US Pharm. 2012;37(11):HS-23-HS-26.
ABSTRACT: Primary joint replacement surgery is commonly performed in patients with various arthropathies. A rare but devastating complication of these procedures is the development of a prosthetic joint infection. Use of prophylactic antibiotics has been shown to reduce the risk of infection. For most patients, current guidelines recommend cefazolin or cefuroxime administered IV within 60 minutes of skin incision as prophylaxis during hip or knee arthroplasty. Prophylaxis should be discontinued within 24 hours of the procedure. Local delivery of antibiotics via antibiotic-loaded bone cement (ALBC) has proven beneficial in the treatment of joint infections, but current guidelines do not recommend the use of ALBC for prophylaxis. Appropriate antibiotic selection, timing, and duration of antimicrobial prophylaxis are essential components of antimicrobial stewardship and have been included as performance measures in the Surgical Care Improvement Project.
Primary joint replacement, or arthroplasty, is commonly performed to relieve disabling joint pain in patients experiencing various arthropathies.1 In 2003, more than 200,000 primary hip arthroplasties and more than 400,000 primary knee arthroplasties were performed.2 As the population ages, it is predicted that demand for both procedures will rise—a 174% increase in the number of hip arthroplasties and a 673% increase in the number of knee arthroplasties—by the year 2030.2
One of the most devastating complications of these procedures is the development of a prosthetic joint infection. The rate of infection after primary arthroplasty ranges from 1% to 3% in low-risk patients, but increases to 8% to 13% in patients with risk factors such as diabetes, rheumatoid arthritis (RA), immunosuppression, and others.3,4 Patients with prosthetic joint infections commonly require revision surgery to remove the infected prosthesis, along with prolonged antibiotic courses.5 Medical costs associated with revision procedures are almost five times that of a primary arthroplasty, with a longer duration of hospitalization, more complications, and more outpatient visits.6
The benefit of prophylactic antibiotics used preoperatively was demonstrated in trials conducted in the 1970s and 1980s.7,8 Prophylactic antibiotics reduce the absolute risk of postoperative infection by 8%, with a number needed to treat of only 13 people to prevent one infection.9 This review will focus on the appropriate antibiotic selection, timing, and duration of prophylaxis in primary arthroplasty, including a brief review of prophylactic use of antibiotic-loaded bone cement (ALBC) and a discussion of current regulatory initiatives to improve appropriate preoperative care.
Antibiotic Prophylaxis
The goals of prophylaxis are to prevent postoperative infection at the surgical site, prevent postoperative morbidity and mortality, and reduce the duration and cost of health care.10 Surgical-site infections account for nearly 25% of all nosocomial infections each year and increase hospital readmissions and death.11 Prophylactic antimicrobials must be active against the organisms most likely to cause a postoperative infection, maintain adequate concentrations that exceed the minimum inhibitory concentration (MIC) at the contamination site for the duration of the procedure, be safe for the patient at the recommended dosage, minimize the development of resistance, and be cost effective.10,12
Spectrum of Activity
Prior to choosing an antimicrobial for prophylaxis, it is imperative to identify common microorganisms that may be encountered during surgery. In joint arthroplasty, microorganisms that colonize the skin, predominantly Staphylococcus aureus (35%) and Staphylococcus epidermidis (40%), are those most commonly encountered. Other less commonly identified bacteria include Enterococcus, gram-negative bacilli like Escherichia coli, and Pseudomonas.3 Antibiotics chosen for prophylaxis do not have to be active against all organisms that may be encountered, but they should have reliable activity against those most commonly encountered.12
