US Pharm. 2012;37(11):39-44.
ABSTRACT: Several metabolic consequences of antipsychotic use have been described within the literature, including weight gain, hyperglycemia, and dyslipidemia. While the exact mechanisms of antipsychotic-induced metabolic dysfunction are unknown, there are a variety of proposed mechanisms. Since weight gain is considered a precipitating factor for the development of diabetes and dyslipidemia, monitoring of weight changes with antipsychotic treatment is an important screening measure. As some antipsychotic agents are less likely to contribute to weight gain, such as aripiprazole and ziprasidone, it seems prudent to choose a lower-risk antipsychotic in people who are at high risk for or have a preexisting diagnosis of diabetes or in whom hyperglycemia, dyslipidemia, and/or substantial weight gain develops.
Diabetes mellitus (DM) is the seventh leading cause of death in the United States and is a major cause of renal failure, new onset blindness, and nontraumatic limb amputations.1 DM additionally places people at risk for adverse cardiovascular outcomes and is a major contributor to the incidence of stroke and heart disease. National estimates indicate that DM affects 8.3% of the U.S. population, which equates to a staggering 25.8 million people, and the incidence of DM is continuing to grow at an alarming rate.1
While the health and economic consequences of type 2 DM (T2DM) are widely known, the contribution of medications to the development of T2DM is sometimes less well recognized. One class of drugs for which there are large amounts of clinical data to substantiate a link between their use and the development of T2DM is antipsychotic medications. This article will provide an overview of antipsychotic-induced DM and current recommendations pertaining to its management.
Antipsychotic-Induced T2DM: How Common Is It?
There are many potential metabolic consequences of antipsychotic use that have been described within the literature. Such metabolic disturbances primarily include weight gain, hyper-glycemia, and dyslipidemia.2-4 As a consequence of these metabolic disturbances, the risk of comorbid hypertension and heart disease are also reportedly increased in people using antipsychotic drugs.5
The association of antipsychotic use to T2DM development is supported by retrospective epidemiologic studies as well as post-marketing surveillance.5 Identifying the prevalence of antipsychotic-induced DM versus the background prevalence of DM in people with schizophrenia and other mental health disorders, however, is confounded by a number of factors. Many studies to date have included patients with schizophrenia receiving treatment with antipsychotic therapy; thus, determining the baseline prevalence of DM in this population in the absence of antipsychotic therapy is difficult. Data indicate the prevalence of DM and obesity to be approximately 1.5 to 2 times higher in people with schizophrenia or affective disorders when compared to the general population.6
In an analysis of the Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) study, patients with schizophrenia were found to have higher rates of T2DM (13%) when compared to age, race, and gender-matched controls from the National Health and Nutrition Examination Survey (NHANES) III (3%; P <.001).7 Similarly, a cross-sectional study enrolling outpatients with a diagnosis of schizophrenia found an 11.5% lifetime prevalence of T2DM in this patient population.5 Cases of patients developing hyperglycemia upon initiation of antipsychotic therapy followed by the resolution of hyperglycemia upon discontinuation of the offending medication have also been reported.6 A cohort study of antipsychotic-naïve patients followed participants for 3 years after the initiation of antipsychotic therapy.8 After 3 years of follow-up, the incidence rate of DM was found to be 0.65% per patient-year. Of note, in older participants the time to DM onset was significantly shorter when compared to that in younger participants, indicating that age may be a risk factor for antipsychotic-induced DM.8
People at highest risk for antipsychotic-induced DM are those diagnosed with schizophrenia, bipolar disorder, psychotic depression, dementia, autism, or developmental disorders.2,6 Other less common uses of antipsychotic medications, which can also result in the development of treatment-induced metabolic disturbances, are post-traumatic stress disorder (PTSD), delirium, personality disorder, and aggressive behavior.6 Risk factors for the development of antipsychotic-induced DM are complex and may involve an interplay between sedentary lifestyle, the pharmacologic properties of the antipsychotic agent being used (e.g., drug-specific risk of contributing to drug-induced metabolic disturbances), worsening metabolic homeostasis, and possible genetic causes.8,9
How Do Antipsychotics Contribute to the Development of Weight Gain, T2DM, and Dyslipidemia?
