US Pharm. 2010;35(7):49-53.
Uncontrolled asthma has been responsible for 1.8 million emergency department (ED) visits and almost 500,000 hospitalizations in the United States in 2004 alone.1,2 Furthermore, in 2006, the number of physician office, hospital outpatient, and ED visits with asthma as the primary diagnosis was 13.3 million.3 During the last seven years, three asthma maintenance medications have been approved and marketed: ciclesonide (Alvesco), budesonide/formoterol (Symbicort), and omalizumab (Xolair). This article will systematically review the safety, tolerability, efficacy, price, simplicity of use, and some of the major published studies of the newest treatment regimens for asthma maintenance.4
Ciclesonide
Ciclesonide (Alvesco; Sepracor), an inhaled corticosteroid, was approved by the FDA in 2006 for maintenance treatment of asthma in patients aged 12 years and older.5 Ciclesonide is a prodrug that is hydrolyzed to its active metabolite, C21-desisobutyryl-ciclesonide, following oral inhalation. Its mechanism of action includes inflammatory-cell migration and activation, reduced airway hyperresponsiveness, and blockade of late-phase allergic reactions.5,6
Safety: Ciclesonide is not indicated for the treatment of acute bronchospasm or status asthmaticus, and it is contraindicated in patients with known hypersensitivity to ciclesonide. Precautions should be taken in patients at risk for worsening adrenal function, immunosuppression, decreased bone mineral density (BMD), glaucoma, intraocular pressure, or cataract. The Expert Panel Report 3 acknowledged that long-term use of high-dose inhaled corticosteroids (ICS) can lead to adverse effects on linear growth; therefore, adolescents receiving ICS should be consistently monitored for any deviations from predicted growth patterns.6 Studies were inconsistent regarding ciclesonide's efficacy in pediatric patients aged less than 12 years; for this reason, ciclesonide is not approved for use in this population.5 Ciclesonide is classified as pregnancy category C, and the extent of its secretion into breast milk is unknown; therefore, caution should be exercised in nursing women.5
Tolerability: The most common adverse reactions experienced by patients (>3%) taking ciclesonide are headache, nasopharyngitis, sinusitis, pharyngolaryngeal pain, upper respiratory infection, arthralgia, nasal congestion, and pain in the back and extremities. Less common side effects (<1%) include oral candidiasis, cough, dry mouth and throat, nausea, chest discomfort, and dysphonia. Discontinuation rates were greater in patients taking ciclesonide 160 mcg once daily compared with ciclesonide 320 mcg twice daily owing to an increase in asthma exacerbations.5,6
Efficacy: In the U.S., ciclesonide is approved for twice-daily administration for asthma maintenance. In other countries, the drug is approved for once-daily administration, and many published clinical trials have investigated this frequency of administration. A 16-week, randomized, double-blind, placebo-controlled study examined 691 patients with mild-to-moderate persistent asthma who had been treated with bronchodilator therapy only. Patients were randomized to one of four groups: ciclesonide 160 mcg once daily in the morning for 16 weeks; ciclesonide 80 mcg twice daily for 16 weeks; ciclesonide 80 mcg twice daily for 4 weeks followed by 160 mcg once daily in the morning for 12 weeks; or placebo for 16 weeks. All ciclesonide regimens showed statistical improvement in predose forced expiratory volume in 1 second (FEV1) by week 16 compared with placebo. Morning predose FEV1 was greater in patients treated with 80 mcg twice daily versus those taking 160 mcg once daily. Respective increases in morning predose FEV1 for ciclesonide 160 mcg once daily, 80 mcg twice daily for 16 weeks, and 80 mcg twice daily for 4 weeks followed by 160 mcg once daily for 12 weeks were 5%, 10.4%, and 5%.5
A phase III, randomized, placebo-controlled trial evaluated 141 patients with severe, persistent, prednisone-dependent asthma each week to determine whether their prednisone dose could be reduced. The use of ciclesonide 320 mcg twice daily and 640 mcg twice daily led to a 47% and 63% reduction in prednisone dose, respectively, as opposed to a 4% increase in prednisone dose with placebo.7
Another randomized, open-label study compared ciclesonide 320 mcg twice daily and fluticasone propionate 330 mcg twice daily in 528 patients with moderate-to-severe asthma. Pulmonary function, peak flow, asthma symptoms, exacerbations requiring oral steroids, use of rescue medications, discontinuation rates, and quality of life were assessed. The authors concluded that there were no significant differences between the two corticosteroids in the measured outcomes.8
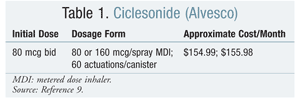
Price: The monthly cost of ciclesonide metered dose inhaler (MDI) is based on 60 actuations per canister (TABLE 1).9 In comparison, the cost of mometasone (Asmanex Twisthaler) is based on 120 actuations per canister. A single canister of mometasone will last 4 months when administered at 220 mcg once daily or 2 months when administered at 440 mcg twice daily; it is available for $210.19 for 120 actuations.10 Although mometasone is available only in 120-actuation increments, this equates to $52.55/month for once-daily dosing and $105.10/month for twice-daily dosing.
