US Pharm. 2021;46(7):43-47.
ABSTRACT: Eosinophilic asthma (EA) is a severe subphenotype of asthma. Patients with EA historically suffer from frequent and severe asthma exacerbation, declining lung function, and oral-steroid dependency, as well as decreased quality of life and the risk for poor health outcomes. Biologic therapies target the eosinophil inflammatory response, and they have been found to reduce asthma exacerbations and hospitalizations, decrease oral-corticosteroid use, improve lung function, and result in better overall quality of life for these refractory patients.
Asthma is a chronic, inflammatory, respiratory disease affecting over 339 million persons globally with nearly 25 million people affected in the United States—7.5% of whom are children.1,2 It is characterized by hallmark symptoms such as wheezing, shortness of breath, chest tightness, and cough, which vary in frequency and intensity. Respiratory symptoms of asthma are usually triggered by environmental factors, such as indoor and outdoor allergens, chemical irritant exposures, air pollution, and tobacco smoke. Additional triggers include certain medication (e.g., aspirin, nonsteroidal anti-inflammatory drugs, and beta blockers), extreme changes in weather, emotions, and physical exercise.3,4 Since asthma can manifest in different ways and at varying times during life, it is now recognized as a heterogenous disease that involves both genetic and environmental factors. With each phenotype of the disease, airway inflammation is uniquely driven by a specific pathophysiologic mechanism, or endotype, that may render conventional therapies ineffective. In persons with chronic conditions, this limitation in airflow may become persistent, without regard to symptoms or lung function, but lessens with treatment.3
To achieve optimal asthma control in patients, a better understanding of phenotypes is necessary using targeted therapies, especially benefitting severe refractory asthma that does not respond to standard treatments. This population represents approximately 10% of all persons with asthma who have increased morbidity and overall healthcare costs due to their disease.5
Eosinophilic Asthma
Esinophilic asthma (EA) is a severe asthma phenotype. It typically presents in adults but can also be seen in children and young adults. The cause is unknown, as most of these individuals do not have underlying allergic triggers. As the name suggests, persons with severe EA have increased eosinophils found in the sputum with underlying type 2 inflammation. Type 2 inflammation is attributed to cytokine-producing T-cells and interleukin (IL)-4, IL-5, and IL-13, stimulating the formation and release of eosinophils from bone marrow in response to allergen exposure. Exposure to viruses, bacteria, and irritants also activates the immune system, producing IL-33, IL-25, and thymic stromal lymphopoietin in epithelial cells. This increased eosinophil production increases mucus production, airway hyperresponsiveness, and airway remodeling. The presence of excess eosinophils is an indicator for poor asthma control and marker of disease severity. Airway obstruction in EA patients also involves the sinus and small or distal airways. In addition to severe and persistent wheezing, coughing, shortness of breath, difficulty breathing, chest tightness, and airflow obstruction, these individuals are also affected by chronic sinus disease, inflamed nasal mucosa, and nasal polyps (TABLE 1).3-6

Diagnosis
Eosinophil levels can be measured in the sputum, blood, and exhaled fractional nitric oxide (FeNO) concentration in the breath. Noninvasive, patient-induced sputum is the most reliable measure of eosinophil cells, despite the difficulty in obtaining from patients in clinical settings. Sputum eosinophils counts >1%–3% correlates with severe disease and is a marker for biologic treatments, which have shown to reduce the rate of asthma exacerbations.3,4,7
Peripheral blood eosinophil counts can also be used to determine the presence of severe EA likely requiring biologic treatment. Elevated blood levels ≥150 cells/µL are a good indicator for airway eosinophilia. In contrast to sputum eosinophils, low blood levels do not accurately reflect absence of airway eosinophilia and, therefore, correlate poorly to sputum eosinophil levels. Blood eosinophil levels are also affected by high-dose inhaled and oral corticosteroids. Hence, although peripheral blood eosinophilia may be a marker of disease severity in asthma, it does not correlate consistently with sputum eosinophilia.3,4,7
FeNO is another marker of airway inflammation and is measured through breath tests. Measurements are rapid, simple, and noninvasive. Nitric oxide (NO) is a reactive molecule synthesized by NO synthase expressed on epithelial cells in the airway. An FeNO level ³20 ppb is a good indicator for sputum eosinophilia. While NO levels correlate with airway inflammation, it is not solely linked to eosinophils but rather epithelial cells and macrophages.