Antibiotic Selection
Surgical-prophylaxis guidelines published by the American Society of Health-System Pharmacists and the American Academy of Orthopaedic Surgeons recommend cefazolin or cefuroxime as first-line antimicrobial prophylaxis for hip or knee arthroplasty.10,11 These cephalosporins have reliable activity against gram-positive organisms and clinically relevant gram-negative bacilli (E coli), with the exception of methicillin-resistant S aureus (MRSA) and Enterococcus. Both agents have demonstrated excellent penetration into the bone, synovium, and muscle within minutes of infusion.13,14 They also exhibit bactericidal activity through inhibition of cell-wall synthesis, which is a favorable characteristic for antimicrobials used for prophylaxis.12 Both antimicrobials pose a low risk to patients when administered for a short duration. No significant difference has been found in infection rates between the two cephalosporins, but most studies in the United States used cefazolin.9,12
Dosages used for prophylaxis are outlined in TABLE 1. Antibiotic doses should be repeated if the operation continues beyond two half-lives after the initial dose, to ensure concentrations above the MIC for the duration of surgery. Cefazolin should be redosed intraoperatively every 2 to 5 hours, and cefuroxime every 3 to 4 hours.12,15
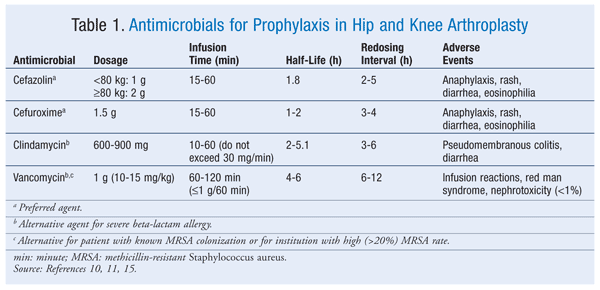
In patients with an established severe or life-threatening anaphylactic allergy to penicillins or cephalosporins, the recommended alternatives are clindamycin and vancomycin. Clindamycin is preferred unless the patient has a known history of MRSA colonization or the institution has a high rate of MRSA.10,11 MRSA and S epidermidis infections have been on the rise, with some facilities reporting a prevalence of up to 66%.12 According to the expert consensus, a MRSA rate exceeding 20% of all institutional isolates is considered high.10 Despite this recommendation, trials conducted in institutions with MRSA rates greater than 20% have consistently demonstrated that there is no difference between cefazolin and vancomycin prophylaxis in the rate of surgical-site infections.1,16 One study reported that 138 patients would need to be treated with vancomycin instead of cefazolin to prevent one MRSA infection in a hospital with 30% MRSA prevalence.17 These data have called into question whether the use of prophylactic vancomycin is necessary in institutions with high MRSA rates. Excessive use of vancomycin leading to antimicrobial resistance is a reason for judicious use of this antibiotic. With the emergence of vancomycin-intermediate S aureus and rare vancomycin-resistant strains, it is imperative to ensure appropriate use of this antimicrobial to prevent further resistance from developing.
Timing of Antibiotic Therapy
Antibiotics should be administered within 60 minutes prior to skin incision.10,11 The exception is vancomycin, which should be administered within 2 hours of incision to ensure adequate infusion time. An early trial found that the crucial time for antibacterial activity is between incision and 3 hours postincision.18 Antimicrobials administered after this period are less effective. A study comparing preoperative (0-2 h prior to incision), perioperative (during the 3 h after incision), and postoperative (3-24 h after incision) antibiotics found significantly fewer infections in patients who received antimicrobials preoperatively.19
Duration of Antibiotic Therapy
Antimicrobial prophylaxis should be discontinued within 24 hours after the operation.10,11 In a trial comparing 1 day versus 3 days of cefazolin, the infection rate was identical (0.6%).20 In another trial, a single preoperative dose of cefazolin prevented infection just as well as 1 day of cefuroxime.21 Since judicious use of antibiotics is imperative to reduce the risk of antimicrobial resistance, antimicrobials should be used for the shortest duration possible without increasing the risk of infection. In orthopedic procedures requiring drain placement, there is no evidence suggesting that antimicrobials should be continued until all drains are removed; thus, antibiotics should be discontinued within 24 hours.11
