US Pharm. 2022;47(3):18-24.
ABSTRACT: Rheumatoid arthritis is among the most common arthritic diseases, with 71 cases diagnosed per 100,000 people. To alleviate symptoms and reduce long-term damage, treatment options available to patients include biologics, particularly adalimumab (Humira)—a tumor necrosis factor inhibitor. The drug has played a vital role in the treatment of rheumatoid arthritis over the years, but recently adalimumab-adbm (Cyltezo) was developed as the first interchangeable biosimilar that mimics the structure and function of the original product.
According to the National Institutes of Health, autoimmune and inflammatory diseases have affected more than 24 million people in the United States since 2005.1 These conditions are caused by the immune system’s inability to differentiate true foreign invaders. Therefore, the immune system mistakes parts of the body as threats and releases autoantibodies to destroy the healthy cells. Although the cause for these disease states remains a mystery, medications and new therapeutic guidelines have emerged over time to combat these conditions.
Rheumatoid arthritis (RA), for instance, is one of the most prevalent autoimmune disorders in the U.S. Although there is currently no cure for this disease, the goal is to achieve a state of remission in which symptoms are less likely to occur. Adalimumab (Humira), introduced in 2002, was one of the many common therapies used to reduce signs and symptoms of RA.2 Despite providing relief to a large number of patients over the years, there were consistent barriers that prevented individuals from accessing appropriate care.
The FDA recently approved the first interchangeable biosimilar to adalimumab, creating a major breakthrough in the realm of autoimmune disease treatments.
Rheumatoid Arthritis
Although the cause of autoimmune and inflammatory diseases remains unknown, there are more than 80 clinical diseases that originate from an autoimmune response.3 The variety of diseases stems from the fact that autoimmune disorders can affect any tissue or organ in the body. Hence, the range of symptoms between each disease state can differ, with some being mild and manageable while others are debilitating and life-threatening. An example of this is the common condition RA, an autoimmune and inflammatory disease in which the immune system attacks healthy cells, causing inflammation mainly in the joints of the body.4 The synovium, the tissue around the joint that produces fluid allowing for smooth movement, is especially damaged, and the inflamed area makes the joint sore and painful, causing difficult movement.5,6 The most common joints affected by this condition are the hands, knees, and wrists, but in more severe cases, this form of arthritis can also affect other parts of the body, such as the lungs, eyes, and heart.5,6 Internal organs that are affected become subject to persistent tissue damage, and RA can cause chronic pain or severe deformities.
In the U.S., approximately 1.3 million patients suffer from RA.7 While the number of cases continues to surge, the total healthcare costs increase as well.7 The objective of ensuring that all patients have affordable access to treatment that can relieve their symptoms and improve their quality of life remains unmet. In most cases, the treatment goal, at a minimum, is to reduce inflammation and swelling while improving joint function. However, the end result that clinicians hope for is to slow the progression of the disease and reduce further damage to the affected areas.6
Management of RA
Currently, there are three main classes of therapeutic options available to treat RA: nonsteroidal anti-inflammatory agents (NSAIDs), corticosteroids, and disease-modifying antirheumatic drugs (DMARDs).5,6 NSAIDs and corticosteroids are used to relieve pain and help mitigate inflammation at the site of injury. Side effects most commonly seen with the use of NSAIDs are stomach and kidney damage. For patients who have localized gastric side-effect concerns, cyclooxygenase 2 (COX-2) anti-inflammatory agents are seen as an alternative. These agents restrain COX-2 enzyme and decrease the number of prostaglandins.5 In doing so, prostaglandins that are protective of the stomach and kidney remain, and inflammation is reduced. Meanwhile, low-dose corticosteroids can be utilized to decrease the amount of inflammation and suppress flares by regulating the autoimmune system.4
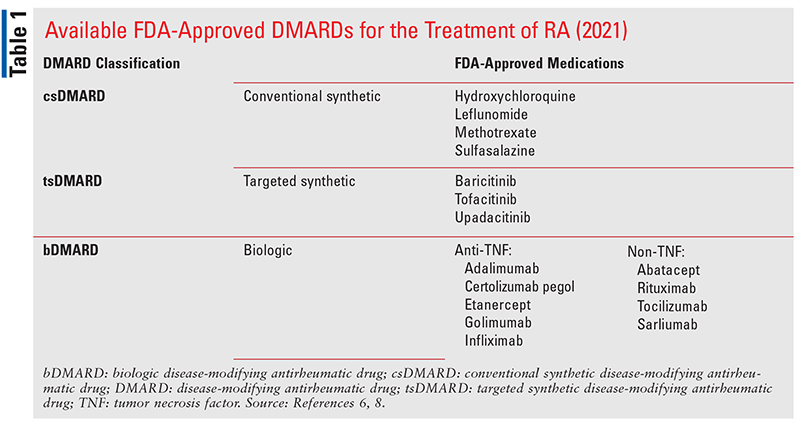
DMARDs are a major class of drugs that halt the progression of disease by altering the immune system. There are two categories in this class: traditional DMARDs and biologics. The severity of the ailment, side effects, and patient preferences all play a role in medicine selection. While traditional DMARDs have been used extensively throughout treatment, biologics are becoming increasingly popular. Biologics are produced using recombinant DNA and prevent or reduce inflammation in the damaged sites.8 They attack more specific inflammation-causing cells and, therefore, are considered more effective.8 One example of a biologic drug used for RA is adalimumab (Humira). TABLE 1 shows all of the available FDA-approved DMARDs for the treatment of RA.6,8
Adalimumab