Metabolic disturbances seen with antipsychotic use typically manifest initially as weight gain that can advance to obesity. Cases of new onset DM associated with weight gain and independent of weight gain have been reported in the literature.3 Second-generation antipsychotic (SGA) therapy can contribute to rapid weight gain in the first few months of treatment, with weight gain continuing in some patients for up to one year. Variation in weight gain exists and can range from gains of 0.5 to 5.0 kg after 10 weeks of antipsychotic therapy.6 Given these risks, distinguishing the presence of metabolic syndrome is useful to proactively identify metabolic risk in people with mental illness. In addition to the presence of mental illness, lifestyle factors such as increased food intake, intake of particularly high carbohydrate content drinks and snacks, limited physical activity, drug abuse, or tobacco smoking can all contribute to the development of weight gain and obesity in this population.2
While the exact mechanisms of antipsychotic weight gain are unknown, there are a variety of proposed mechanisms whereby antipsychotics may contribute to weight gain, DM, and dyslipidemia. It is widely believed that antipsychotic drugs contribute to weight gain and corresponding hyperglycemia and dyslipidemia via effects mediated by binding to serotonin (5-HT2), norepinephrine, dopamine, and/or histamine receptors. Serotonin activity at receptor sites is a potent satiety signal, with the most implicated receptors being 5-HT1A and 5-HT2C. Stimulation of 5-HT1A is associated with an increase in food intake whereas stimulation of 5-HT2C is related to a decrease in food intake. Antagonism of the 5-HT2C receptor can, in turn, lead to an increase in food intake, with most SGAs possessing 5-HT2C antagonist activity.10 The histamine receptor has additionally been theorized to mediate medication-induced weight gain, with histamine receptor affinity correlating with drug-induced weight gain in some studies.11 Olanzapine, for instance, possesses a high affinity for histamine receptors, while agents with a lower risk of contributing to weight gain, such as ziprasidone and aripiprazole, have lower histamine receptor affinities.10
Hyperglycemia, impaired fasting glucose, and DM are likely caused and/or exacerbated by drug-induced insulin resistance stemming from weight gain; however, a direct effect of antipsychotic drugs on insulin resistance may also play a role.6 Mechanistic studies have demonstrated both insulin resistance and increased insulin secretion (an indirect indicator of increased insulin resistance) with antipsychotics.3,12 Likewise, changes in body weight may also contribute to antipsychotic-induced dyslipidemia.6 With weight gain considered a key adverse effect that potentiates the development of dyslipidemia, hyperglycemia, and DM, additional research is warranted to unravel mechanisms contributing to the metabolic complications associated with antipsychotic pharmacotherapy.
Monitoring Recommendations
Recommendations for patient monitoring for the development of dyslipidemia, weight gain, and DM have been published.6 They consider the potential metabolic risks when initiating an SGA; patient, family, and caregiver education; baseline screening; and referral to a specialist, if needed, as key components of patient care.6 As weight gain is considered a precipitating factor for the development of DM and dyslipidemia, monitoring of weight changes with treatment is an important screening measure.
Baseline and follow-up monitoring is recommended by the joint consensus statement from the American Diabetes Association, the American Psychiatric Association, the American Association of Clinical Endocrinologists, and the North American Association for the Study of Obesity to help reduce the risk of developing diabetes, cardiovascular disease, and other complications of DM.6 According to the consensus statement, screening measures are recommended to be obtained before or soon after the initiation of antipsychotic medication. The recommended screening measures are summarized in TABLE 1.6

Accordingly, clinicians are encouraged to record and track the patient’s height and weight (BMI) before and during treatment and to encourage patients to self-monitor weight change by charting their weight.6 Follow-up monitoring is additionally recommended at various intervals depending on the measure being evaluated. Fasting plasma glucose and blood pressure are assessed at baseline, at 3 months, and then annually, while lipids are assessed at baseline, at 3 months, and then every 5 years. If patients are at higher risk to develop DM or hypertension, more frequent monitoring is advised. Using these measures, clinicians can better assess patients’ baseline status for weight (overweight defined as BMI 25-29), prediabetes (fasting plasma glucose 100-125 mg/dL) or DM (fasting plasma glucose ≥126 mg/dL), hypertension (>140/90 mmHg), or dyslipidemia to help determine their risk status and the contribution and/or causation of antipsychotic therapy.6
Depending on the presence of any metabolic disorders at baseline, clinicians can make an informed decision regarding the selection of appropriate antipsychotic therapy (see the following section). For patients who are overweight, referral for a consult with a nutritionist and for activity counseling is advised, especially if an SGA with a high likelihood of contributing to weight gain is initiated. The panel goes on to state that education is a very important aspect of management, and all patients, caregivers, and family members should be counseled regarding the recognition of the signs and symptoms of DM and diabetic ketoacidosis (DKA) (TABLE 2).6 Abrupt development of DKA as the only presenting sign of glucose abnormalities has been reported. For patients who do develop hyperglycemia, prompt referral to a clinician experienced in the management of DM is recommended.

