US Pharm. 2016;41(6):HS14-HS18.
ABSTRACT: Graves’ disease is an autoimmune disorder that occurs when the immune system attacks the thyroid gland and causes it to produce too much thyroid hormone (hyperthyroidism). The exact cause of Graves’ disease is not known. The disorder can affect anyone at any age, but it is more common in women. If left untreated, Graves’ disease can cause a severe, life-threatening crisis (i.e., thyroid storm). The primary goals of treatment are to eliminate excess thyroid hormone and minimize the long-term consequences of the disease. Treatments include radioactive iodine, antithyroid medications (propylthiouracil, methimazole), and surgery. Beta-blockers may be used to provide symptomatic relief.
Graves’ disease is named for the physician who first described the condition in Ireland in 1835—Robert J. Graves.1 It is also referred to as Basedow’s disease—named after a German doctor, Karl Adolph von Basedow, who also described the disorder in 1840. He was unaware that Graves had described the same disease a few years earlier. The term Basedow’s disease is more commonly used in continental Europe; in the United States, it is called Graves’ disease.1
The thyroid, a butterfly-shaped gland located at the front of the neck, is an important organ of the endocrine system.2 This gland releases the hormones thyroxine (T4) and triiodothyronine (T3), which control body metabolism.3-6 Thyroid hormone helps the body use energy, stay warm, and keep the brain, heart, muscles, and other organs functioning properly. Controlling metabolism is important for regulating mood, weight, and mental and physical energy levels. When the body produces too much thyroid hormone, the condition is called hyperthyroidism. An underactive thyroid gland leads to hypothyroidism.
Because thyroid hormones affect various body systems, signs and symptoms associated with Graves’ disease can be wide-ranging and significantly influence a person’s overall well-being. Common signs of hypothyroidism include lethargy, dry skin, brittle hair and fingernails, and cold temperature sensitivity.3,4 Typical signs of hyperthyroidism often are just the opposite and include tachycardia, heat sensitivity, and moist, oily skin.3-7 Although a number of disorders may result in hyperthyroidism, Graves’ disease is a common cause.
Although Graves’ disease may affect anyone, it is more common among women and has an onset before the age of 40 years.1,7-9 The incidence is about 1 in 2,000 people (0.05%). Factors that increase the risk of Graves’ disease are described in TABLE 1.4,8,10,11

Pathophysiology
Graves’ disease is an autoimmune disorder that can occur when the immune system mistakenly attacks healthy thyroid tissue, leading to overactivity of the thyroid gland. Normally, thyroid function is regulated by a hormone released by the pituitary gland. When needed, the body’s immune system produces antibodies designed to target a specific virus, bacterium, or other foreign substance. In Graves’ disease, the body produces an antibody to some of the cells in the thyroid gland. The antibody associated with Graves’ disease, thyrotropin receptor antibody, mimics the regulatory pituitary hormone and overrides the body’s normal regulation of the thyroid, causing an overproduction of thyroid hormones.2,6,8,9
These antibodies, called thyroid-stimulating immunoglobulins (TSIs), cause the thyroid to grow and make more thyroid hormone than the body needs. TSIs bind to thyroid cell receptors, which are normally “docking stations” for thyroid-stimulating hormone (TSH). TSIs then trick the thyroid into growing and producing too much thyroid hormone, leading to hyperthyroidism. The thyroid gland can become enlarged when the patient’s diet is lacking sufficient iodine or when levels of TSH become elevated in response to a defect in normal hormone synthesis by the thyroid gland. In Graves’ disease, B- and T-lymphocyte–mediated autoimmunity is known to be directed at familiar thyroid antigens—thyroglobulin, thyroid peroxidase, sodium-iodide symporter, and the thyrotropin receptor. The thyrotropin receptor is the primary autoantigen of Graves’ disease and is responsible for the manifestation of hyperthyroidism. Cardinal symptoms of hyperthyroidism include bulging eyes (exophthalmos), heat intolerance, increased energy, tachycardia, difficulty sleeping, diarrhea, and anxiety. Signs and symptoms specific to Graves’ disease are listed in TABLE 2.4,10,11 If left untreated, Graves’ disease can cause severe thyrotoxicosis or thyroid storm.9,10,12,13

