US Pharm. 2006;12:HS32-HS41.
Orthostatic hypotension is defined by the Consensus Committee of the American Autonomic Society and the American Academy of Neurology as a reduction of systolic blood pressure of at least 20 mmHg or a reduction of diastolic blood pressure of at least 10 mmHg within three minutes of standing. For individuals who cannot stand, a drop in blood pressure within three minutes of tilting the head up to at least a 60° angle is indicative of orthostatic hypotension. The Consensus Committee describes orthostatic hypotension as a physical sign, rather than a disease, that may be symptomatic or asymptomatic.1 Common symptoms of orthostatic hypotension include light-headedness, dizziness, weakness, fatigue, cognitive impairment, nausea, palpitations, and tremulousness.1
The prevalence of orthostatic hypotension may increase with age, and studies report that as many as 13% to 30% of elderly people have orthostatic hypotension.2-5 Orthostatic hypotension as defined in these studies could be systolic or diastolic orthostatic hypotension, both systolic and diastolic orthostatic hypotension, or symptomatic orthostatic hypotension. Thus, depending on the definition of orthostatic hypotension in the cohort under study, the prevalence of orthostatic hypotension might vary.2-5 Food ingestion, time of day, state of hydration, hypertension medication, gender, and age can also affect the incidence of orthostatic hypotension.1 In a prospective study of 60 hypertensive individuals 65 or older, Vara-Gonzalez et al. determined the reproducibility (intra-individual variability) of postural changes in blood pressure in a primary care setting. Findings suggested that the reproducibility of orthostatic hypotension in the cohort was poor. The researchers recommended that orthostatic blood pressure be confirmed on at least two occasions in elderly individuals.6
Several studies have reported that orthostatic hypotension is a potential risk factor for vascular-related death, myocardial infarction, coronary heart disease, transient ischemic attack, falls, and fractures.7-11 Orthostatic hypotension has also been associated with increased mortality.12,13 Thus, orthostatic hypotension is associated with serious risks in the older population, adversely affecting patients' health and quality of life and increasing financial and social burdens.14
Pathophysiology
In healthy individuals,
gravitational venous pooling in the legs and abdomen begins immediately when
they change from supine to standing position.15 The subsequent
transient decrease in venous return and cardiac output results in reduced
blood pressure. Most of the venous pooling occurs within the first 10 seconds,
and venous pooling is completed within three to five minutes of orthostatic
stress.16 The initial reaction to orthostatic stress is mediated by
the autonomic nervous system. During prolonged orthostatic stress
(longer than three minutes), additional adjustments are mediated by the
humoral limb of the autonomic nervous system. The main sensory receptors
involved in orthostatic neural reflex adjustment are the arterial
mechanoreceptors (baroreceptors) located in the aortic arch, and it is
believed that the carotid sinuses and mechanoreceptors located in the heart
and lungs (cardiopulmonary receptors) play a minor role.17 During
orthostatic stress, these receptors activate autonomic reflexes that cause a
sympathetic mediated increase in catecholamine levels and result in increased
heart rate, myocardial contractility, and enhanced cardiac output. The
increase in catecholamine levels also induce arterial and venous
vasoconstriction that rapidly normalize blood pressure.18 During
prolonged orthostatic stress, additional activation of the neurohumoral system
reinforces the activation of the cardiovascular reflexes through additional
constriction of blood vessels and minimization of water loss by activation of
the renin-angiotensin-aldosterone system and vasopressin.15 When
any of these responses to orthostatic stress is impaired, blood pressure and
organ perfusion may be reduced upon standing, and the patient may display
symptoms of orthostatic hypotension.
Etiologies
There are many causes for
orthostatic hypotension, which can be categorized into neurogenic and
nonneurogenic causes.19 Neurogenic causes can be subdivided into
primary or secondary causes. Primary neurogenic causes include autonomic
nervous system dysfunction and multiple system atrophy (formerly known as
Shy-Drager syndrome). Patients with multiple-system atrophy have a loss of
neurons in nuclei of the central nervous system and present with parkinsonian
features, cerebellar dysfunction, or pyram idal symptoms. Secondary
neurogenic causes include systemic diseases resulting in lesions of the
central or peripheral nervous system (e.g., diabetes mellitus, Guillian-Barre
syndrome, alcoholic polyneuropathy).
