ABSTRACT: Atrial fibrillation is one of the most common forms of cardiac disturbances, diagnosed in an estimated 2.7 million to 6.1 million people in the United States, with prevalence increasing with age. Based on the individual’s symptoms, medical treatment is available to help control the heart’s rate and rhythm and reduce the risk of clots or strokes. Successful strategies to restore and maintain sinus rhythm have been shown to improve symptoms and quality of life. However, selection of antiarrhythmic drugs should be based on underlying heart disease and comorbidities, due to drug interactions, adverse reaction profile, and monitoring requirements with antiarrhythmic agents. Pharmacists can play a vital role in therapy selection and recommendations for continuation of therapy.
US Pharm. 2020;45(2):24-27.
Atrial fibrillation (AF) is one of the most common forms of cardiac disturbances, diagnosed in an estimated 2.7 million to 6.1 million people in the United States, with prevalence increasing with age.1,2 AF is an arrhythmia or irregular heartbeat with uncoordinated atrial activation and ineffective atrial contraction, leading to inadequate blood flow into the ventricles.1,2 Symptoms may include irregular heartbeat, heart palpitations, extreme fatigue, shortness of breath, and chest pain.2 AF is associated with an increased risk of stroke, heart failure, dementia, hospitalization, and death.1 As the death rate has risen over the past two decades, the cost of AF in the U.S. has also increased, with annual medical costs more than $8,000 per patient higher when compared with those without AF.1-5
A multipronged approach is needed for the management of AF. Based on the individual’s symptoms, medical treatment is available to help control the heart’s rate (rate control) and rhythm (rhythm control) and to reduce the risk of clots or stroke (anticoagulants).1,2
ANTIARRHYTHMIC MANAGEMENT
Successful strategies to restore and maintain sinus rhythm have been shown to improve symptoms and quality of life; however, these strategies don’t improve survival or reduce the risk of stroke.1,6,7 The Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) study found no evidence to support the prevention of stroke or improvement in mortality with rhythm control when compared with a rate-control strategy. Also, a rhythm-control strategy may be associated with more hospitalizations, warranting its use only in appropriate candidates.1 Persistent symptoms associated with AF, difficulty achieving adequate rate control, younger patients, first episode of AF, tachycardia-mediated cardiomyopathy, and AF precipitated by an acute illness may indicate the need for rhythm-control management.1 Early intervention with rhythm-control strategy to prevent progression of AF has also been shown to be beneficial.1,8-10
ANTIARRHYTHMIC AGENTS
Selection of antiarrhythmic drugs should be based on underlying heart disease and comorbidities, and therapy should be guided first by safety and then by efficacy.1
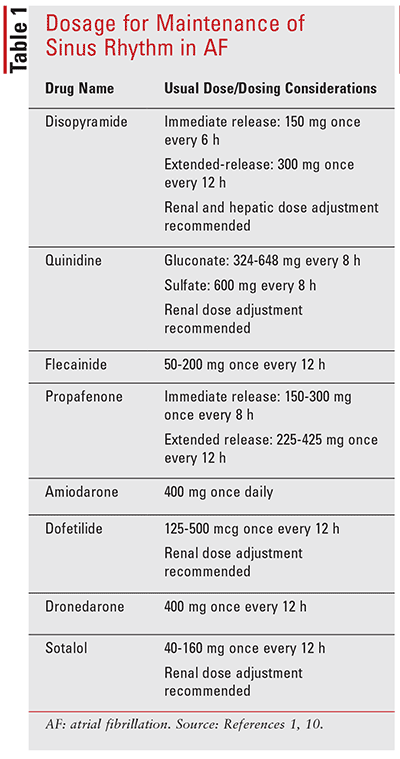
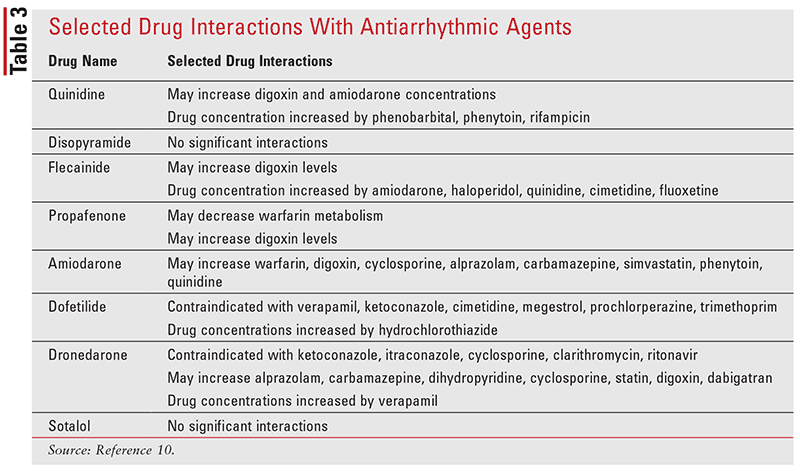
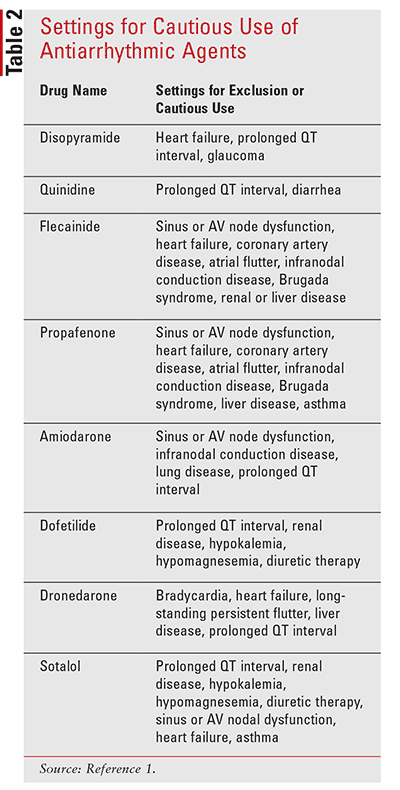
Many of these agents affect ion channels and adrenergic receptors, causing a range of cardiac and noncardiac side effects.10 However, antiarrhythmic agents may reduce frequency and duration of AF and improve the quality of life.1 Table 1 provides an overview of the typical dosing of antiarrhythmic agents used for the maintenance of sinus rhythm in AF. Important factors to consider when selecting an antiarrhythmic agent (Table 2) include whether patients have coronary artery disease, heart failure, left ventricular hypertrophy, risk of bradyarrhythmias or excessive QT prolongation, history of torsades de pointes, and influences on drug disposition, such s age, renal/hepatic dysfunction, and drug interactions (Table 3).1,10
CLASS
Antiarrhythmic agents work by blocking the sodium, potassium, and calcium channels or the adrenergic receptors. The Vaughan Williams classification system defines where the arrhythmic agents work: Class I agents are sodium-channel blockers, class II are beta-blockers, class III are potassium-channel blockers, and class IV are calcium-channel blockers.11
Vaughan Williams Class IA