Smoking, allergic rhinitis, and female gender can also give inconclusive results. As a result, a combination of biomarkers (sputum eosinophils ³2%, and/or blood eosinophils ³150 cells/µL, and/or FeNO ³20 ppb) may be better than using one alone to achieve a greater predictor of EA and its severity.3,4,7
Management
Most persons with asthma respond well to properly utilized, guideline-based medications. However, an estimated 10% to 20% of these individuals may be refractory to the standards of treatment (including inhaled and oral corticosteroids) and have severe asthma such as EA.2,3 Due to the underlying type 2 inflammation in most severe cases, maintenance therapy with high-dose inhaled corticosteroids (ICS) or daily oral corticosteroids (OCS) may be used for symptom control, minimizing exacerbations, and maintaining normal activity.3
In certain patients, refractory type 2 inflammation may occur and is suspected when initial assessment reveals: 1) levels of blood eosinophils >150 cells/µL; and/or 2) FeNO >20 ppb; and/or 3) sputum eosinophil level >2%; and/or 4) asthma is clinically allergen-driven. It is recommended that blood eosinophil and FeNO levels be repeated up to three times when asthma is worsening, prior to the initiation of OCS and consideration of non–type 2 asthma, as maintenance OCS may cause suppression of the aforementioned biomarkers; therefore, measurements should be taken either before OCS therapy is initiated or with the lowest possible OCS dose.3
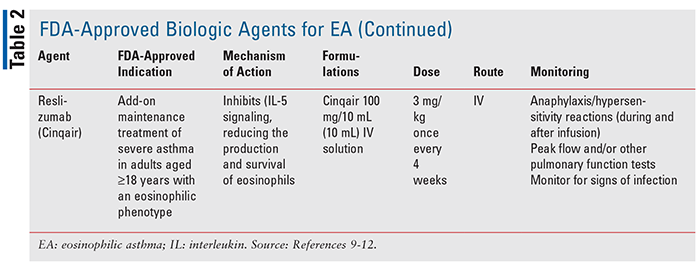
For the management of EA in persons who have not adequately responded to maintenance therapy with high-dose ICS, OCS, or bronchodilators, a type 2 targeted biologic agent may be an option. The selection of a biologic agent for EA is individualized and must include the consideration of factors such as dosing frequency, method of administration, and adverse effects (TABLE 2). Currently, there are four FDA-approved biologic therapies to treat EA: benralizumab, dupilumab, mepolizumab; and reslizumab.3,5 These agents require testing for parasitic infections (and treatment if warranted) prior to initiation as eosinophilia may be a consequence of such infections. Use of benralizumab, dupilumab, mepolizumab, or reslizumab in persons with untreated parasitic infections can lead to disseminated disease.
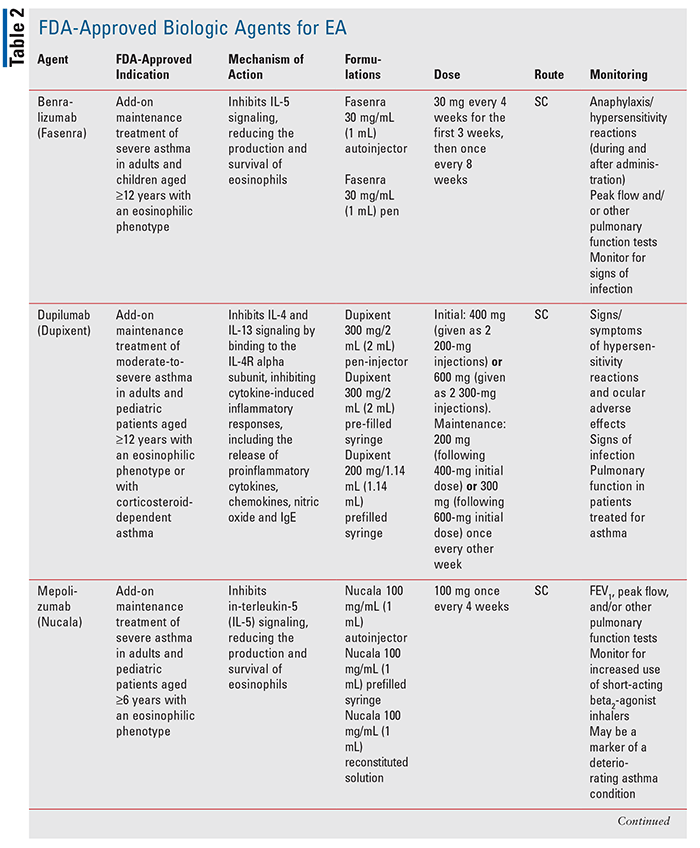
It is important to note that omalizumab has also been found to reduce airway and blood eosinophils, reduce asthma exacerbations, and decrease OCS use in children and adults with asthma; however, this monoclonal antibody aims to reduce serum IgE levels and is indicated for the treatment of moderate-to-severe persistent asthma in patients aged 6 years or older with a positive skin test or in-vitro reactivity to a perennial aeroallergen and symptoms that are inadequately controlled by ICS.5,8 Although a decrease in IgE may result in a reduction in eosinophilia, most persons with EA do not have an IgE-mediated allergy and will likely not benefit from treatment with omalizumab.5
Benralizumab
Benralizumab (Fasenra) is a monoclonal antibody that selectively targets the IL-5 receptor alpha subunit on eosinophils, which leads to rapid and direct depletion of the eosinophils. It is indicated for use as adjunctive maintenance treatment for severe asthma (including EA) in patients aged 12 years or older. This biologic agent can be self-administered via SC injection at a dosage of 30 mg every 4 weeks for the first three doses and every 8 weeks thereafter. It is available as a prefilled syringe and an autoinjector.9