One feature that distinguishes a nonneurogenic cause from a neurogenic cause is that the postural drop in blood pressure that is associated with nonneurogenic etiologies is accompanied by a compensatory increase in heart rate. This increase in heart rate does not occur in individuals with neurogenic causes of orthostatic hypotension.19,20 Nonneurogenic causes of orthostatic hypotension include cardiac pump failure, reduced intravascular volume, venous pooling, or causative medications.
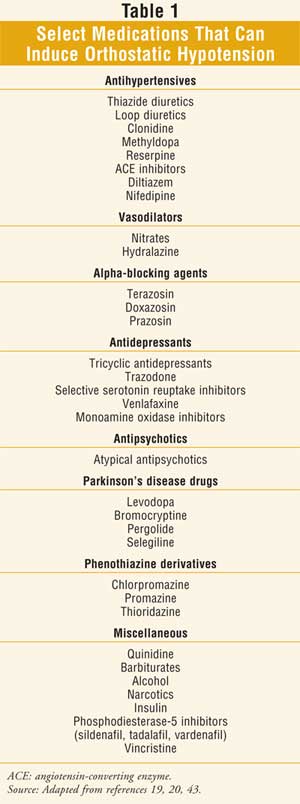
It is estimated that about 50% of orthostatic hypotension cases involve causative medications (see table 1 for a list of select causative medications).21 Antihypertensives that impair autonomic reflex mechanisms are frequent causes of orthostatic hypotension. Drugs that induce diuresis can reduce intravascular volume and cause orthostatic hypotension. Peripheral vasodilation resulting from the use of non-selective alpha-blocking agents is associated with first-dose syncope, and therefore, dose should be started low and slowly titrated upwards.18 In a retrospective study of veterans 75 and older, Poon and Braun reported that the incidence of orthostatic hypotension increased with increasing numbers of concurrent medications used that are known to cause orthostatic hypotension.21 Therefore, the use of potentially causative medications for orthostatic hypotension should be carefully evaluated in older patients, particularly those who have concurrent illnesses that can cause orthostatic hypotension, such as cardiac impairment, low intravascular volume, or vasodilation.
Comorbidities and Consequences
There are a number of associated
comorbidities of orthostatic hypotension. Stroke, isolated systolic
hypertension, and dementia have all been associated with orthostatic
hypotension. In a study by Eigenbrodt et al., 11,707 patients who were
initially stroke-free were followed for a period of 7.9 years.11
Orthostatic hypotension was found to be statistically predictive of ischemic
stroke, even after adjustment for numerous other stroke risk factors (hazard
ratio, 2.0; 95% CI, 1.2 to 3.2). In another study by Rutan et al., 5,201 men
and women who were 65 or older at initial examination were studied to
determine the prevalence of orthostatic hypotension and its associations with
various cardiovascular risk factors and symptomatology.8 In this
study, orthostatic hypotension was significantly associated with isolated
systolic hypertension (odds ratio, 1.35; CI, 1.09 to 1.68). Studies have also
been conducted to determine the effect of orthostatic hypotension on cognition
and dementia. In a study by Elmstahl and Rosen, 33 healthy women ages 75 to 95
with no signs of cerebrovascular disease or dementia at baseline examination
were enrolled into a longitudinal five-year follow-up study.22
Seven women had documented cognitive decline at the five-year follow-up and a
greater decrease in orthostatic blood pressure during tilting at baseline (16
mmHg) than did controls (1 mmHg; P<.01).
There are also a number of consequences of orthostatic hypotension that can lead to decreased quality of life, such as headaches, blurred vision, syncope, falls, fractures, and dyspnea. These consequences can lead to significant debilitation necessitating long-term care and are especially concerning and problematic to the caregivers of these patients.
Evaluation
History and Physical
Examination:
The initial step in evaluating patients with orthostatic hypotension is the
identification of causative medications that induce orthostatic hypotension.