ALBC
Bone cement is a polymer-based material often used in arthroplasty for fixation of the joint implant to the bone.5 The addition of antibiotics to bone cement has been a treatment modality for established joint infections for more than 30 years.22 Biomaterials used in joint arthroplasty increase the risk of bacterial adhesion because of polarity or surface roughness.4 Once bacteria adhere to the surface of the implant, they can encase themselves in a polysaccharide matrix (biofilm), making them less susceptible to antibiotics.4 Local antibiotic delivery using ALBC has proven beneficial in the treatment of established infections, but prophylactic use is not as well defined. Common antibiotics added to cement include gentamicin, tobramycin, and cefuroxime.4 For prophylactic use, a low dose of antibiotic is added to the cement—up to 2 g antibiotic in 40 g cement.5 Once implanted, the flow of physiological fluids throughout the polymer causes antibiotic release from the surface, cracks, and pores of the cement. The majority of the antibiotic is released within the first days of implantation, but low levels may remain in the cement, with release occurring years after implantation.4,5
The use of ALBC in primary arthroplasty remains controversial owing to low infection rates with current prophylactic measures and risks associated with ALBC. One prospective, randomized trial demonstrated a significant reduction in deep infections in patients treated with cefuroxime ALBC versus plain cement, but all patients who developed infections were at high risk because of diabetes.4 A review of nearly 11,000 primary hip replacements in the Norwegian Arthroplasty Register documented that the lowest rates of infection occurred in patients who were given systemic antibiotics along with ALBC.23 Other trials have found no significant benefit, or only a short-term benefit, with the use of prophylactic ALBC.4,24
The prophylactic use of ALBC has a few disadvantages. The first is a potentially detrimental effect on the strength of the polymer cement. To maintain the cement’s strength and integrity, it is important to avoid using liquid antibiotics and to follow appropriate mixing procedures.4 Low-dose formulations may be less likely to alter the mechanical strength than higher doses, which are used to treat joint infections.
The second disadvantage is the risk of systemic toxicity or allergic reaction to ALBC. To date, no serious systemic toxicities or allergic reactions to ALBC have occurred. Systemic concentrations of the antibiotics are far below toxic concentrations even with high-dose ALBC, but high local concentrations in the bone have raised questions about toxicity to osteoblasts and osteocytes.5 Aminoglycosides have a low risk of allergy, but cefuroxime-loaded cements raise concern. An allergic reaction to the cement would most likely necessitate removal of the prosthesis and all the cement.4
Another concern is the development of antimicrobial resistance with widespread use of ALBC. In one study analyzing infections identified after primary arthroplasty, 88% of coagulase-negative Staphylococcus infections were resistant to gentamicin in patients who received gentamicin ALBC, versus only 16% in patients receiving plain cement.4 Therefore, gentamicin ALBC would be ineffective in treating the subsequent infection.
Lastly, regarding the cost-effectiveness of prophylactic ALBC, it is predicted that a fivefold decrease in the rate of postoperative infection would be needed to justify the increased cost of adding an antibiotic to bone cement prophylactically.4 With low rates of infection in most patients, it is unclear whether this approach would be cost-effective in every patient.