For patients who do develop metabolic abnormalities stemming from antipsychotic therapy, switching to another antipsychotic agent associated with less hyperglycemia, weight gain, and dyslipidemia is a potential management strategy. The consensus statement recommends switching an agent if the patient gains ≥5% of his or her baseline body weight with therapy.6
Are There Differences Between Antipsychotic Agents?
SGAs most implicated in leading to metabolic abnormalities are clozapine, olanzapine, risperidone, quetiapine, aripiprazole, and ziprasidone.10 There is variability between first-generation antipsychotics (FGAs) and SGAs regarding their metabolic and other adverse effects. FGAs are known to contribute to extrapyramidal symptoms (EPS) such as dystonia, pseudoparkinsonism, akathisia, and late-onset tardive dyskinesia.6 High-potency FGAs such as haloperidol and fluphenazine also have higher occurrences of EPS in comparison to other, low-potency FGAs.13 SGAs, while generally associated with fewer EPS and better overall tolerability when compared to FGAs, are also associated with a higher risk of contributing to metabolic disturbances.
Clozapine and olanzapine are considered the worst offenders for contributing to hyperglycemia, dyslipidemia, and weight gain. Intermediate effects are seen with risperidone and quetiapine, while ziprasidone and aripiprazole are considered to convey the smallest relative risk.13 Interestingly, the relative risks of these agents parallel their relative effects on weight gain. A report of 1-year treatment data compiled from industry-sponsored trials showed more weight gain with clozapine and olanzapine (~12 kg) when compared to quetiapine and risperidone (~2 to 3 kg), aripiprazole (slightly less than quetiapine or risperidone), or ziprasidone (~2 to 3 kg weight loss).2 These differences should be considered in light of the fact that they were obtained from separate industry-sponsored trials.
While data are limited, the American Diabetes Association states that the effects of SGA therapies on lipids are concordant with their effects on body weight; thus, monitoring is recommended.6 Mixed findings exist for risperidone and quetiapine in regard to risk for the development of new-onset or worsening T2DM, and they are considered to have intermediate effects on increasing lipids.6 While ziprasidone and aripiprazole are considered to possess the lowest risk, case studies of new-onset hyperglycemia have been reported in the literature, including a report of aripiprazole precipitating a case of DKA.13 A summary of the reported metabolic effects of SGAs is provided in TABLE 3.6
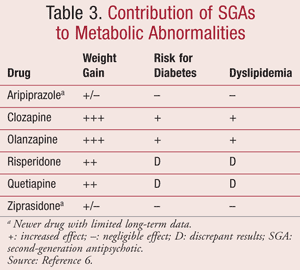
Overall, it seems prudent to choose a lower-risk antipsychotic such as aripiprazole or ziprasidone for an individual who has preexisting DM or is otherwise at high risk. If hyperglycemia, dyslipidemia, and/or substantial weight gain develops, switching to a lower-risk antipsychotic drug should be considered.6,14 While data regarding potential metabolic consequences of long-term treatment with newer SGAs (such as asenapine, iloperidone, lurasidone, and paliperidone) are currently lacking, the relative effects of these agents on the development of diabetes and other metabolic derangements will become more clear with additional study and clinical experience.