19,23 The history should also include a review of stimuli that increase
the incidence of orthostatic hypotension. Such stimuli include meals, hot
environments (after showers), alcohol consumption, hyperventilation, and time
of the day. Once the stimuli are identified, safety precautions should be
practiced to prevent any falls secondary to orthostatic hypotension. For
example, orthostatic hypotension occurs more commonly in the early morning;
therefore, patients should schedule activities in the afternoon.24
A comprehensive review of the patient's medical history should be performed to
identify neurogenic and nonneurogenic causes of orthostatic hypotension. The
physical examination should include measurement of supine and standing pulse
and blood pressure, with an interval of three minutes between measurements in
each position. A thorough neurologic evaluation--including a mental status
examination to identify cognitive impairment, a cranial nerve assessment to
evaluate down gaze, a motor examination to identify parkinsonian features such
as tremor, rigidity, and bradykinesia, and a sensory evaluation to identify
polyneuropathies--should be completed.19
Diagnostic Tests: There are many autonomic function tests available to assist the practitioner in identifying the cause of orthostatic hypotension, especially when history and physical examination are inconclusive.21 A commonly used test is Valsalva's maneuver, which assesses the functional integrity of the baroreceptor reflex. During the test, the patient exerts a constant expiratory pressure for 15 seconds while blood pressure and heart rate changes are recorded.19 Other autonomic function tests such as the tilt table test, the quantitative sudomotor axon reflex test, and the cold pressor test can be used to assess orthostatic hypotension.
Treatment of Orthostatic Hypotension
Nonpharmacologic Treatment:
Nonpharmacologic measures are the
mainstay of treatment in the management of orthostatic hypotension. The
predominant cause of the signs and symptoms of orthostatic hypotension is the
inability of the body to compensate the normal gravitational pooling of the
blood in the lower extremities and abdomen. Therefore, nonpharmacologic
approaches should focus on decreasing venous pooling, avoiding volume
depletion, and increasing plasma volume.
Patients should be encouraged to change position slowly. When rising from a supine position, patients should be encouraged to sit on the side of the bed for a few minutes before standing up. If getting up after a period of prolonged sitting, patients should cross their legs a couple of times prior to standing. Lift chairs may also be useful for these situations. Use of compression stocking and abdominal binders are encouraged; however, they must be worn all day to be useful, and many patients find them uncomfortable to wear, especially during hot weather.25 Physical adjustments and use of restrictive agents raise cardiac output and mean blood pressure.26
Additionally, adequate fluid intake should be maintained to prevent dehydration and to improve the symptoms of orthostatic hypotension. The usual recommendation is to drink 2.0 to 2.5 L (66 to 83 ounces) of fluids during the day, while avoiding drinking one hour prior to bedtime to minimize nighttime trips to the bathroom. In a study by Shannon et al., 11 patients with severe orthostatic hypotension due to autonomic failure ingested 480 mL (16 ounces) of tap water over a period of five minutes.27 Mean (± standard deviation) blood pressure after one minute of standing was 83 ± 6/53 ± 3.4 mmHg at baseline, which increased to 114 ± 30/66 ± 18 mmHg ( P <.01) 35 minutes after drinking water (P<.01).
Individuals experiencing postprandial orthostatic hypotension due to gastrointestinal and hepatic pooling should be counseled to eat more frequent, smaller meals and to plan on a 60-minute postprandial resting period.28 Shannon et al. tested the effect of drinking water on postprandial hypotension in seven patients who had autonomic failure. After they had eaten a meal, blood pressure decreased by 43 ± 36/20 ± 13 mmHg without patients drinking water, compared with 22 ± 10/12 ± 5 mmHg with drinking (P<.001).27 Sodium intake should also be increased, as long as there are no contraindications.
Patients should be advised to avoid hot showers or exercising in warm environments. Patients with orthostatic hypotension often have an inability to sweat, which causes a rise in core body temperature that may lead to compensatory vasodilation and subsequent fall in blood pressure. 26 While patients are in bed, the head should be elevated 10° to 20°. This elevation reduces renal arterial pressure and causes release of rennin, resulting in aldosterone release, retention of sodium and water, and thereby, increasing blood pressure.28 Coffee drinking is encouraged to enhance peripheral resistance and reduce postprandial exacerbations. In addition, eating black licorice has been documented to exhibit mineralocorticoid effects.28,29 Straining during micturition and defecation should be minimized; therefore, these patients should be advised to increase fiber consumption, especially in the form of wheat and oat bran. All of these nonpharmacologic treatment strategies minimize the impact of postural orthostatic hypotension on activities of daily living.