Simplicity: Ciclesonide is inhaled twice daily, which is similar to other inhaled corticosteroids that range from one to four inhalations per day. The inhaler should be primed prior to first use or if it has not been used for more than 10 days. The inhaler has an indicator display window that counts down the number of inhalations left in the canister. Patients should use ciclesonide like most oral inhalers, with the exception that it does not need to be shaken prior to use. Patients should also be instructed to rinse the mouth with water after each dose to avoid thrush.5
Place in Therapy: ICS are the most potent and effective anti-inflammatory medications available. Low-dose ICS, which are the preferred maintenance medications, are added to a patient's quick-relief, short-acting beta2-agonist once the patient has progressed from intermittent to mild persistent asthma. As a patient progresses to moderate or severe persistent asthma, the ICS dose generally is increased before other medications are added to the regimen. ICS for asthma therapy include beclomethasone, budesonide, ciclesonide, flunisolide, fluticasone, mometasone, and triamcinolone.11
Budesonide/Formoterol
Budesonide/formoterol fumarate dihydrate (Symbicort; AstraZeneca), a combination MDI, was approved by the FDA in 2006 for the maintenance treatment of asthma in patients 12 years of age and older. Budesonide is a corticosteroid with anti-inflammatory properties, and formoterol is a long-acting beta2-agonist (LABA) that functions as a bronchodilator.12
Safety: Budesonide/formoterol is not indicated for the treatment of acute bronchospasm or status asthmaticus, and it is contraindicated in patients with known hypersensitivity to budesonide or formoterol.12 All LABAs carry a black box warning owing to their potential to increase the risk of asthma-related death.12 In February 2010, because of safety concerns, the FDA mandated a risk-management strategy and class-labeling changes for all LABA-containing products, including a revised medication guide for patients and a plan to educate health care professionals about the appropriate use of LABAs. The requirement is based on findings from several clinical studies--including the Salmeterol Multicenter Asthma Research Trial, the Salmeterol Nationwide Surveillance study, and an FDA-initiated meta-analysis of 110 additional studies--indicating an increased risk of severe exacerbation of asthma symptoms leading to hospitalization, and even death, in some patients using LABAs for the prophylactic treatment of asthma.13-15
Similar to other corticosteroids, precautions should be taken owing to the potential for worsening adrenal function, immunosuppression, decreased BMD, glaucoma, intraocular pressure, cataracts, and adverse effects on growth. The safety and efficacy of budesonide/formoterol has not been established in pediatric patients aged less than 12 years. Budesonide/formoterol is classified as pregnancy category C, and budesonide has been shown to be secreted in human milk; therefore, it should not be used in nursing women.12 Drug interactions that warrant caution include strong CYP450 3A4 inhibitors, tricyclic antidepressants, beta-blockers, and diuretics.12
Tolerability: Common adverse reactions experienced by patients (>3%) using budesonide/formoterol 80/4.5 mcg or 160/4.5 mcg twice daily include nasopharyngitis, headache, upper respiratory tract infections, pharyngolaryngeal pain, sinusitis, influenza, and back pain. Less common side effects include nasal congestion, stomach discomfort, vomiting, and oral candidiasis.12 Patients should be informed of adverse effects associated with LABAs, such as palpitations, chest pain, rapid heart rate, tremor, and nervousness.12
Efficacy: Budesonide/formoterol is approved for twice-daily administration as maintenance therapy for asthma. In a 6-month, multicenter, parallel-group, double-blind, double-dummy, randomized trial, 1,391 patients aged 12 years and older with persistent asthma who had experienced an exacerbation within the past year received 2 inhalations of budesonide/formoterol 160/4.5 mcg twice daily or 1 inhalation of salmeterol/fluticasone 500/50 mcg twice daily. Patients' mean rate of exacerbations was similar in both treatment groups. There was a 30% lower annual rate of moderate or severe exacerbations in the salmeterol/fluticasone group compared with the budesonide/formoterol group. The authors concluded that asthma symptoms and lung function were improved in both treatment groups; however, salmeterol/fluticasone was significantly superior to budesonide/formoterol in reducing the rate of exacerbations.16