Uncommon manifestations of Graves’ disease include Graves’ dermopathy (a reddening and thickening of the skin, often on the shins or tops of the feet) and Graves’ ophthalmopathy (a buildup of certain carbohydrates in the skin).2,8
Complications of Graves’ Disease
There are many complications associated with Graves’ disease, including pregnancy issues, heart disorders, osteoporosis, and thyroid storm. Possible complications during pregnancy include miscarriage, preterm birth, fetal thyroid dysfunction, poor fetal growth, maternal heart failure, and preeclampsia.4,5,8 If left untreated, Graves’ disease can lead to heart rhythm disorders, changes in the structure and function of heart muscles, and the inability of the heart to pump enough blood to the body.5,8,10 Untreated hyperthyroidism also can lead to weak, brittle bones.5,8
Thyroid storm is a rare, life-threatening complication also known as accelerated hyperthyroidism or thyrotoxic crisis.2,9,13-16 It is more likely when severe hyperthyroidism is untreated or treated inadequately. Serious adverse effects include fever, profuse sweating, vomiting, diarrhea, delirium, severe weakness, seizures, markedly irregular heartbeat, jaundice, severe low blood pressure, and coma. Thyroid storm often requires immediate emergency care.5,8,15
Tests and Diagnosis
The diagnosis of Graves’ disease includes several steps. During a physical examination, the eyes will be examined to see if they are irritated or protruding, and if the thyroid gland is enlarged.8 Heart rate, blood pressure, and any signs of tremors will be monitored. Blood tests indicate that TSH and thyroid levels are usually elevated in Graves’ disease.8 The amount and rate of radioactive iodine taken up by the thyroid helps determine if Graves’ disease or another condition is the cause of the hyperthyroidism. Ultrasound can show if the thyroid gland is enlarged, and is most useful in people who cannot undergo radioactive iodine uptake, such as pregnant women. Imaging tests (CT scan or MRI) can help identify and diagnose Graves’ ophthalmopathy.5
Treatment
The primary goals of treatment for hyperthyroidism are to eliminate excess thyroid hormone and minimize the long-term consequences.9 Treatments include radio-active iodine, antithyroid medications (propylthiouracil [PTU], methimazole [MMI]), and surgery.2,8-10 Beta-blockers are sometimes added to provide symptomatic relief.9,13,15
Radioactive Iodine Therapy (RIT): The usual dose for RIT ranges from 5 to 15 mCi of 131I. In general, higher dosages are required for patients who have large goiters or low radioiodine uptake, or who have been pretreated with antithyroid drugs.15,16
Because the thyroid needs iodine to produce hormones, the radioiodine goes into the thyroid cells and, over time, overactive thyroid cells are destroyed. The thyroid gland shrinks, and in several weeks to months, hyperthyroid symptoms gradually diminish.16 RIT may increase the risk of new or worsened symptoms of Graves’ ophthalmopathy. This adverse effect is usually mild and temporary, but the therapy may not be utilized if the person already has moderate-to-severe eye problems. Other side effects of this therapy include tenderness in the neck and a temporary increase in thyroid hormone levels. Because this treatment can cause overall thyroid function to decline, RIT is not usually utilized in pregnant or nursing women. Other treatments to supply the body with normal amounts of thyroid hormone may be needed. Patients currently taking antithyroid drugs must discontinue the medication at least 2 days prior to taking the radiopharmaceutical.11,16 With radioactive iodine, the goal of therapy is to cause a hypothyroid state due to the destruction of the thyroid gland, which usually occurs 2 to 3 months after administering the drug. The absolute contraindication for radioiodine is pregnancy.14,16,17
Antithyroid Medication (ATM): ATMs work by blocking the thyroid’s use of iodine to produce hormones.5,11 These medications include oral PTU and MMI. PTU may be used when patients cannot tolerate MMI, radioactive iodine, or surgery. When these drugs are used, a relapse of hyperthyroidism may occur over time. ATMs block the coupling reaction of monoiodotyrosine and diiodotyrosine to produce T4 and T3, thus effectively preventing synthesis of thyroid hormone.9,11 Additionally, PTU, but not MMI, inhibits peripheral conversion of T4 to T3.9 Initial PTU oral doses range between 300 and 600 mg daily (maximum daily dose = 1,200 mg). Initial oral doses of MMI, which is 10 times more potent than PTU, range between 30 and 60 mg daily (maximum daily dose = 120 mg).9,11,13 Although these medications are traditionally divided into three to four doses daily, they may be administered as single doses.9 Clinical improvement is usually observed at 4 to 8 weeks, at which time the dose should be tapered on a monthly schedule.9 Typical oral maintenance doses for each agent are 50 to 300 mg for PTU and 5 to 30 mg for MMI. Treatment should be continued for 12 to 24 months if the goal is to achieve lifelong remission.9,11,13