Pharmacologic Treatment: Pharmacologic treatment is aimed at increasing central plasma volume and preventing plasma pooling. A number of agents have been studied for this purpose.
Midodrine, a peripheral selective direct alpha-1 adrenoreceptor agonist, is the only FDA-approved agent that is specifically indicated for treatment of orthostatic hypotension. It works by inducing both arterial and venous constriction. Midodrine is enzymatically hydrolyzed to the pharmacologically active metabolite, desglymidodrine. Desglymidodrine works by binding to alpha-1 receptors on arteries and veins, producing an increase in vascular tone and elevating blood pressure. 30 Dosing should start at 2.5 mg three times per day, while gradually increasing to a target dose of 10 mg three times per day. Midodrine should be dosed in three- to four-hour intervals and should not be administered within four hours of bedtime or in the evening, because the drug can increase the incidence of supine hypertension.28 In a randomized, double-blind, placebo-controlled study by Low et al., 171 patients with orthostatic hypotension were randomized to receive either placebo or midodrine 10 mg three times per day for a period of six weeks.31 On day 15 of the treatment period, midodrine improved systolic blood pressure while standing by 22.4 mmHg (24%) versus 6 mmHg (6%) with placebo (P <.01). Midodrine improved diastolic blood pressure during standing by 13.3 mmHg (21%) versus 4.3 mmHg (7%) with placebo (P <.01). Reports of light-headedness by the end of the second week of treatment was lower (P = .001) and the global symptom relief score rated by both the patient (P = 0.03) and the investigator (P <.001) was higher in the midodrine group. The most common adverse effects were piloerection, urinary retention, and supine hypertension.
Fludrocortisone, a synthetic mineralocorticoid, is used as a first-line agent. Small oral doses produce marked sodium retention and increased urinary potassium excretion with a resultant rise in blood pressure which is apparently due to increased angiotensin activity and fluid retention.32,33 Treatment is started at 0.1 mg daily, and maintenance doses usually range from 0.1 to 0.4 mg daily, with titration of 0.1 mg occurring at about one- to two-week intervals.28 Side effects include hypokalemia, which may necessitate potassium supplementation, as well as edema, dizziness, headaches, and supine hypertension. Electrolytes should be checked one week following initiation of therapy and approximately each week following a dose increase.
Dihydroergotamine (DHE) causes vasoconstriction by stimulating alpha-adrenergic receptors and by inhibiting reuptake of norepinephrine, leading to increases in venous return and decreases in venous stasis and pooling.34 Dosing should start at 1.0 mg intramuscularly (IM) once daily or at 0.0065 to 0.013 mg/kg subcutaneously (SC) once daily in the morning. Maximum dosage is 3.0 mg IM or SC in a 24-hour period and 6.0 mg IM or SC on a weekly basis. Side effects include myalgia, numbness and tingling in extremities, edema, pruritus, dizziness, and hypertension.
Erythropoietin has also been utilized for management of orthostatic hypotension. Anemia is now considered to be an important cause of orthostatic hypotension, and treatment with erythropoietin increases intravascular volume, leading to improvements in orthostatic tolerance.35 The recommended dose is 25 to 50 U/kg SC three times per week. In a small clinical study by Perera et al., treatment with erythropoietin (4,000 U SC biweekly for six weeks) increased hematocrit and blood pressure in all patients.36 Hematocrit increased from 33.9% ± 0.7% to 44.3% ± 1.4%, blood pressure in supine position increased from 150 ± 8 mmHg/87 ± 8 mmHg to 166 ± 25 mmHg/92 ± 12 mmHg (P <.05), and after three minutes in the head-up tilt position, from 86 ± 21 mmHg/47 ± 15 mmHg to 102 ± 23 mmHg/63 ± 12 mmHg, (P <.05). Side effects include headache, seizures, edema, and hypertension.