Price: The monthly cost of budesonide/formoterol MDI is based on 120 inhalations per canister (TABLE 2).17 In comparison, the other available combination MDI, fluticasone/salmeterol hydrofluoroalkane (Advair HFA)--available in strengths of 45/21 mcg, 115/21 mcg, and 230/21 mcg--is administered as 1 puff twice daily and costs, respectively, $187.95, $219.40, and $269.91 for a 1-month supply.18

Simplicity: Budesonide/formoterol is administered as 2 inhalations twice daily. The inhaler should be primed prior to first use or if it has not been used for more than 7 days. Similar to ciclesonide, the inhaler has an indicator display window that counts down the number of inhalations left in the canister. Patients should use the inhaler like other MDIs and should shake the inhaler for approximately 5 seconds prior to each use. As with all ICS, patients should be instructed to rinse the mouth with water after each dose to avoid thrush.12
Place in Therapy: Of the adjunctive therapies available, LABAs are the preferred choice to combine with ICS in patients aged 12 years and older.11 LABAs are never to be used as monotherapy. Combination products containing an ICS and an LABA include fluticasone/salmeterol (Advair) and budesonide/formoterol (Symbicort).12
Omalizumab
Omalizumab (Xolair; Genentech) is a novel injectable recombinant DNA-derived humanized immunoglobulin G monoclonal antibody that binds to human immunoglobulin E (IgE). It was approved by the FDA in 2003 for maintenance treatment of moderate-to-severe persistent asthma in patients aged 12 years and older with a positive skin test or in vitro reactivity to a perennial aeroallergen and symptoms that are inadequately controlled with ICS.19 Omalizumab inhibits the binding of IgE to the high-affinity receptor on the surface of mast cells and basophils, which limits the release of allergic response mediators and reduces serum free IgE levels. In clinical studies, total IgE levels did not return to pretreatment levels for up to 1 year after discontinuation of omalizumab.19
Safety: The product carries a black box warning that anaphylaxis presenting as bronchospasm, hypotension, syncope, urticaria, and/or angioedema of the throat or tongue has been reported after administration. Anaphylaxis can occur as early as after the first dose, but also has occurred beyond 1 year after starting regularly scheduled treatment, and its frequency is estimated at 0.2%.19 A task force investigated the actual incidence of anaphylaxis and determined that postmarketing reports from 2003 to 2005 corresponded to a rate of 0.09% of patients.20 Task force recommendations include observing the patient for 2 hours for the first three injections and 30 minutes thereafter and educating the patient regarding the signs and treatment of anaphylaxis, including the proper use of the epinephrine autoinjector. Omalizumab is contraindicated in patients with a history of severe hypersensitivity reaction.19
Malignant neoplasms have been observed in 0.5% of omalizumab-treated patients. Omalizumab has not been shown to alleviate asthma exacerbations acutely, so is not indicated to treat acute bronchospasm or status asthmaticus. Some patients who have had a reduction of oral corticosteroid therapy may present with systemic eosinophilia, vasculitic rash, worsening pulmonary symptoms, cardiac complications, or neuropathy.19 Omalizumab is classified as pregnancy category B, and caution should be exercised in nursing women. The concomitant use of omalizumab with other medications or allergen immunotherapy has not been evaluated.19
The FDA is currently evaluating interim safety findings from the ongoing study Evaluating the Clinical Effectiveness and Long-Term Safety in Patients with Moderate to Severe Asthma (EXCELS), an observational study of approximately 5,000 omalizumab-treated patients and a control group of 2,500. The primary objective of EXCELS is to assess the long-term safety profile of omalizumab in patients followed for 5 years. Interim data suggest a disproportionate increase in ischemic heart disease; arrhythmias; cardiomyopathy and cardiac failure; pulmonary hypertension; cerebrovascular disorders; and embolic, thrombotic, and thrombophlebotic events.21 At this time, the FDA is not recommending any changes to the prescribing information for omalizumab and is not advising patients to stop taking the drug.