Currently, prophylactic use of ALBC in the U.S. is low, with only 11% of physicians using ALBC during primary arthroplasty. In contrast, in European countries it is a common practice, with up to 85% of physicians documenting use.4 The FDA has approved six formulations of premixed low-dose ALBC for use in revision procedures; they are not currently approved for routine prophylactic use. Based on recent data, there may be a role for prophylactic ALBC in patients at high risk for joint infection, including those with prior joint infection, diabetes, immunosuppression, malnutrition, or RA.4 Currently, guidelines do not recommend the use of prophylactic ALBC.10,11
Evaluation of Appropriate Prophylaxis
In 2002, the Centers for Medicare and Medicaid Services and the CDC implemented the National Surgical Infection Prevention Project (SIP). The goal of this project was to reduce postoperative morbidity and mortality through appropriate selection of antimicrobials, appropriate timing of prophylactic doses, and proper discontinuation of antimicrobials.25 In accordance with national guidelines, the three performance measures included use of cefazolin or cefuroxime unless the patient had a documented severe allergy or the risk of MRSA was high; a preoperative antimicrobial dose given within 1 hour prior to incision; and discontinuation of antimicrobial prophylaxis within 24 hours.25 A baseline sample of Medicare patients assessed in 2003 showed that the majority of antimicrobials used were consistent with the guidelines (97.2%), but only 59.7%% of orthopedic patients received their antibiotic within 60 minutes of surgery and only 36.7% of antibiotics were discontinued within 24 hours.25
In 2003, the three SIP performance measures were added to the core measure set used by the Joint Commission on Accreditation of Healthcare Organizations (JCAHO).26 The SIP measures have transitioned into a more comprehensive initiative, the Surgical Care Improvement Project (SCIP), which outlines seven specific measures to prevent surgical-site infections, focusing on antibiotic and nonantibiotic recommendations.27 The purpose of these quality-improvement measures is to standardize preoperative care and increase compliance in hospitals nationwide.27
Conclusion
The risk of joint infection after primary hip or knee arthroplasty is low. If infection does occur, however, the consequences are devastating, often necessitating prosthesis removal and prolonged hospital stays. To prevent these adverse consequences, it is recommended that patients receive appropriate prophylactic antibiotics, either cefazolin or cefuroxime in most cases, administered within 60 minutes of the incision and continued for 24 hours. Patients with a prior history of severe anaphylactic reaction to penicillins or cephalosporins may receive clindamycin or vancomycin, with clindamycin favored in patients without a history of MRSA. Vancomycin should be used judiciously, even in hospitals with a high prevalence of MRSA, in order to prevent resistance. ALBC is not approved for use in primary arthroplasty, but it may have some benefit in high-risk patient groups. The SCIP, which aims to improve the appropriate use of prophylactic antibiotics and preoperative care, has been incorporated as a core measure by the JCAHO.
REFERENCES
1. Tyllianakis ME, Karageorgos AC, Marangos MN, et al. Antibiotic
prophylaxis in primary hip and knee arthroplasty: comparison between
cefuroxime and two specific antistaphylococcal agents. J Arthroplasty. 2010;25:1078-1082.
2. Kurtz S, Ong K, Lau E, et al. Projections of primary and revision
hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg. 2007;89:780-785.
3. Peel TN, Cheng AC, Buising KL, Choong PF. Microbiological
aetiology, epidemiology, and clinical profile for prosthetic joint
infections: are current antibiotic prophylaxis guidelines effective? Antimicrob Agents Chemother. 2012;56:2386-2391.
4. Jiranek WA, Hanssen AD, Greenwald AS. Antibiotic-loaded bone cement for infection prophylaxis in total joint replacement. J Bone Joint Surg Am. 2006;88:2487-2500.
5. Bistolfi A, Massazza G, Vernè E, et al. Antibiotic-loaded cement in orthopedic surgery: a review. ISRN Orthopedics. 2011;2011:1-8.
6. Bozic KJ, Ries MD. The Impact of infection after total hip arthroplasty on hospital and surgeon resource utilization. J Bone Joint Surg. 2005;87:1746-1751.
7. Fogelburg EV, Zitzmann EK, Stinchfield FE. Prophylactic penicillin in orthopaedic surgery. J Bone Joint Surg Am. 1970;52:95-98.
8. Hill C, Flamant R, Mazas F, Evrard J. Prophylactic cefazolin
versus placebo in total hip replacement. Report of a multicentre
double-blind randomised trial. Lancet. 1981;1:795-796.
9. AlBuhairan B, Hind D, Hutchinson A. Antibiotic prophylaxis for wound infections in total joint arthroplasty: a review. J Bone Joint Surg Br. 2008;90:915-919.