Octreotide is a long-acting somatostatin analogue that inhibits the secretion of hormones involved in vasodilation, including vasoactive intestinal peptide. Octreotide increases splanchnic arteriolar resistance and decreases gastrointestinal blood flow, resulting in increases in both semirecumbent and standing blood pressure, and is most useful in patients with autonomic orthostatic hypotension.37,38 The recommended doses range from 0.2 to 0.4 mcg/kg SC, with a maximum dose of up to 1.6 mcg/kg. The effectiveness of octreotide was studied in a small uncontrolled study by Hoeldtke and Israel.38 Doses ranging from 0.2 to 0.4 mcg/kg resulted in an average increase in semirecumbent mean blood pressure of 15 to 20 mmHg in patients with autonomic failure and diabetic autonomic neuropathy. Its use is limited by the fact that gastrointestinal symptoms worsen in patients who initially have these symptoms. Other side effects include nausea, vomiting, diarrhea, and hyperglycemia.
Clonidine, an alpha-2 receptor agonist, is used to treat orthostatic hypotension in patients with severe autonomic dysfunction. Clonidine stimulates alpha-1 receptors in peripheral vascular smooth muscle, resulting in acute vasoconstriction and an increase in blood pressure.39 The central hypotensive effect of this drug is reduced in this patient population.26The doses of clonidine that have been studied for this indication include 0.4 and 0.8 mg daily, in twice-daily dosing. Side effects include sedation, dry mouth, altered mental status, and excessive hypertension. 39
Pyridostigmine, an acetylcholinesterase inhibitor, may be utilized for the treatment of orthostatic hypotension. This drug works by enhancing sympathetic ganglion transmission, thereby leading to increased systemic resistance. In a study by Singer et al., 15 patients with neurogenic orthostatic hypotension who were treated with pyridostigmine 60 mg showed increases in their orthostatic pressure and improvements in their orthostatic symptoms (P >.05).40 Side effects include abdominal cramps, blurred vision, diaphoresis, diarrhea, and miosis.
Other agents that have been utilized in a small number of patients include desmopressin acetate, yohimbine, indomethacin, ephedrine, pseudoephedrine, ergotamine, vitamin B12, fluoxetine, and nonselective beta-blockers such as pindolol.28 Combination therapy consisting of midodrine and fludrocortisone, midodrine and pyridostigmine, or midodrine and octreotide has demonstrated increases in blood pressure compared with monotherapy.28,37,41 Studies evaluating these combination therapies have been of short duration; therefore, the impact of combination therapy on adverse events has been minimal. Another treatment option for patients with severe orthostatic hypotension is placement of dual-chamber pacemakers.25
The Pharmacist's Role
Education and prevention are
important components of caring for patients with orthostatic hypotension, and
that is where pharmacists should and must play a major role. Pharmacists can
educate patients and caregivers about the appropriateness of following
nonpharmacologic treatment strategies. Patients and caregivers must be
educated about following preventive strategies, including proper diet and
physical activity.
In addition, pharmacists must take an active role in educating, identifying, and assisting in the selection and modification of appropriate medication for hypertension. Pharmacists should take a complete medical history to determine if the cause of orthostatic hypotension is drug induced and contact a physician to assist in the selection of alternative treatments, with the goal of minimizing the number of medications that may induce orthostatic hypotension and its complications.
If a patient is taking a potentially causative medication, the primary treatment of choice is discontinuation of the drug. In fact, simply stopping antihypertensive therapy can successfully treat orthostatic hypotension in half the patients with these symptoms.42,43 Ending treatment with certain classes of antihypertensives, such as diuretics, vasodilators, nondihydropyridine calcium channel antagonists, and alpha-blockers has shown greater promise in treating orthostatic hypotension, compared with others such as angiotensin-converting enzyme inhibitors, beta-antagonists with intrinsic sympathomimetic activity, and angiotensin-receptor antagonists.
Conclusion
Orthostatic hypotension is a
common cause of increased morbidity and mortality, especially in the elderly
population. There are many reversible and nonreversible causes of orthostatic
hypotension, both neurogenic and nonneurogenic. Pharmacists have a role in
educating patients about both pharmacologic and nonpharmacologic modalities
for controling and preventing the incidence and consequences of orthostatic
hypotension.
References
1. Consensus statement on the
definition of orthostatic hypotension, pure autonomic failure, and multiple
system atrophy. The Consensus Committee of the American Autonomic Society and
the American Academy of Neurology. Neurology. 1996;46:1470.
