US Pharm. 2021;46(6):HS11-HS16.
ABSTRACT: Male breast cancer (MBC) comprises less than 1% of breast carcinomas in the United States, but the incidence has been slowly rising. Despite lower incidence rates, male patients present with more progressive disease and face higher mortality rates compared with female breast cancer (FBC) patients. Although treatment strategies for MBC are largely derived from therapeutic trials involving FBC, new evidence is being used to guide clinical decisions for MBC. Pharmacist recognition of the clinical signs and symptoms of MBC may lead to earlier detection and improved patient outcomes. Additionally, the pharmacist can counsel on therapies and side-effect management. In the health-system setting, the highest priority is to optimize treatment and minimize adverse drug events.
Male breast cancer (MBC) comprises less than 1% of breast carcinomas in the United States, but the incidence has been slowly rising over the years.1,2 Because of its relatively low rate of occurrence, MBC research and awareness have been minimal. Diagnosis and treatment of MBC are largely adapted from evidence on female breast cancer (FBC), but key differences in risk factors and tumor characteristics have been noted.3 Men are more likely to present with more progressive disease, which may contribute to the disparity in survival rates.
Epidemiology and Risk Factors
It is estimated that, in the U.S., MBC will be diagnosed in 2,650 patients and cause 530 deaths in 2021.3 The lifetime risk of breast cancer (BC) in men is significantly lower than in women (1:1,000 vs. 1:8, respectively).1 As with other malignancies, the risk of developing BC increases with age. The average age at diagnosis is 67 years for males, compared with 62 years for females. In the U.S., black males are at higher risk than non-Hispanic white males.4
Although germline mutations increase overall BC risk, men are more likely to present with a BRCA2 mutation (7%-8% risk) than a BRCA1 mutation (1% risk).5 (Interestingly, the reverse is observed in FBC.) BRCA1 and BRCA2 are tumor-suppressing genes that play a role in the encoding of DNA repair proteins. Intrinsic risk factors for MBC are older age, black race, family history, germline genetic mutation (e.g., BRCA2, BRCA1, CHEK2, PALB2), diseases associated with hyperestrogenism (e.g., Klinefelter’s syndrome), liver disease, obesity, and testicular abnormalities; extrinsic risk factors include radiation exposure to the breast or chest and exogenous estrogen use.1,3
Signs and Symptoms
Because routine BC screening is not recommended in men, recognition of the clinical presentation of MBC is imperative. A palpable, firm, painless mass with or without axillary lymphadenopathy is the most common presentation.6 Other signs and symptoms of MBC may include pain; nipple discharge (principally bloody); change in breast shape or size; nipple retraction; ulceration; and changes in color, texture, or topography of the breast, nipple, or areola. Gynecomastia, pseudogynecomastia, lipoma, posttraumatic hematoma, fat necrosis, and epidermal inclusion cysts are benign conditions that may have overlapping presentations with MBC.6
Diagnosis
Gynecomastia, the most common cause of breast masses in men, is coexistent in up to 40% of MBC cases.6 Diagnostic evaluation and follow-up strategies are guided by presenting signs or symptoms and patient age.7,8 For men with bilateral breast enlargement consistent with a benign condition, clinical management is recommended. In patients presenting with asymmetric gynecomastia or an alternative sign of MBC, diagnostic mammography is recommended for those aged 25 years or older; for younger patients, ultrasonography is the initial diagnostic test. Core needle biopsy is conducted if imaging is suspicious or highly suggestive of malignancy. If core needle biopsy cannot be performed, alternatives include fine-needle aspiration and surgical excisional biopsy.7,8
Most BCs are carcinomas, epithelial tumors that develop in cells lining the ducts (ductal carcinoma) or lobules (lobular carcinoma).3 In situ disease means that the tumor does not extend past the basement membrane of a duct or lobule, whereas invasive disease is characterized by infiltration through the membrane. The most common histopathologic type of MBC is invasive ductal carcinoma, followed by ductal carcinoma in situ.3 Lobular carcinomas are seen much less frequently in men than in women (1%-2% vs. ~12%).