10. ASHP therapeutic guidelines on antimicrobial prophylaxis in surgery. American Society of Health-System Pharmacists. Am J Health Syst Pharm. 1999;56:1839-1888.
11. American Academy of Orthopaedic Surgeons. Recommendations for the
use of intravenous antibiotic prophylaxis in primary total joint
arthroplasty. www.aaos.org/about/papers/advistmt/1027.asp. Accessed May
16, 2012.
12. Meehan J, Jamali AA, Nguyen H. Prophylactic antibiotics in hip and knee arthroplasty. J Bone Joint Surg Am. 2009;91:2480-2490.
13. Schurman DJ, Hirshman HP, Kajiyama G, et al. Cefazolin concentrations in bone and synovial fluid. J Bone Joint Surg Am. 1978;60:359-362.
14. Williams DN, Gustilo RB, Beverly R, Kind AC. Bone and serum
concentrations of five cephalosporin drugs. Relevance to prophylaxis and
treatment in orthopedic surgery. Clin Orthop Relat Res. 1983;179:253-265.
15. Bratzler DW, Houck PM. Antimicrobial prophylaxis for surgery: an
advisory statement from the National Surgical Infection Prevention
Project. Am J Surg. 2005;189:395-404.
16. Finkelstein R, Rabino G, Mashiah T, et al. Vancomycin versus
cefazolin prophylaxis for cardiac surgery in the setting of a high
prevalence of methicillin-resistant staphylococcal infections. J Thorac Cardiovasc Surg. 2002;123:326-332.
17. Sewick A, Makani A, Wu C, et al. Does dual antibiotic prophylaxis
better prevent surgical site infections in total joint arthroplasty? Clin Orthop Relat Res. 2012;Epub. DOI 10.1007/s11999-012-2255-1.
18. Burke JF. The effective period of preventive antibiotic action in experimental incisions and dermal lesions. Surgery. 1961;50:161-168.
19. Classen DC, Evans RS, Pestonik SL, et al. The timing of
prophylactic administration of antibiotics and the risk of
surgical-wound infection. New Engl J Med. 1992;326:281-286.
20. Williams DN, Gustilo RB. The use of preventive antibiotics in orthopaedic surgery. Clin Orthop Relat Res. 1984;190:83-88.
21. Tang WM, Chiu KY, Ng TP, et al. Efficacy of a single dose of
cefazolin as a prophylactic antibiotic in primary arthroplasty. J Arthroplasty. 2003;18:714-718.
22. Clyburn TA, Cui Q. Antibiotic laden cement: current state of the
art. www.aaos.org/news/bulletin/may07/clinical7.asp. Accessed May 21,
2012.
23. Espehaug B, Engesaeter LB, Vollset SE, et al. Antibiotic
prophylaxis in total hip arthroplasty. Review of 10,905 primary cemented
total hip replacements reported to the Norwegian Arthroplasty Register,
1987 to 1995. J Bone Joint Surg Br. 1997;79:590-595.
24. Namba RS, Chen Y, Paxton EW, et al. Outcomes of routine use of antibiotic-loaded cement in primary total knee arthroplasty. J Arthroplasty. 2009;24(suppl 6):44-47.
25. Bratzler DW, Houck PM, Richards C, et al. Use of antimicrobial
prophylaxis for major surgery: baseline results from the National
Surgical Infection Prevention Project. Arch Surg. 2005;140:174-182.
26. The Joint Commission. Surgical Care Improvement Project.
www.jointcommission.org/surgical_care_improvement_project. Accessed May
17, 2012.
27. Rosenberger LH, Politano AD, Sawyer RG. The surgical care
improvement project and prevention of post-operative infection,
including surgical site infection. Surg Infect (Larchmt). 2011;12:163-168.
To comment on this article, contact rdavidson@uspharmacist.com.