Tolerability: The adverse events most frequently resulting in clinical intervention were injection-site reaction (45%); viral infections (23%); upper respiratory tract infection (20%); sinusitis (16%); headache (15%); and pharyngitis (11%). The most common adverse reactions occurring in more than 1% of omalizumab-treated patients include body, leg, and arm pain; arthralgia; fatigue; dizziness; dermatitis; and earache.19
Efficacy: Omalizumab reduces the rate of serious asthma exacerbations and the need for ED treatment and hospitalization in asthma patients.22 The safety and efficacy of omalizumab were originally evaluated in three randomized, double-blind, placebo-controlled, multicenter trials using a maximum dose of 750 mg for 4 weeks.19 Recently, observational studies have been initiated to determine safety and effectiveness in clinical practice. The PERSIST study, a prospective, open-label, observational, multicenter study of severe persistent allergic asthma, assessed the 16-week and 52-week effectiveness of add-on omalizumab treatment using physician-rated global evaluation of treatment effectiveness (GETE).23 At both 16 and 52 weeks, significant improvement was observed, as evidenced by good/excellent GETE, better quality of life, percentage of patients free of severe exacerbations, and reductions in healthcare utilization.23 As mentioned previously, the FDA is following the EXCELS study, an ongoing multicenter, prospective, observational cohort study that will provide information regarding the clinical safety and effectiveness of omalizumab in a real-world clinical-practice setting.24
Price: The wholesale acquisition cost of one 150-mg vial of omalizumab was $649.16 as of 2008 (TABLE 3).25 A patient would pay approximately $10,000 to $20,000 annually for omalizumab therapy, excluding health care provider administration fees; this rate is much higher than for other available asthma medication regimens. Genentech offers financial assistance for health care providers and patients through its Xolair Access Solutions program.

Simplicity: Dose (150-375 mg) and dosing frequency (2 or 4 weeks) are determined by baseline serum total IgE level (30-700 IU/mL) and body weight (30-150 kg). Patients must have a positive skin test or in vitro reactivity to a perennial aeroallergen with asthma symptoms that are inadequately controlled with ICS.19 Upon initiation of omalizumab, ICS should not be abruptly discontinued, and no more than 150 mg should be subcutaneously injected per site, with a maximum dose of 375 mg every 2 weeks.19 Administration should occur only in a health care provider's office to facilitate observation and treatment for anaphylaxis.
Place in Therapy: Omalizumab is the only adjunctive therapy to demonstrate added efficacy to high-dose ICS plus LABA in patients who have severe persistent allergic asthma; therefore, it may be considered as additional therapy for severe persistent asthma patients aged 12 years and older who are not controlled on high-dose ICS plus LABA.11 From a pharmacoeconomic standpoint, omalizumab should be used only in patients with allergic asthma who are poorly controlled despite maximal therapy.26
REFERENCES
1. Akinbami L. Centers for Disease Control and Prevention (CDC). Asthma prevalence, health care use and mortality: United States, 2003-05.