Adalimumab is a registered trademark of AbbVie Biotechnology Ltd.9 Adalimumab is a tumor necrosis factor (TNF) blocker indicated for an array of conditions, such as moderate-to-severe RA, juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, Crohn’s disease, ulcerative colitis, plaque psoriasis, hidradenitis supprativa, and uveitis.2 Its mechanism of action revolves around TNF, a naturally occurring cytokine involved in normal inflammatory and immune responses.2 Elevated levels of TNF are seen in many autoimmune and inflammatory diseases and play a role in pathological inflammation and joint destruction.2 Adalimumab binds to TNF-alpha and blocks its interaction with TNF receptors on the surface of p55 and p75 cells.2 Specifically, for RA, adalimumab works by inhibiting the progression of structural damage and improving physical function in adult patients. It can be used alone or in combination with a nonbiologic DMARD.2 Adalimumab is administered as an SC injection, and the dose varies depending on the patient’s condition, weight, and age. The most common adverse effects associated with adalimumab are injection-site reactions, infections, headache, and rash.2
Adalimumab-adbm
On October 15, 2021, the FDA approved the first interchangeable biosimilar to adalimumab, called adalimumab-adbm (Cyltezo). A biosimilar is a biological product that carries no clinical difference from the already-approved medication but includes similar components.10 The idea behind creating interchangeable biosimilars is to offer products that can be substituted for the reference product without the worry of a change in clinical efficacy. Adalimumab-adbm was originally approved in August 2017 for the treatment of multiple chronic inflammatory diseases.11 However, in October 2021, the FDA granted its approval through the supplemental Biologics License Application to Boehringer Ingelheim.9 The new biosimilar falls within its mission’s intention by providing an alternative therapeutic option to help with many chronic inflammatory conditions. Both products share the same clinical results, including expected boxed warnings. The approval of these interchangeable biosimilars creates increased access to treatment options for patients with serious medical conditions.4
Adalimumab-adbm is also a TNF inhibitor and is approved to treat the following conditions: moderate-to-severe active RA, psoriatic arthritis, ankylosing spondylitis, moderate-to-severe active Crohn’s disease, moderate-to-severe active ulcerative colitis, moderate-to-severe chronic plaque psoriasis, moderate-to-severe active polyarticular juvenile idiopathic arthritis in patients aged 2 years and older, and pediatric patients aged 6 years and older with Crohn’s disease.10,12 The formulation is a single-dose, prefilled glass syringe with two available strengths: 40 mg/0.8 mL and 20 mg/0.4 mL.12 Adalimumab-adbm works in the same way as adalimumab; both bind specifically to TNF-alpha and block interactions with TNF receptors on the p55 and p75 cell surfaces and lyse in vitro–expressed surface TNFs.12 Likewise, treatment can reduce the thickness of the epidermis and cause infiltration of inflammatory cells.12 Infections, injection-site reactions, headache, and rash are all common side effects.11,12
Adalimumab-adbm represents the first interchangeable monoclonal antibody. With its approval, the FDA’s Purple Book, the official directory of all approved biosimilars and interchangeables, is updated.13 Adalimumab-adbm will not be available until July 2023, but the therapeutic impact could be staggering.9 Replacing benchmark products with biosimilars could further ease the financial burden of patients. As the chairman of the Division of Dermatology at Baylor University Medical Center exclaimed, “As the first interchangeable biosimilar of adalimumab, Cyltezo (adalimumab-adbm) represents an important step toward bringing patients more affordable treatment options for complex, and often expensive, biologic reference products.”9
VOLTAIRE-RA Trial
The VOLTAIRE-RA trial was completed in 2015, with the objective to prove the clinical equivalence of adalimumab-adbm and adalimumab. In a test group of 645 patients with clinically diagnosed active RA, this study used a double-blind, randomized, parallel-group intervention approach.14 It was noted that the patients were stable on methotrexate and randomized to continue with either the biosimilar adalimumab-adbm or adalimumab within a 24-hour treatment time frame.14 The primary endpoint was illustrated through the percentage of patients achieving the American College of Rheumatology 20% response criteria (ACR20) at Weeks 12 and 24.14 Efficacy, safety, and immunogenicity were also observed up to Week 58 of treatment.14
Results show that 67.0% (n = 324) of those who received adalimumab-adbm and 61.1% (n = 321) of those who received adalimumab achieved ACR20 (90% CI 0.9-12.7).14 Meanwhile, at the 24th week, 69.0% and 64.5% (95% CI 3.4-12.5) were achieved, respectively.14 Changes in mean response rates for ACR20 were similar when re-randomized at 24 weeks.14 The significance of this evidence indicates a positive outcome for the therapeutic equivalence of the two agents. Likewise, safety, tolerability, and immunogenicity were similar between the two treatment groups.