Patients should not discontinue corticosteroid therapy abruptly when starting benralizumab but rather discuss the appropriateness of gradual discontinuation with their healthcare provider. Headache and pharyngitis are among the most reported adverse effects. Hypersensitivity reactions are also possible and include anaphylaxis, angioedema, urticaria, and rash. FDA approval of this biologic agent in 2017 was based on three phase III studies that demonstrated up to a 51% reduction in annual asthma exacerbation rates compared with placebo, significant improvement in lung function, mean reduction of daily OCS use of 75%, discontinuation of OCS use in 52% of patients, and safety profile similar to placebo.9,13
Dupilumab
Dupilumab (Dupixent) is an IL-4 receptor alpha antagonist that was approved by the FDA in 2018 as an add-on maintenance treatment for moderate-to-severe asthma in patients aged 12 years or older with an eosinophilic-phenotype or OCS-dependent asthma. It is administered SC and is available as a prefilled syringe and a prefilled pen. The initial dosage for patients with asthma is 400 mg followed by 200 mg every other week or 600 mg followed by 300 mg every other week. In patients with comorbid moderate-to-severe atopic dermatitis (for which dupilumab is also indicated) or concomitantly on OCS, the initial dose is the latter (600 mg, followed by 300 mg every other week).
The most reported adverse reaction in patients with asthma have been injection-site reactions, oropharyngeal pain, and eosinophilia. Hypersensitivity reactions can also occur and include anaphylaxis, urticaria, rash, erythema nodosum, and serum sickness. Patients who are on corticosteroids should not discontinue therapy abruptly when initiating dupilumab; gradual discontinuation should be discussed with a healthcare provider. In addition, patients on dupilumab should avoid live vaccines, report any new or worsening ocular symptoms due to the potential for conjunctivitis and keratitis, and watch for any signs of eosinophilic conditions such as vasculitic rash, worsening pulmonary symptoms, and/or neuropathy (especially with OCS dose reductions).10
FDA approval of dupilumab for OCS-dependent asthma and moderate-to-severe asthma in patients with eosinophilic phenotype was based on three randomized, placebo-controlled, multicenter trials. It was found to improve lung function by up to 33%, reduce the need for OCS by 70%, and decrease exacerbations by up to 67% compared with placebo. It was also reported that more than 50% of patients treated with dupilumab eliminated the use of OCS. In addition, the efficacy of dupilumab improved in patients with higher eosinophil levels.10,14
Mepolizumab
In 2015, mepolizumab (Nucala) was approved by the FDA as the first anti–II-5 adjunctive treatment for severe asthma in patients aged 12 years or older with an eosinophilic phenotype at a dosage of 100 mg SC once every 4 weeks.15 In 2019, it became the first biologic treatment approved for children aged 6 to 11 years with severe eosinophilic asthma at a dosage of 40 mg SC once every 4 weeks.16 It is available as lyophilized powder in a single-dose vial for reconstitution, prefilled syringe, and autoinjector.