ATMs may be used before or after RIT as a supplemental treatment. Adverse effects of both drugs include rash, joint pain, liver failure, and a decrease in disease-fighting white blood cells. Because of the risk of birth defects, MMI is not usually used to treat pregnant women in the first trimester. PTU may be considered the preferred drug of choice just before and during the first trimester of pregnancy. The FDA has issued a black box warning on PTU indicating its ability to cause potentially fatal or severe liver injury and acute liver failure in adults and pediatric patients.18
Beta-Blockers: These medications do not inhibit the production of thyroid hormones, but they do block the effect of hormones on the body. They may be used to provide relief of irregular heartbeats, tremors, anxiety or irritability, heat intolerance, sweating, diarrhea, and muscle weakness. Beta-blockers commonly used include propranolol, atenolol, metoprolol, lopressor, and nadolol. Because beta-blockers may complicate management of asthma and diabetes, those patients should use these drugs with caution.5,11
Surgery: Surgery to remove all or part of the thyroid gland (thyroidectomy or subtotal thyroidectomy) is also an option for the treatment of Graves’ disease. After the procedure, patients will likely need treatment to supply the body with normal amounts of thyroid hormone. Risks of surgery include potential damage to the vocal cords and parathyroid glands. The parathyroid glands produce a hormone that controls the level of calcium in the blood. Complications are rare under the care of a surgeon experienced in thyroid surgery.5,11
Treatment of Graves’ Ophthalmopathy
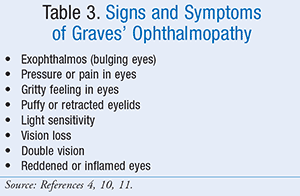
Graves’ ophthalmopathy can be divided into two clinical phases: the inflammatory stage and the fibrotic stage.5 The inflammatory stage is marked by edema and deposition of glycosaminoglycan in the extraocular muscles. This results in the clinical manifestations of orbital swelling, stare, diplopia, periorbital edema, and, at times, pain. Graves’ ophthalmopathy does not always improve with treatment for Graves’ disease. Symptoms of Graves’ ophthalmopathy (TABLE 3) may even worsen for 3 to 6 months. After that, the signs and symptoms usually stabilize for a year or so and then begin to get better, often on their own. Mild symptoms of Graves’ ophthalmopathy may be managed by using OTC artificial tears during the day and lubricating gels at night.5
If symptoms are more severe, the doctor may recommend several treatments, including corticosteroids, orbital decompression surgery, orbital radiotherapy, and nonpharmacologic therapies. Treatment with prednisone, may be needed to reduce swelling behind the eyeballs.11 Adverse effects of corticosteroids include fluid retention, weight gain, elevated blood glucose levels, increased blood pressure, and mood swings. Orbital decompression surgery is a procedure that removes the bone between the eye socket (orbit) and sinuses, the air spaces next to the orbit.11 This gives the eyes room to move back to their original position. This treatment is usually used if pressure on the optic nerve threatens the loss of vision. Possible complications include double vision. Orbital radiotherapy uses targeted x-rays over the course of several days to destroy some of the tissue behind the eyes.2,11
Lifestyle and Home Remedies: Eating well and exercising can enhance the improvement in some symptoms while being treated. Because the thyroid controls metabolism, the patient may have a tendency to gain weight when the hyperthyroidism is corrected. Brittle bones can also occur with Graves’ disease, and weight-bearing exercises can help maintain bone density.2,4,6
Patients can apply cool compresses to their eyes, as the added moisture may be soothing. Patients should also be advised to wear sunglasses to lessen sensitivity to ultraviolet (UV) rays and bright light. Lubricating eye drops may relieve the dry, scratchy sensation on the surface of the eyes. A paraffin-based gel, such as Refresh Lacri-Lube, can be applied at night. Patients should elevate the head of their bed. Keeping the head higher than the rest of the body lessens fluid accumulation in the head and may relieve the pressure on the eyes. Finally, patients should be counseled not to smoke, since smoking worsens Graves’ ophthalmopathy.