In MBC, tumor classification and characterization of a confirmed malignancy are similar to those used for FBC and are based on the American Joint Committee on Cancer (AJCC) TNM staging system (TABLE 1), histopathologic type, and immunohistochemical subtyping.9 TNM stands for primary tumor (T), regional lymph node (N) involvement, and presence of distant metastasis (M). Immunohistochemical staining of the biopsy specimen detects the presence of hormone receptors (HRs)—i.e., estrogen receptor (ER) and progesterone receptor (PR)—and human epidermal growth factor receptor 2 (HER2), which are used in pharmacotherapy determinations. Recent studies replicated previous findings confirming that most BCs in men are ER/PR-positive (luminal intrinsic subtype) but HER2-negative.
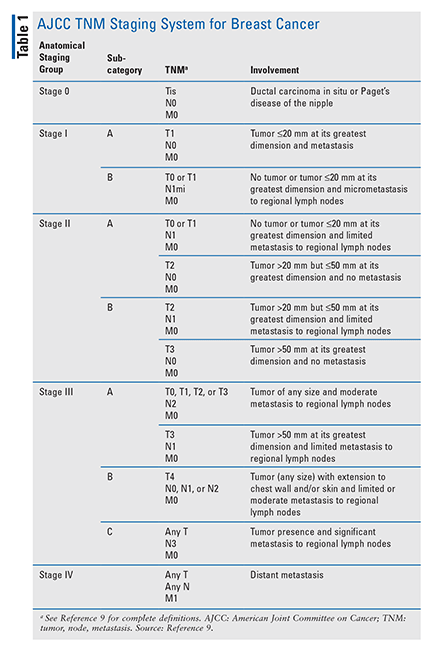
Prognosis
Although BC survivability has improved over the past decades irrespective of sex, conflicting evidence has led to uncertainty regarding similarities in prognosis for MBC versus FBC.10,11 In the U.S., the Surveillance, Epidemiology, and End Results (SEER) database is used to track relative survival rates for cancer. The database does not follow AJCC TNM stages; instead, data are organized into localized, regional, and distant stages. SEER 5-year relative survival rates for BC show a slight variability favoring women (TABLE 2).12
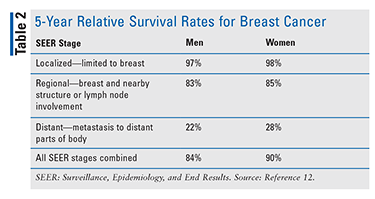
Nonpharmacologic Treatment
Surgery remains a mainstay of treatment for early BC. Historically, total mastectomy with either sentinel lymph node biopsy or axillary lymph node dissection has been considered first-line and the most common surgical approach used in male patients. Based on the recent proliferation of MBC research and the identification of differences in tumor characteristics between men and women, the use of breast-conserving therapy (BCT) in men is now being investigated. BCT (partial mastectomy followed by radiation therapy [RT]) has been shown to be therapeutically equivalent to total mastectomy regardless of sex and has been heavily employed in female patients for years. In one study analyzing the National Cancer Database, the small percentage of male patients who received BCT had greater survival rates, but larger studies are needed to support this finding.13
Indications for postoperative RT in men with BC are the same as for women with BC.14 In the U.S., the percentage of MBC patients who receive RT ranges from 36% to 42%.2,3,13 General indications include BCT, involvement of skin and nipple, positive lymph node, tumor size greater than 5 cm, and positive surgical margins.15
Pharmacologic Treatment
A wide array of systemic therapies are available for BC, with appropriate timing and selection determined by disease stage and tumor characterization. Pharmacologic treatment strategies for MBC follow the same therapeutic principles as for FBC. Perioperative or adjunctive treatment may include endocrine therapy, targeted therapy, chemotherapy (CT), or immunotherapy (TABLE 3).