Conclusion
DM is reaching epidemic proportions worldwide, and the contributions of medications to the development of hyperglycemia and other metabolic derangements is receiving more attention. Pharmacists are in a unique position to counsel and encourage appropriate self-monitoring in patients receiving certain drugs, such as antipsychotics, that can contribute to the development of weight gain, hyperglycemia, and dyslipidemia. Pharmacists can serve as a catalyst for patients receiving antipsychotics to communicate adverse events to other health care providers and seek therapeutic substitutions, counseling, and/or treatment if adverse events result from treatment. As a population, people with schizophrenia often have poor access to care and lower levels of psychosocial support; thus, the pharmacist’s recognition and intervention in this population have the potential to improve quality of care.
Providers should be aware that the onset of DM can be rapid and severe in some patients, with case reports of life-threatening DKA reported, sometimes within weeks after starting antipsychotic treatment.12,14 Rarely, such cases have occurred even with low-risk SGAs such as ziprasidone and aripiprazole, so that diligence in monitoring is warranted even with agents less likely to contribute to weight gain, hyperglycemia, and dyslipidemia.13,15
While the exact mechanisms contributing to antipsychotic-induced metabolic changes are unknown, research is ongoing to pinpoint the mechanisms at play. Overall, appropriate monitoring, reporting, and patient and clinician awareness are critical to the early recognition and treatment of antipsychotic-induced DM.
REFERENCES
1. Centers for Disease Control and Prevention. National
diabetes fact sheet: national estimates and general information on
diabetes and prediabetes in the United States, 2011. Atlanta, GA: U.S.
Department of Health and Human Services, CDC; 2011.
2. Reynolds GP, Kirk SL. Metabolic side effects of antipsychotic drug treatment—pharmacological mechanisms. Pharmacol Ther. 2010;125:169-179.
3. Luna B, Feinglos MN. Drug-induced hyperglycemia. JAMA. 2001;286:1945-1948.
4. Kohen I, Gampel M, Reddy L, Manu P. Rapidly developing hyperglycemia during treatment with olanzapine. Ann Pharmacother. 2008;42:588-591.
5. Argo T, Carnahan R, Barnett M, et al. Diabetes prevalence estimates in schizophrenia and risk factor assessment. Ann Clin Psychiatry. 2011;23:117-124.
6. American Diabetes Association, American Psychiatric
Association, American Association of Clinical Endocrinologists, North
American Association for the Study of Obesity. Consensus development
conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27:596-601.
7. Goff DC, Sullivan LM, McEvoy JP, et al. A comparison of
ten-year cardiac risk estimates in schizophrenia patients from the
CATIE study and matched controls. Schizophr Res. 2005;80:45-53.
8. Nielsen J, Skadhede S, Correll CU. Antipsychotics
associated with the development of type 2 diabetes in
antipsychotic-naïve schizophrenia patients. Neuropsychopharmacology. 2010;35:1997-2004.
9. Burden of mental illness. Schizophrenia. CDC. Updated
July 1, 2011. www.cdc.gov/mentalhealth/basics/burden.htm. Accessed July
30, 2012.
10. Llorente MD, Urrutia V. Diabetes, psychiatric disorders, and the metabolic effects of antipsychotic medications. Clin Diabetes. 2006;24:18-24.
11. Wirshing DA, Wirshing WC, Kysar L, et al. Novel antipsychotics: comparison of weight gain liabilities. J Clin Psychiatry. 1999;100:3-16.
12. Lebovitz HE. Metabolic consequences of atypical antipsychotic drugs. Psychiatr Q. 2003;74:277-290.
13. Kibbey K, Roberts AM, Nicholson G. Diabetic ketoacidosis and elevated serum lipase in the setting of aripiprazole therapy. Diabetes Care. 2010;23:e96.
14. Lean ME, Pajonk FG. Patients on atypical antipsychotic drugs: another high-risk group for type 2 diabetes. Diabetes Care. 2003;26:1597-1605.
15. Letourneau G, Abdel-Baki A, Dubreucq S, et al.
Hyperosmolar hyperglycemic state associated with ziprasidone treatment: a
case report. J Clin Psychopharmacol. 2011;31:671-673.
To comment on this article, contact rdavidson@uspharmacist.com.