2. Hale WA, Chambliss ML. Should
primary care patients be screened for orthostatic hypotension? J Fam Pract
. 1999;48:547-552.
3. Alli C, Avanzini F, Bettelli G, et
al. Prevalence and variability of orthostatic hypotension in the elderly.
Results of the ‘Italian study on blood pressure in the elderly (SPAA)'. The
‘Gruppo di Studio Sulla Pressione Arteriosa nell'Anziano'.
Eur Heart J. 1992;13:178-182.
4. Raiha I, Luutonen S, Piha J, et
al. Prevalence, predisposing factors, and prognostic importance of orthostatic
hypotension. Arch Intern Med. 1995;155:930-935.
5. Tilvis RS, Hakala SM, Valvanne J,
Erkinjuntti T. Postural hypotension and dizziness in a general aged
population: a four-year follow-up of the Helsinki Aging Study. J Am Geriatr
Soc. 1996;44:809-814.
6. Vara-Gonzalez L, Arauzo Alonso S,
Gonzalez Fernandez RM, et al. Reproducibility of postural changes of blood
pressure in hypertensive elderly patients in primary care. Blood Press Monit
. 2006;11:17-20.
7. Luukinen H, Airaksinen KE.
Orthostatic hypotension predicts vascular death in older diabetic patients.
Diabetes Res Clin Pract. 2005;67:163-166.
8. Rutan GH, Hermanson B, Bild DE, et
al. Orthostatic hypotension in older adults. The Cardiovascular Health Study.
CHS Collaborative Research Group. Hypertension. 1992;19(6 Pt 1):508-519.
9. Rose KM, Tyroler HA, Nardo CJ, et
al. Orthostatic hypotension and the incidence of coronary heart disease: the
Atherosclerosis Risk in Communities study. Am J Hypertens. 2000;13(6 Pt
1):571-578.
10. Luukinen H, Koski K, Laippala P,
Airaksinen KE. Orthostatic hypotension and the risk of myocardial infarction
in the home-dwelling elderly. J Intern Med. 2004;255:486-493.
11. Eigenbrodt ML, Rose KM, Couper
DJ, et al. Orthostatic hypotension as a risk factor for stroke: the
atherosclerosis risk in communities (ARIC) study, 1987-1996. Stroke.
2000;31:2307-2313.
12. Davis BR, Langford HG, Blaufox
MD, et al. The association of postural changes in systolic blood pressure and
mortality in persons with hypertension: the Hypertension Detection and
Follow-up Program experience. Circulation. 1987;75:340-346.
13. Masaki KH, Schatz IJ, Burchfiel
CM, et al. Orthostatic hypotension predicts mortality in elderly men: the
Honolulu Heart Program. Circulation. 1998;98:2290-2295.
14. Tinetti ME, Williams CS. The
effect of falls and fall injuries on functioning in community-dwelling older
persons. J Gerontol A Biol Sci Med Sci. 1998;53:M112-M119.
15. Smit AA, Halliwill JR, Low PA,
Wieling W. Pathophysiology basis of orthostatic hypotension in autonomic
failure. J Physiol. 1999;519 Pt 1:1-10.
16. Mathias CJ. Diseases of the
autonomic nervous system. In: Warrell DA, Cox TM, Firth JD, Benz EJ. Oxford
Textbook of Medicine. 4th ed. Oxford: Oxford University Press; 2003:13,
14, 24.
17. Wieling W, Wesseling KH.
Importance of reflexes in the circulatory adjustments to postural changes.
Cardiovascular reflex control in health and disease. W.B. Saunders: London;
1993:35-63.
18. Orthostatic hypotension and
syncope. The Merck Manual Online Medical Library. Section 16, chapter
200. Merch Research Laboratories. 2006. Available at: www.merck.com. Accessed
on December 1, 2006.
19. Engstrom JW, Aminoff MJ.
Evaluation and treatment of orthostatic hypotension. Am Fam Physician.
1997;56:1378-1384.
20. Mathias CJ. Orthostatic
hypotension: causes, mechanisms, and influencing factors. Neurology.
1995;45:S6-S11.