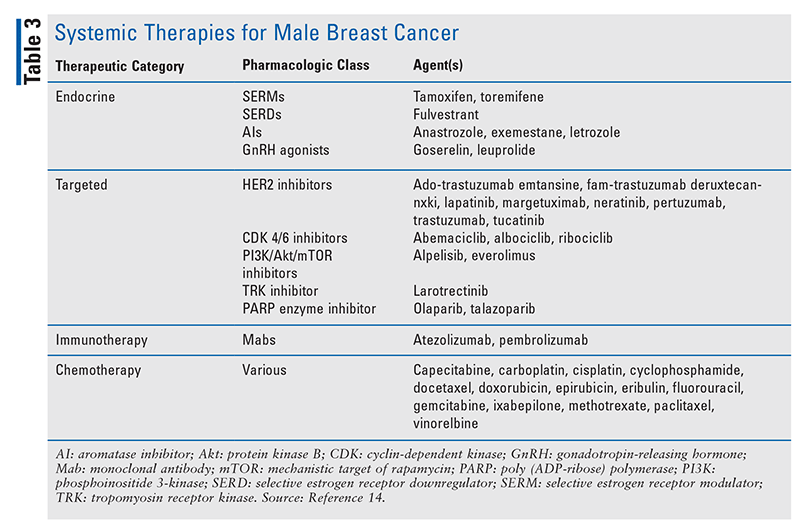
Endocrine Therapy: The presence of HRs is associated with improved outcomes in BC patients. These intracellular receptors that selectively bind estrogen and progesterone will become the target for endocrine (hormone) therapy. Although variations exist in reported rates, 90% or more of MBCs are ER-positive and about 80% are PR-positive, values similar to those for postmenopausal FBC. Despite the eligibility of a large proportion of MBC patients for endocrine therapy, approximately 30% do not receive adjuvant endocrine therapy.16
In men who are candidates for endocrine therapy, tamoxifen should be used preferentially owing to the survival benefit seen in observational studies.1 Tamoxifen, a selective ER modulator, competitively binds to ERs on tumors, causing a decrease in ER signaling–dependent growth in breast tissue. Tamoxifen 20 mg once daily should be used for 5 years, with an additional 5 years offered to patients who are at risk for recurrence.14 Low-dose tamoxifen (5 mg/day for 3 years) may be considered in patients who are symptomatic on the 20-mg dosage or are unwilling or unable to take the standard dosage. Tamoxifen adverse drug effect (ADE) incidence and related drug discontinuation occur at similar or increased rates in men.17 The most notable effects include weight gain, anxiety, insomnia, hot flashes, sexual dysfunction, and thromboembolic events.18 Management of endocrine-therapy ADEs should not include testosterone or androgen supplementation and should follow the approach used in women.1 Venlafaxine has been shown to be effective for treating tamoxifen-induced hot flashes in men.
For patients in whom tamoxifen is contraindicated, use of a gonadotropin-releasing hormone (GnRH) agonist/antagonist and an aromatase inhibitor (AI) should be considered.1 AIs (i.e., anastrozole 1 mg once daily, letrozole 2.5 mg once daily, or exemestane 25 mg once daily) should not be used alone because of the inferior survival in trials comparing tamoxifen with AIs and the lack of complete estradiol suppression. Treatment with AIs also was found to lead to an increase in testosterone and follicle-stimulating hormone, which allowed further aromatization to occur, increasing estrogen and possibly contributing to the demonstrated 1.5-fold increase in mortality.19 AIs are associated with arthralgias and bone-density alterations, whereas GnRH agonists have been associated with metabolic changes (hyperinsulinemia, hypercholesterolemia, weight gain) and increased thrombosis (myocardial infarction and stroke). Bone-modifying agents are indicated only for treatment and prevention of osteoporosis, not for prevention of cancer recurrence. Fulvestrant is used in patients who have not responded to other hormonal therapies; the most common ADE of this agent is hot flashes.