www.cdc.gov/nchs/data/hestat/
2. American Academy of Allergy, Asthma & Immunology. Asthma statistics.
www.aaaai.org/media/resources/
3. CDC. Asthma.
www.cdc.gov/nchs/fastats/
4. Shaughnessy AF. STEPS drug updates [editorial]. Am Fam Physician. 2003;68:2342-2348.
5. Alvesco (ciclesonide) package insert. Marlborough, MA: Sepracor Inc; July 2008.
6. Bateman ED, Cheung D, Lapa e Silva J, et al. Randomized comparison of ciclesonide 160 and 640 mcg/day in severe asthma. Pulm Pharmacol Ther. 2008;21:489-498.
7. Bateman E, Karpel J, Casale T, et al. Ciclesonide reduces the need for oral steroid use in adult patients with severe, persistent asthma. Chest. 2006;129:1176-1187.
8. Bateman ED, Linnhof AE, Homik L, et al. Comparison of twice daily ciclesonide and fluticasone propionate in patients with moderate-to-severe persistent asthma. Pulm Pharmacol Ther. 2008;21:264-275.
9. Drugstore.com. Alvesco.
www.drugstore.com/pharmacy/
10. Drugstore.com. Asmanex 120 metered doses.
www.drugstore.com/pharmacy/
11. National Heart Lung and Blood Institute. Expert Panel Report 3: guidelines for the diagnosis and management of asthma. NIH Publication 07-4051.
www.nhlbi.nih.gov/guidelines/
12. Symbicort (budesonide/formoterol) package insert. Wilmington, DE: AstraZeneca LP; February 2009.
13. Food and Drug Administration (FDA). FDA drug safety communication: new safety requirements for long-acting inhaled asthma medications called Long-Acting Beta-Agonists (LABAs).
www.fda.gov/Drugs/DrugSafety/
14. Castle W, Fuller R, Hall J, Palmer J. Serevent nationwide surveillance study: comparison of salmeterol with salbutamol in asthmatic patients who require regular bronchodilator treatment. BMJ.
15. Nelson HS, Weiss ST, Bleecker ER, et al. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest.
16. Dahl R, Chuchalin A, Gor D, et al. EXCEL: a randomised trial comparing salmeterol/fluticasone propionate and formoterol/budesonide combinations in adults with persistent asthma. Respir Med.
17. Drugstore.com. Symbicort.
www.drugstore.com/pharmacy/ 1993;306:1034-1037. 2006;129:15-26. 2006;100:1152-1162.
18. Drugstore.com. Advair HFA.
www.drugstore.com/pharmacy/
19. Xolair (omalizumab) package insert. South San Francisco, CA: Genentech, Inc; January 2010.
20. Cox L, Platts-Mills TA, Finegold I, et al. American Academy of Allergy, Asthma & Immunology/American College of Allergy, Asthma and Immunology Joint Task Force report on omalizumab-associated anaphylaxis. J Allergy Clin Immunol. 2007;120:1373-1377.
21. FDA. Omalizumab (marketed as Xolair) information.
www.fda.gov/Drugs/DrugSafety/
22. Corren J, Casale T, Deniz Y, et al. Omalizumab, a recombinant humanized anti-IgE antibody, reduces asthma-related emergency room visits and hospitalizations in patients with allergic asthma. J Allergy Clin Immunol. 2003;111:87-90.
23. Brusselle G, Michils A, Louis R, et al. “Real-life” effectiveness of omalizumab in patients with severe persistent allergic asthma: the PERSIST study. Respir Med. 2009;103:1633-1642.
24. Long AA, Fish JE, Rahmaoui A, et al. Baseline characteristics of patients enrolled in EXCELS: a cohort study. Ann Allergy Asthma Immunol. 2009;103:212-219.
25. Red Book 2008. Montvale, NJ: Thomson Healthcare; 2008.
26. Oba Y, Salzman GA. Cost-effectiveness analysis of omalizumab in adults and adolescents with moderate-to-severe allergic asthma. J Allergy Clin Immunol. 2004;114:265-269.
To comment on this article, contact rdavidson@uspharmacist.com.






