or more, depending on the infant’s individual symptoms and the maternal substance(s) used. The Federal Interagency Narcotic Treatment Policy Review Board decided in 2001 that infants and children experiencing opioid withdrawal do come under the NATA because they are not addicts and cannot be enrolled in narcotic treatment programs. This means that physicians can write prescriptions for any opioid, including methadone, to treat pediatric patients with opioid-withdrawal syndrome.24
Buprenorphine: Buprenorphine was the first medication approved for the treatment of opioid addiction by practitioners outside of narcotic treatment programs. The Drug Addiction Treatment Act of 2000 guides recommendations for the prescribing of buprenorphine.25 Buprenorphine products may be prescribed only by a physician with a unique identification number (X-number) following his or her DEA number.23
Physicians who prescribe buprenorphine for opioid dependence must undergo specific education for prescribing and are limited to 30 patients in the first year and 100 patients thereafter after submitting a second notification.12 In August 2016, the U.S. Department of Health and Human Services released a final rule stating that physicians already treating up to 100 patients may apply to increase that number to 275 patients.26
Role of the Pharmacist
It is important for pharmacists to remain respectful and nonjudgmental when patients present in acute withdrawal. Careful medication reconciliation can identify the potential for withdrawal with proper use of validated scoring systems. Given the growing opioid-abuse epidemic, healthcare systems are increasingly managing the complications of acute opioid withdrawal. The management of acute opioid withdrawal relies on knowledge of laws affecting prescribing medications for opioid withdrawal, which medications or other substances the patient may be withdrawing from, and pharmacologic management options.
REFERENCES
1. CDC. Today’s heroin epidemic. www.cdc.gov/vitalsigns/heroin. Accessed August 1, 2016.
2. CDC. Opioid overdose: prescribing data. www.cdc.gov/drugoverdose/data/prescribing.html. Accessed October 17, 2016.
3. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49.
4. Nicholls L, Bragaw L, Ruetsch C. Opioid dependence treatment and guidelines. J Manag Care Pharm. 2010;16(1 suppl B):S14-S21.
5. Kocherlakota P. Neonatal abstinence syndrome. Pediatrics. 2014;134:e547-e561.
6. Galinkin J, Koh JL; Committee on Drugs; Section on Anesthesiology and Pain Medicine; American Academy of Pediatrics. Recognition and management of iatrogenically induced opioid dependence and withdrawal in children. Pediatrics. 2014;133:152-155.
7. Vernon MK, Reinders S, Mannix S, et al. Psychometric evaluation of the 10-item Short Opiate Withdrawal Scale-Gossop (SOWS-Gossop) in patients undergoing opioid detoxification. Addict Behav. 2016;60:109-116.
8. FDA. FDA requires strong warnings for opioid analgesics, prescription opioid cough products, and benzodiazepine labeling related to serious risks and death from combined use. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm518697.htm. Accessed September 30, 2016.
9. Reed LJ, Glasper A, de Wet CJ, et al. Comparison of buprenorphine and methadone in the treatment of opiate withdrawal: possible advantages of buprenorphine for the treatment of opiate-benzodiazepine codependent patients? J Clin Psychopharmacol. 2007;27:188-192.
10 Choo C. Medications used in opioid maintenance treatment. US Pharm. 2009;34(11):40-53.
11. Lintzeris N, Clark N, Winstock A, et al. National clinical guidelines and procedures for the use of buprenorphine in the maintenance treatment of opioid dependence. http://nationaldrugstrategy.gov.au/internet/drugstrategy/publishing.nsf/Content/buprenorphine-guide/$FILE/bupren.pdf. Accessed October 14, 2016.
12. Clinical Pharmacology (online database). www.clinicalpharmacology.com. Tampa, FL: Gold Standard, Inc; 2015. Accessed July 29, 2016.
13. Dunlap B, Cifu AS. Clinical management of opioid use disorder. JAMA. 2016;316:338-339.
14. Buprenorphine package insert. Parsippany, NJ: Actavis Pharma, Inc; January 2015.
15. Daniulaityte R, Carlson R, Brigham G, et al. “Sub is a weird drug:” a web-based study of lay attitudes about use of buprenorphine to self-treat opioid withdrawal symptoms. Am J Addict. 2015;24:403-409.
16. Methadone package insert. Newport, KY: Xanodyne Pharmaceuticals, Inc; March 2006.
17. Gowing L, Farrell M, Ali R, White JM. Alpha2-adrenergic agonists for the management of opioid withdrawal. Cochrane Database Syst Rev. 2016;(5):CD002024.
18. Berna C, Kulich RJ, Rathmell JP. Tapering long-term opioid therapy in chronic noncancer pain: evidence and recommendations for everyday practice. Mayo Clin Proc. 2015;90:828-842.
19. Wesson DR, Ling W. The clinical opiate withdrawal scale (COWS). J Psychoactive Drugs. 2003;35:253-259.
20. Bradley BP, Gossop M, Phillips GT, Legarda JJ. The development of an opiate withdrawal scale (OWS). Br J Addict. 1987;82:1139-1142.
21. Franck LS, Harris SK, Soetenga DJ, et al. The Withdrawal Assessment Tool-1 (WAT-1): an assessment instrument for monitoring opioid and benzodiazepine withdrawal symptoms in pediatric patients. Pediatr Crit Care Med. 2008;9:573-580.
22. Hudak ML, Tan RC; Committee on Drugs; Committee on Fetus and Newborn; American Academy of Pediatrics. Neonatal drug withdrawal. Pediatrics. 2012;129:e540-e560.
23. Drug Enforcement Administration. Practitioner’s manual: section VI–opioid (narcotic) addiction treatment programs. www.deadiversion.usdoj.gov/pubs/manuals/pract/section6.htm. Accessed August 1, 2016.
24. Methadone prescriptions for neonatal abstinence syndrome. West Virginia Board of Pharmacy News. https://nabp.pharmacy/wp-content/uploads/2016/06/WV032012.pdf. Accessed July 31, 2016.
25. Substance Abuse and Mental Health Services Administration. Buprenorphine. http://buprenorphine.samhsa.gov/faq.html. Accessed July 31, 2016.
26. Department of Health and Human Services. Medication assisted treatment for opioid use disorders. https://s3.amazonaws.com/public-inspection.federalregister.gov/2016-16120.pdf. Accessed September 30, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.