Targeted Therapy: Similar to the use of directed hormone therapy for luminal A and B type BC, targeted therapy is used for cancers that express certain characteristics.20 HER2 has a role in normal cell growth and function; however, in BC, overexpression and amplification are associated with increased tumor size, high tumor grade, lymph node involvement, and elevated cell-proliferation rate. About 10% of male patients present with HER2 overexpression, less than the rate seen in females.
Agents in the HER2-inhibitor class work via several different mechanisms.20 Trastuzumab, pertuzumab, and margetuximab are monoclonal antibodies (Mabs) that bind to the extracellular domain of HER2; antitumor effects are mediated via mechanisms including inhibition of downstream signaling pathways, engagement of antibody-dependent cellular cytotoxicity, and inhibition of receptor dimerization. Common ADEs include fatigue, gastrointestinal upset, and hematologic abnormalities; serious ADEs include infusion-related reactions, cardiotoxicity, and pulmonary toxicity (trastuzumab). The HER2 drug conjugates ado-trastuzumab emtansine and fam-trastuzumab deruxtecan-nxki are Mabs formulated with cytotoxic agents for increased effects. The tyrosine kinase inhibitors (TKIs) neratinib, lapatinib, and tucatinib inhibit signaling by binding to intracellular tyrosine kinase receptors on HER2.21 These oral agents were approved for use following or concurrently with trastuzumab and CT in residual, recurrent, or metastatic HER2-positive BC. Common ADEs associated with TKIs are nausea, vomiting, and diarrhea. Newer agents with alternative targets and immunotherapy are most often used in patients with advanced or metastatic disease. Although these agents have not been systematically evaluated in men, guidelines recommend that they be used as recommended for female patients based on real-world data.
CT: Neoadjuvant CT and adjuvant CT have proven mortality benefits for invasive MBC, and treatment strategies should follow FBC guidelines.15,17 Anthracycline-based CT regimens, in combination with taxanes and cyclophosphamide, are preferred first-line for most patients with HER2-negative disease.14 Observational data suggest an increased incidence of anthracycline-induced cardiotoxicity; however, current evidence is not strong enough to recommend against use. TABLE 3 lists additional agents that may be employed in CT regimens. When indicated, targeted therapy may be employed in conjunction with CT, particularly in HER2-positive cancers. For patients who are ER/PR-positive, endocrine therapy should be given subsequent to CT when both forms of treatment are indicated.
Gene-expression assays such as Oncotype DX, MammaPrint, Prosigna, and EndoPredict are being used to help provide prognostic and therapy-predictive information in BC.14 Oncotype DX, the test preferred by the National Comprehensive Cancer Network, provides a mean recurrence score based on a 21-gene assay, with a scoring range of 0 to 100 used to delineate the therapeutic benefit of CT. When results of the TAILORx trial were extrapolated to men, a score of 26 or higher implied that a benefit would be derived from CT. Available data show that men are more likely to have clearly delineated risk scores (high or low), and scoring accuracy is similar to that for women.3 For this reason, Oncotype DX use should be considered in all MBC patients with early-stage, HR-positive/HER2-negative disease.
The Pharmacist’s Role
Pharmacists are frequently consulted by patients and providers for their expertise. As the most accessible healthcare provider for many patients, the community pharmacist often fields questions regarding health and wellness. Pharmacist awareness and recognition of the clinical signs and symptoms of MBC, along with public education, may lead to earlier detection and improved patient outcomes. Additionally, the pharmacist can counsel on therapies and side-effect management. In the health-system setting, the highest priority is to optimize treatment and minimize ADEs. Data reveal underutilization of endocrine therapy and possible connections to a difference in ADE rates for specific agents. Pharmacists can help ensure the judicious use of CT by recommending genomic testing in appropriate patients.
REFERENCES
1. Hassett MJ, Somerfield MR, Baker ER, et al. Management of male breast cancer: ASCO guideline. J Clin Oncol. 2020;38(16):1849-1863.