Treatment of Graves’ Dermopathy
Use of OTC creams or ointments containing hydro-cortisone may help relieve swelling and reddening. In addition, using compression wraps on the legs may be beneficial.5,11
Conclusion
Pharmacotherapy for Graves’ disease is complex but manageable. Since the desired result of antithyroid medication is to destroy the thyroid gland and cause a hypothyroid state, patients will require continual clinical monitoring, counseling (TABLE 4), and lifelong thyroid replacement with thyroid hormone.

REFERENCES
1. Ellis H. Robert Graves: 1796-1852. Br J Hosp Med (Lond). 2006;67(6):313.
2. Yeung SJ. Graves disease. Overview. Medcape. November 18, 2015. http://emedicine.medscape.com/article/120619-overview. Accessed March 8, 2016.
3. Garber JR, Cobin RH, Gharib H, et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18(6):988-1028.
4. Graves disease. MedlinePlus. April 21, 2015. www.nlm.nih.gov/medlineplus/ency/article/000358.htm. Accessed March 8, 2016.
5. Cockerham KP, Chan SS. Thyroid eye disease. Neurol Clin. 2010;28:729-755.
6. Yeung SJ. Graves disease. Epidemiology. Medcape. November 18, 2015. http://emedicine.medscape.com/article/120619-overview#a6. Accessed March 8, 2016.
7. Woeber KA. Update on the management of hyperthyroidism and hypothyroidism. Arch Fam Med. 2007;9(8):743-747.
8. Graves disease. Risk factors. Mayo Clinic. www.mayoclinic.org/diseases-conditions/graves-disease/basics/risk-factors/con-20025811. Accessed March 8, 2016.
9. Yeung SJ. Graves disease. Background. Medcape. November 18, 2015. http://emedicine.medscape.com/article/120619-overview#a4. Accessed March 8, 2016.
10. Yeung SJ. Graves disease. Pathophysiology. Medcape. November 18, 2015. http://emedicine.medscape.com/article/120619-overview#a5. Accessed March 8, 2016.
11. Yeung SJ. Graves disease treatment & management. November 18, 2015. http://emedicine.medscape.com/article/120619-treatment. Accessed March 8, 2016.
12. Nayak B, Burman K. Thyrotoxicosis and thyroid storm. Endcrinol Metab Clin North Am. 2006;35(4):663-686.
13. Mandel SJ, Larsen PR, Davies TF. Thyrotoxicosis. In: Melmed S, Polonsky KS, Larsen PR, Kronenberg HM, eds. Williams Textbook of Endocrinology. 12th ed. Philadelphia, PA: Elsevier Saunders; 2011:362-405.
14. Bhattacharyya A, Wiles PG. Thyrotoxic crisis presenting as acute abdomen. 1997;90(12):681-682.
15. Bahn RS, Burch HB, Cooper DS, et al. Hyperthyroidism and other causes of thyrotoxicosis: management guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Endocr Pract. 2011;17(3):456-520.
16. McDermott MT, Kidd GS, Dodson LE, et al. Radioiodine-induced thyroid storm. Case report and literature review. Am J Med. 1983;75(2):353-359.
17. Read CH Jr, Tansey MJ, Menda Y. 36-year retrospective analysis of the efficacy and safety of radioactive iodine in treating young Graves’ patients. J Clin Endocrinol Metab. 2004;89(9):4229-4233.
18. FDA Drug Safety Communication. New boxed warning on severe liver injury with propylthiouracil. April 21, 2010. www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm209023.htm. Accessed March 8, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.