VOLTAIRE-RAext Trial
A phase IIIb VOLTAIRE-RAext trial, based on previous studies, supported the basis of adalimumab-adbm and its indications for the treatment of RA. This study was conducted for 48 weeks using an open, multicenter interventional approach.15 The sample size was 430 adults with a diagnosis of moderate-to-severe RA who wished to continue treatment with adalimumab-adbm.15 These patients were extracted from their participation in the VOLTAIRE-RA trial. The patients received a prefilled, 40-mg syringe of adalimumab-adbm and administered it every 2 weeks.15 The primary endpoint was the number of patients with adverse drug reactions from the start of the trial to the end of the 10-week follow-up period.15
Results were divided into three groups: continuous administration of adalimumab-adbm, conversion from adalimumab reference product (RP) to adalimumab-adbm, and continuous administration of adalimumab RP in the previous study.15 Among these groups, 20.2% experienced ³1 drug-related adverse effect, with a lower proportion of 17.6% in the group with the switched therapy.15 Secondary endpoints were investigated to assess efficacy using disease activity score 28, erythrocyte sedimentation rate, and achievement of the ACR/European League Against Rheumatism response.15,16 Expansion of this study did not lead to significant new findings in terms of efficacy, safety, or immunogenicity.15,16
VOLTAIRE-X Trial
The positive data from the VOLTAIRE-X phase III study demonstrated that interchangeability was achieved. The VOLTAIRE-X trial was designed to assess the pharmacokinetic similarity between patients who regularly took adalimumab and those who alternated between the biosimilar adalimumab-adbm and adalimumab.11,16 The treatment groups were clinically diagnosed with moderate-to-severe chronic plaque psoriasis and received 48 weeks of treatment with a 10-week follow-up period.17 A double-blind, randomized, parallel intervention model was used in this study.17 There were 259 enrolled participants between ages 18 and 80 years.17 The primary endpoint was demonstrated by the pharmacokinetics measurements of adalimumab over a dosing interval of Weeks 30 to 32: area under the plasma concentration (AUC) and maximum observed concentration (Cmax).17
Secondary endpoints were assessed through the efficacy, safety, and immunogenicity profiles, and other pharmacokinetic measurements, such as minimum observed plasma concentration (Cmin) and time to maximum observed plasma concentration (Tmax).17 The results supported the primary and secondary endpoints. There were three endpoint-reporting groups: run-in period with adalimumab (loading dose on first day), treatment switching between adalimumab and adalimumab-adbm, and continued administration of adalimumab.17 Equivalence was estimated based on a 90.2% CI.16 This interval corresponds to the AUC and Cmax tests, and CI was calculated based on the ratio of switching treatments to continuous dosing.17 The results show that both the primary and secondary endpoints were satisfied. Adalimumab-adbm was very similar to adalimumab in terms of safety, efficacy, and immunogenicity. This study, along with previous studies, showed that adalimumab-adbm is a biosimilar and adalimumab is therapeutically equivalent.
The three trials elaborated here—VOLTAIRE-RA, VOLTAIRE-RAext, and VOLTAIRE-X—all demonstrated supporting evidence for the efficacy and safety profiles of adalimumab-adbm for RA patients. In addition, the clinical trials also attested to adalimumab-adbm being an interchangeable biosimilar. However, it is noted that there are other clinical trials reviewing adalimumab-adbm’s role and effectiveness compared with adalimumab in other disease states, such as Crohn’s (VOLTAIRE-CD).18
Conclusion
With positive evidence exhibited in various studies, adalimumab-adbm is the first interchangeable monoclonal antibody. This citrate-free adalimumab-adbm has been proven to exude the same efficacy and safety as the original medication. The introduction of the first interchangeable adalimumab biosimilar offers an alternative and inexpensive option for many and could potentially lead to other scientific breakthroughs.
REFERENCES
1. Xu Y, Wu Q. 2021. Prevalence Trend and Disparities in Rheumatoid Arthritis among US Adults, 2005–2018. Journal of Clinical Medicine, [online] 10(15):3289. www.mdpi.com/2077-0383/10/15/3289/pdf.