2. Sarmiento S, McColl M, Musavi L, et al. Male breast cancer: a closer look at patient and tumor characteristics and factors that affect survival using the National Cancer Database. Breast Cancer Res Treat. 2020;180(2):471-479.
3. Giordano SH. Breast cancer in men. N Engl J Med. 2018;378(24):2311-2320.
4. American Cancer Society. Cancer facts & figures 2021. www.cancer.org/research/cancer-facts-statistics/all-cancer-facts-figures/cancer-facts-figures-2021.html. Accessed March 28, 2021.
5. Ibrahim M, Yadav S, Ogunleye F, Zakalik D. Male BRCA mutation carriers: clinical characteristics and cancer spectrum. BMC Cancer. 2018;18(1):179.
6. Chau A, Jafarian N, Rosa M. Male breast: clinical and imaging evaluations of benign and malignant entities with histologic correlation. Am J Med. 2016;129(8):776-791.
7. National Comprehensive Cancer Network (NCCN). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Breast cancer screening and diagnosis. Version 1.2020. www.nccn.org/professionals/physician_gls/pdf/breast-screening.pdf. Accessed March 30, 2021.
8. Mainiero MB, Lourenco AP, Barke LD, et al. ACR appropriateness criteria evaluation of the symptomatic male breast. J Am Coll Radiol. 2015;12(7):678-682.
9. Amin MB, Edge SB, Greene FL, et al, eds. AJCC Cancer Staging Manual. 8th ed. New York, NY: Springer; 2017.
10. Yadav S, Karam D, Bin Riaz I, et al. Male breast cancer in the United States: treatment patterns and prognostic factors in the 21st century. Cancer. 2020;126(1):26-36.
11. Bender PF, de Oliveira LL, Costa CR, et al. Men and women show similar survival rates after breast cancer. J Cancer Res Clin Oncol. 2017;143(4):563-571.
12. Howlader N, Noone AM, Krapcho M, et al, eds. SEER Cancer Statistics Review, 1975-2017. Bethesda, MD: National Cancer Institute; 2020.
13. Bateni SB, Davidson AJ, Arora M, et al. Is breast-conserving therapy appropriate for male breast cancer patients? A National Cancer Database analysis. Ann Surg Oncol. 2019;26(7):2144-2153.
14. NCCN. NCCN Clinical Practice Guidelines in Oncology. Breast cancer. Version 3.2021. www.nccn.org/professionals/physician_gls/pdf/breast.pdf. Accessed March 31, 2021.
15. Yalaza M, Inan A, Bozer M. Male breast cancer. J Breast Health. 2016;12(1):1-8.
16. Venigalla S, Carmona R, Guttmann DM, et al. Use and effectiveness of adjuvant endocrine therapy for hormone receptor–positive breast cancer in men. JAMA Oncol. 2018;4(10):e181114.
17. Yadav S, Giridhar KV, Taraba J, et al. Safety, efficacy, and tolerability of systemic therapies in male breast cancer: are there sex-specific differences? Expert Opin Drug Saf. 2020;19(8):923-926.
18. Wibowo E, Pollock PA, Hollis N, Wassersug RJ. Tamoxifen in men: a review of adverse events. Andrology. 2016;4(5):776-788.
19. Eggemann H, Ignatov A, Smith BJ, et al. Adjuvant therapy with tamoxifen compared to aromatase inhibitors for 257 male breast cancer patients. Breast Cancer Res Treat. 2013;137(2):465-470.
20. Oh DY, Bang YJ. HER2-targeted therapies—a role beyond breast cancer. Nat Rev Clin Oncol. 2020;17(1):33-48.
21. Yang X, Wu D, Yuan S. Tyrosine kinase inhibitors in the combination therapy of HER2 positive breast cancer. Technol Cancer Res Treat. 2020;19:1533033820962140.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