As with the other biologic agents, corticosteroid therapy should not be abruptly discontinued when starting mepolizumab; if appropriate, gradual discontinuation may be considered. Headache, injection-site reaction, back pain, and fatigue have been the most reported adverse reactions. Possible hypersensitivity reactions include anaphylaxis, angioedema, brochospasm, hypotension, urticaria, and rash.11 FDA approval of mepolizumab in adults, adolescents, and children was based on three double-blind, randomized, placebo-controlled studies in which patients receiving the biologic agent had fewer exacerbations requiring hospitalization and/or emergency department visits, longer time to first asthma exacerbation, and greater reductions in daily OCS doses compared with patients receiving placebo.11,15,16
Reslizumab
Reslizumab (Cinqair) is an IL-5 antagonist monoclonal antibody (IgG4 kappa) that is indicated for adjunctive maintenance treatment of severe asthma in patients aged 18 years or older with an eosinophilic phenotype. The recommended dosage is 3 mg/kg once every 4 weeks via IV infusion over 20 to 50 minutes. Reslizumab should be administered by a healthcare provider in a clinical setting, and patients should be observed after infusion as anaphylaxis has occurred in 0.3% of patients in clinical trials. The most common adverse reaction reported with the use of reslizumab has been oropharyngeal pain. Malignancies have also been observed in clinical studies and are included in warnings and precautions for use. Similar to the other biologic agents used in the treatment of EA, corticosteroids should not be discontinued abruptly with the initiation of reslizumab, although gradual discontinuation may be discussed with a healthcare provider.12
Reslizumab was approved by the FDA in 2016 based on data from four double-blind, randomized, placebo-controlled trials. Compared with placebo, patients with severe asthma were found to have fewer asthma exacerbations and longer times to first exacerbation. There was also a significant improvement in lung function within the active treatment groups and clinically important improvements in patient-reported outcomes (e.g., quality of life).12,17
Role of the Pharmacist
Pharmacists play a critical role in the selection of treatment for EA, as well as the education and assessment of patients with EA. Although the benefits and risks of biologic therapy for EA must be carefully weighed, other patient factors must also be carefully considered, such as affordability, access, and preference. Pharmacists should also assess adherence to ICS treatment (e.g., correct inhaler use/technique) and rule out other causes of asthma exacerbation (e.g., respiratory comorbidities, infectious diseases) prior to adjunctive treatment recommendations for severe EA. In addition, pharmacists can assist patients with referrals to support services as well as psychological services to manage emotional, social, and other burdens of EA and its management.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.REFERENCES
1. World Health Organization. Asthma. World Health Organization. www.who.int/news-room/fact-sheets/detail/asthma. Accessed March 15, 2021.
2. CDC. Most recent national asthma data. www.cdc.gov/asthma/most_recent_national_asthma_data.htm. Accessed March 15, 2021.
3. Global Initiative for Asthma. Global strategy for asthma management and prevention, 2021. www.ginasthma.org. Accessed June 24, 2021.
4. Walford HH, Doherty TA. Diagnosis and management of eosinophilic asthma: a US perspective. J Asthma Allergy. 2014;7:53-65.
5. American Partnership for Eosinophilic Disorders. Eosinophilic asthma. https://apfed.org/about-ead/eosinophilic-asthma/. 2021. Accessed March 15, 2021.
6. Possa SS, Leick EA, Prado CM, et al. Eosinophilic inflammation in allergic asthma. Front Pharmacol. 2013;4:46.
7. Bakakos A, Loukides S, Bakakos P. Severe eosinophilic asthma. J Clin Med. 2019;8(9):1375.
8. Xolair (omalizumab) [package insert]. South San Francisco, CA: Genentech USA, Inc; April 2021.
9. Fasenra (benralizumab) [package insert]. Wilmington, DE: AstraZeneca Pharmaceuticals LP; October 2019.
10. Dupixent (dupilumab) [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals, Inc; January 2021.
11. Nucala (mepolizumab) [package insert]. Research Triangle Park, NC: GlaxoSmithKline LLC; September 2019.
12. Cinqair (reslizumab) [package insert]. West Chester, PA: Teva Respiratory, LLC; February 2020.
13. AstraZeneca Media. Fasenra (benralizumab) receives US FDA approval for eosinophilic asthma. www.astrazeneca.com/media-centre/press-releases.html. 2017. Accessed March 15, 2021.
14. Regeneron. FDA approves asthma indication for Dupixent (dupilumab). https://investor.regeneron.com/. 2018. Accessed March 15, 2021.
15. GlaxoSmithKline. GSK’s Nucala (mepolizumab) receives approval from US FDA. www.gsk.com/en-gb/media. 2015. Accessed March 15, 2021.
16. GlaxoSmithKline. Nucala is the first biologic approved in the US for six to 11-year-old children with severe eosinophilic asthma. www.gsk.com/en-gb/media. 2019. Accessed March 15, 2021.
17. FDA. FDA approved Cinqair to treat severe asthma. www.fda.gov/news-events/press-announcements/fda-approves-cinqair-treat-severe-asthma. 2016. Accessed March 15, 2021.