2. Accessdata.fda.gov. 2021. HUMIRA® (adalimumab) injection. [online] www.accessdata.fda.gov/drugsatfda_docs/label/2021/125057s417lbl.pdf. Accessed December 15, 2021.
3. National Institute of Environmental Health Sciences: Health and Education. Autoimmune diseases. 2021. www.niehs.nih.gov/health/topics/conditions/autoimmune/index.cfm, Accessed December 10, 2021.
4. CDC. Rheumatoid arthritis. 2021. www.cdc.gov/arthritis/basics/rheumatoid-arthritis.html. Accessed December 10, 2021.
5. American College of Rheumatology. Rheumatoid Arthritis. www.rheumatology.org/I-Am-A/Patient-Caregiver/Diseases-Conditions/Rheumatoid-Arthritis. Accessed November 29, 2021.
6. Fraenkel L, Bathon JM, England BR, et al. 2021 American College of Rheumatology Guideline for the Treatment of Rheumatoid Arthritis. Arthritis Rheumatol. 2021;73(7):1108-1123.
7. Myasoedova E, Crowson CS, Kremers HM, et al. Is the incidence of rheumatoid arthritis rising?: results from Olmsted County, Minnesota, 1955-2007. Arthritis Rheum. 2010;62(6):1576-1582.
8. Cohen S, Cannella A, 2021. Patient education: disease-modifying antirheumatic drugs (DMARDs) in rheumatoid arthritis (beyond the basics). Uptodate.com. www.uptodate.com/contents/disease-modifying-antirheumatic-drugs-dmards-in-rheumatoid-arthritis-beyond-the-basics#H17. Accessed December 20, 2021.
9. FDA. U.S. FDA approves Cyltezo® (adalimumab-adbm) as first interchangeable biosimilar with Humira. www.boehringer-ingelheim.us/press-release/us-fda-approves-cyltezo-adalimumab-adbm-first-interchangeable-biosimilar-humira. 2021. Accessed November 5, 2019.
10. FDA. FDA approves Cyltezo, the first interchangeable biosimilar to Humira. www.fda.gov/news-events/press-announcements/fda-approves-cyltezo-first-interchangeable-biosimilar-humira. 2021. Accessed November 29, 2021.
11. NIH. The Autoimmune Diseases Coordinating Committee. Progress in autoimmune diseases research: report to Congress. www.niaid.nih.gov/sites/default/files/adccfinal.pdf. March 1, 2005. Accessed November 20, 2021.
12. FDA. CYLTEZOTM (adalimumab-adbm) injection. www.accessdata.fda.gov/drugsatfda_docs/label/2021/125057s417lbl.pdf. 2021. Accessed December 15, 2021.
13. FDA. Biosimilar and interchangeable biologics: more treatment choices. www.fda.gov/consumers/consumer-updates/biosimilar-and-interchangeable-biologics-more-treatment-choices.2021. Accessed November 29, 2021.
14. Cohen SB, Alonso-Ruiz A, Klimiuk PA, et al. Similar efficacy, safety and immunogenicity of adalimumab biosimilar BI 695501 and Humira reference product in patients with moderately to severely active rheumatoid arthritis: results from the phase III randomised VOLTAIRE-RA equivalence study. Ann Rheum Dis. 2018;77(6):914-921.
15. Cohen SB, Czeloth N, Lee E, et al. Long-term safety, efficacy, and immunogenicity of adalimumab biosimilar BI 695501 and adalimumab reference product in patients with moderately-to-severely active rheumatoid arthritis: results from a phase 3b extension study (VOLTAIRE-RAext). Expert Opin Biol Ther. 2019 Oct;19(10):1097-1105. Epub 2019 Aug 6.
16. Stanley B, Cohen NC, Czeloth N, et al. Long-term safety, efficacy, and immunogenicity of adalimumab biosimilar BI 695501 and adalimumab reference product in patients with moderately-to-severely active rheumatoid arthritis: results from a phase 3b extension study (VOLTAIRE-RAext). Expert Opin Biol Ther. 2019:10:1097-1105.
17. Clinicaltrials.gov. The VOLTAIRE-X Trial Looks at the Effect of Switching Between Humira® and BI 695501 in Patients With Plaque Psoriasis - Full Text View. clinicaltrials.gov/ct2/show/NCT03210259 Accessed December 20, 2021.
18. Hanauer S, Liedert B, Balser S, et al. Safety and efficacy of BI 695501 versus adalimumab reference product in patients with advanced Crohn’s disease (VOLTAIRE-CD): a multicentre, randomised, double-blind, phase 3 trial. The Lancet Gastroenterology & Hepatology. 2021;6(10):816-825.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