21. Hollister AS. Orthostatic
hypotension. Causes, evaluation, and management. West J Med.
1992;157:652-657.
22. Elmstahl S, Rosen I. Postural
hypotension and EEG variables predict cognitive decline: results from a 5-year
follow-up of healthy elderly women. Dement Geriatr Cogn Disord.
1997;8:180-187.
23. Mansoor GA. Orthostatic
hypotension due to autonomic disorders in the hypertensive clinic. Am J
Hypertension. 2006;19:319-326.
24. Robertson D, Davis TL. Recent
advances in the treatment of orthostatic hypotension. Neurology.
1995;45:S26-S32.
25. Sclater A, Alagiakrishnan K.
Orthostatic hypotension. A primary care primer for assessment and treatment.
Geriatrics. 2004;59:22-27.
26. Sahni M, Lowenthal DT, Meuleman
J. A clinical, physiology and pharmacology evaluation of orthostatic
hypotension in the elderly. Int Urol Nephrol. 2005;37:669-674.
27. Shannon JR, Diedrich A, Biaggioni
I, et al. Water drinking as a treatment for orthostatic syndromes. Am J Med
. 2002;112:355-360.
28. Handler J. Symptomatic
orthostatic hypotension/supine hypertension. J Clin Hypertens (Greenwich)
. 2005;7:612-616.
29. Basso A, Dalla Paola L, Erle G,
et al. Licorice ameliorates postural hypotension caused by diabetic autonomic
neuropathy. Diabetes Care. 1994;17:1356.
30. Midodrine. Clinical Pharmacology
2006. Available at: cp.gsm.com. Accessed on March 22, 2006.
31. Low PA, Gilden JL, Freeman R, et
al. Efficacy of midocrine vs. placebo in neurogenic orthostatic hypotension. A
randomized, double-blind multicenter study. Midocrine Study Group. JAMA
. 1997;277:1046-1051.
32. Fludrocortisone. Clinical
Pharmacology 2006. Available at: cp.gsm.com. Accessed on March 22, 2006.
33. Chobanian AV, Volicer L, Tifft
CP, et al. Mineralocorticoid-induced hypertension in patients with orthostatic
hypotension. N Engl J Med. 1979;301:68-73.
34. Dihydroergotamine. Clinical
Pharmacology 2006. Available at: cp.gsm.com. Last accessed on March 22, 2006.
35. Shibao C, Gamboa A, Diedrich A,
Biaggioni I. Management of hypertension in the setting of autonomic failure: a
pathophysiological approach. Hypertension. 2005;45:469-476.
36. Perera R, Isola L, Kaufmann H.
Effect of recombinant erythropoietin on anemia and orthostatic hypotension in
primary autonomic failure. Clin Auton Res. 1995;5:211-213.
37. Hoeldtke RD, Boden G, O'Dorisio
TM. Treatment of postprandial hypotension with a somatostatin analogue (SMS
201-995). Am J Med. 1986;81(6B):83-87.
38. Hoeldtke RD, Israel BC. Treatment
of orthostatic hypotension with octreotide. J Clin Endocrinol Metab.
1989;68:1051-1059.
39. Robertson D, Goldberg MR,
Hollister AS, et al. Clonidine raises blood pressure in severe idiopathic
orthostatic hypotension. Am J Med. 1983;74:193-200.
40. Singer W, Opfer-Gehrking TL,
McPhee BR, et al. Acetylcholinesterase inhibition: a novel approach in the
treatment of neurogenic orthostatic hypotension. J Neurol Neurosurg
Psychiatry. 2003;74:1294-1298.
41. Singer W, Sandroni P,
Opfer-Gehrking TL, et al. Pyridostigmine treatment trial in neurogenic
orthostatic hypotension. Arch Neurol. 2006;63:513-518.
42. Fotherby MD, Potter JF.
Orthostatic hypotension and anti-hypertensive therapy in the elderly.
Postgrad Med J. 1994;70:878-881.
43. Poon IO, Braun U. High prevalence
of orthostatic hypotension and its correlation with potentially causative
medications among elderly veterans. J Clin Pharm Ther. 2005;30:173-178.
To comment on this article, contact editor@uspharmacist.com.