Quinidine and disopyramide are class IA antiarrhythmic agents that work on the sodium channel. The use of these agents has decreased and is not recommended as first-line. Although quinidine was one of the first antiarrhythmic agents used in AF, it is now reserved as an alternative treatment when newer antiarrhythmics are contraindicated.1 Quinidine may prolong QT interval and can cause torsades de pointes. Disopyramide can also prolong QT interval and is avoided in structural heart disease. Although disopyramide is useful in a high vagal tone setting, there is little evidence available to help support this use.1 A systematic review also suggested that quinidine and disopyramide may increase mortality.12
Vaughan Williams Class IC
Flecainide and propafenone are class IC agents working on the sodium channel. Due to results from the Cardiac Arrhythmia Suppression Trial (CAST) study, these agents are not recommended in patients with concomitant left ventricular dysfunction and coronary artery disease. Flecainide is very effective in treating AF and premature ventricular contractions that are triggered in patients with structurally normal hearts; use is not recommended in those with structural heart disease. In addition, the chance of proarrhythmic toxicity increases with exertion when heart rate increases.11 Propafenone is indicated for more severe or life-threatening ventricular arrhythmias and should be used in conjunction with atrioventricular nodal blocking agents because of the possibility of conversion to atrial flutter. The absorption of both agents may be altered with food, especially with milk for flecainide.11
Vaughan Williams Class III
Amiodarone, dofetilide, dronedarone, and sotalol are class III agents, potassium channel blockers that lead to prolongation of QT interval and possible ventricular arrhythmias or torsades de pointes. Amiodarone is considered the most potent antiarrhythmic agent.11
The adverse event profile with amiodarone can be intolerable, with gastrointestinal effects the most common side effect.11,13 Other side effects that may occur include pulmonary inflammation, thyroid abnormalities, retinal deposits, and congestive and inflammatory hepatopathy.11 Amiodarone may also cause a decrease in pro-arrhythmic effects and effectiveness in maintenance of sinus rhythm compared with other antiarrhythmic agents.14 Dronedarone, an analogue of amiodarone, is less efficacious than amiodarone but has an improved side effect profile. Sotalol and dofetilide are effective in the maintenance of sinus rhythm, but inpatient loading is recommended. Both have also been shown to be effective in preventing arrhythmia in patients with underlying structural heart disease.11 Of all antiarrhythmic agents, dofetilide and amiodarone have been proven safe in patients with heart failure.11
ROLE OF PHARMACISTS
Pharmacists may play a vital role in the management of AF patients taking antiarrhythmics. Discussing adverse events and administration requirements, reviewing drug interactions, and recommending monitoring parameters are just a few of the key responsibilities. A retrospective chart review was conducted to show the impact of a pharmacy-based outpatient clinic for patients receiving antiarrhythmic medications.15 The research showed that pharmacist monitoring increased patient adherence to required monitoring/testing parameters postdischarge, detected unrecognized adverse events such as pulmonary function decline, QT prolongation, and lab abnormalities in 38% of visits, and identified clinically significant drug interactions. A similar retrospective cohort study with a pharmacy-cardiology collaborative program was conducted to assess antiarrhythmic management and also concluded significantly more clinical interventions (P <.01) in the postintervention program due to dose adjustments or discontinuation, monitoring requirements, and electrolyte supplementation.16
CONCLUSION
AF is the most common arrhythmia, with prevalence increasing with age; multiple agents are available for its management. Although data are unavailable to support favorable morbidity and mortality outcomes, restoration of sinus rhythm may prevent the progression of AF and improve the quality of life in early intervention. However, due to the nature of the side effects or monitoring requirements of antiarrhythmics, therapy selection should be based first upon safety and then upon efficacy.
REFERENCES
1. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol. 2014;64(21):e1-e76.
2. CDC. Atrial fibrillation fact sheet. 2017; www.cdc.gov/dhdsp/data_statistics/fact_sheets/fs_atrial_fibrillation.htm. Accessed November 28, 2019.
3. Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics—2015 update: a report from the American Heart Association. Circulation. 2015;131:e29-e322.
4. Agency for Healthcare Research and Quality. Weighted national estimates. HCUP national inpatient sample [online]. 2012. http://hcupnet.ahrq.gov/HCUPnet.jsp. Accessed November 28, 2019.
5. CDC. About multiple cause of death 1999–2011. CDC WONDER Online Database. 2014. http://wonder.cdc.gov/mcd-icd10.html. Accessed December 1, 2019.
6. Corley SD, Epstein AE, DiMarco JP, et al; AFFIRM Investigators. Relationships between sinus rhythm, treatment, and survival in the Atrial Fibrillation Follow-Up Investigation of Rhythm Management (AFFIRM) Study. Circulation. 2004;109:1509-1513.
7. Hagens VE, Ranchor AV, Van Sonderen E, et al. Effect of rate or rhythm control on quality of life in persistent atrial fibrillation: results from the Rate Control Versus Electrical Cardioversion (RACE) Study. J Am Coll Cardiol. 2004;43:241-247.
8. Kirchhof P, Bax J, Blomstrom-Lundquist C, et al. Early and comprehensive management of atrial fibrillation: executive summary of the proceedings from the 2nd AFNET-EHRA consensus conference ‘research perspectives in AF.’ Eur Heart J. 2009;30:2969-2977c.
9. Van Gelder IC, Haegeli LM, Brandes A, et al. Rationale and current perspective for early rhythm control therapy in atrial fibrillation. Europace. 2011;13:1517-1525.
10. Zimetbaum P. Antiarrhythmic drug therapy for atrial fibrillation. Circulation. 2012;125:381-389.
11. Barekatain A, Razavi M. Antiarrhythmic therapy in atrial fibrillation: indications, guidelines and safety. Tex Heart Inst J. 2012;39(4):532-534.
12. Lafuente-Lafuente C, Mouly S, Longas-Tejero MA, et al. Antiarrhythmic drugs for maintaining sinus rhythm after cardioversion of atrial fibrillation: a systematic review of randomized controlled trials. Arch Intern Med. 2006;166:719-728.
13. Doyle JF, Ho KM. Benefits and risks of long-term amiodarone therapy for persistent atrial fibrillation: a meta-analysis. Mayo Clin Proc. 2009;84(3):234-242.
14. Singh BN, Singh SN, Reda DJ, et al, for the Sotalol Amiodarone Atrial Fibrillation Efficacy Trial (SAFE-T) Investigators. Amiodarone versus sotalol for atrial fibrillation. N Engl J Med. 2005;352(18):1861-1872.
15. Snider M, Kalbfleisch S, Carnes CA. Initial experience with antiarrhythmic medication monitoring by clinical pharmacists in an outpatient setting: a retrospective review. Clin Ther. 2009;31(6):1209-1218.
16. Kibert JL, Franck JB, Dietrich NM, et al. Impact of a pharmacy-cardiology collaborative management program during initiation of antiarrhythmic drugs. J Am Coll Clin Pharm. 2019.
To comment on this article, contact rdavidson@uspharmacist.com.