ABSTRACT: Bronchiectasis is a progressive pulmonary disorder with an increasing prevalence affecting individuals of any age. It is characterized by permanent dilation of bronchi due to inflammation and chronic infections. Symptoms include chronic cough, excessive sputum production, shortness of breath, and repeated respiratory infections. There is currently no cure. Therefore, treatment is a stepwise approach focusing on reducing and preventing the frequency of exacerbations, managing airway inflammation, improving airway function by managing airway clearance, and preventing the progression of structural lung damage. The pharmacist’s role in managing patients with bronchiectasis includes counseling on smoking cessation, nutritional needs, proper inhaler usage, adverse events, and keeping current on vaccinations.
Bronchiectasis is a complex, heterogeneous disorder characterized by a dysregulated inflammatory response that results in lung damage; abnormal, irreversible dilation of the bronchi; and recurrent bacterial infection. There are many underlying causes of bronchiectasis and—in up to 50% of patients—there has been no identified cause. Idiopathic bronchiectasis and post infective-related bronchiectasis represent the majority of adult cases in non–cystic fibrosis bronchiectasis. Post infective bronchiectasis can be caused by bacterial organisms, most commonly Pseudomonas aeruginosa and Haemophilus influenzae. Other pathogens including fungi, viruses, and mycobacterium can also colonize and/or infect patients with bronchiectasis.1
There are other less common etiologies associated with causing bronchiectasis. These include genetic disease such as cystic fibrosis; secondary lung diseases like chronic obstructive pulmonary disease (COPD); asthma; interstitial lung disease; immunodeficiency disorders (predominantly Alpha-1 antitrypsin deficiency); alterations in the mucociliary escalator like primary ciliary dyskinesia; inflammatory diseases such as rheumatoid arthritis or connective tissue disease; inflammatory bowel disease; and aspiration syndromes such as esophageal reflux and chronic sinusitis.2
Epidemiology
It is estimated that 350,000 to 500,000 adults in the United States have bronchiectasis. The overall prevalence of bronchiectasis is approximately 139 cases per 100,000 U.S. adults aged 18 years or older and is typically higher for women (180 per 100,000) versus men (95 per 100,000).2 The prevalence of bronchiectasis increases substantially with age, from 7 per 100,000 persons aged 18 to 34 years to 812 per 100,000 persons aged 75 years and older.3
The overall annual incidence of bronchiectasis is approximately 29 cases per 100,000 U.S. adults aged 18 years and older and is also higher for women (34 per 100,000) versus men (23 per 100,000). The incidence of bronchiectasis increases substantially with age, from 2 per 100,000 persons aged 18 to 34 years to 154 per 100,000 persons aged 75 years and older.4
Diagnosis
Bronchiectasis should be suspected in any patient with chronic, wet, purulent cough, sputum production, or frequent respiratory infections. Additional factors suggesting the diagnosis include daily sputum production, rhinosinusitis, fatigue, and hemoptysis. Although chronic sputum production is the classic symptom suggesting the diagnosis, patients may have a chronic cough that is nonproductive. Dyspnea, wheezing, pleuritic chest pain, and digital clubbing are also experienced.4
A high-resolution CT is the test of choice for diagnosing bronchiectasis. The main diagnostic feature of bronchiectasis includes bronchial wall thickening, an internal diameter of a bronchus that is wider than its adjacent pulmonary artery, failure of a bronchi to taper as it travels to the periphery of the lung, and mucus plugs completely filling the airway lumen.4,5
Pathophysiology
Current understanding of bronchiectasis pathophysiology is limited. Cole’s “vicious cycle hypothesis” described in 1986 is widely accepted as the paradigm of bronchiectasis development and progression (see FIGURE 1). In this model, impaired mucociliary clearance results in an accumulation of airway secretions that disrupt normal host defenses, leaving the patient more vulnerable to infection. Persistent infection triggers an inflammation response which causes abnormal airway remodeling and structural damage.6 This cycle of events results in a persistent and progressive process with an “entry point” that can be started anywhere within the underlying etiologies of bronchiectasis.
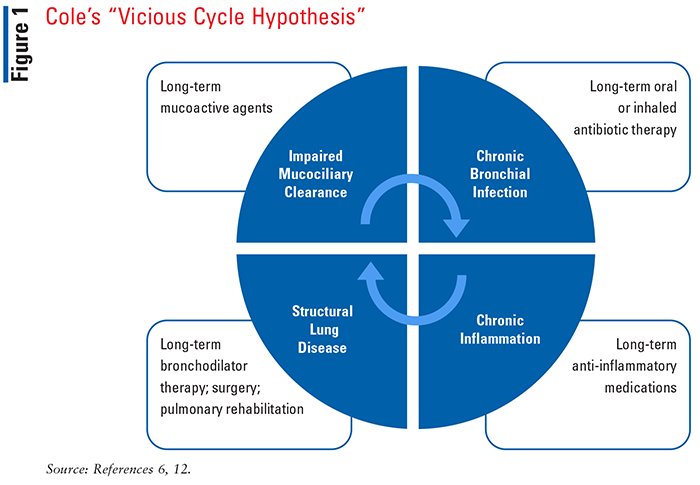
This model was therefore further revised by Flume et al to describe a vicious vortex, where airway dysfunction, airway inflammation, infection, and structural damage are linked.7 As a result, trying to break the cycle with treatments directed to just one entry point has not consistently demonstrated clinical benefits.
Bronchiectasis Exacerbations
There is a need for a clear definition of exacerbations used in patients with bronchiectasis. The identification of specific thresholds for the heterogeneous etiologies and symptoms combined with the minimum time required to start an exacerbation represent important challenges in the clinical management of bronchiectasis.8 A consensus definition offered by Amati et al and Hill et al define an exacerbation as deterioration of ³3 of the following key symptoms for 48 hours or longer: cough, sputum volume and or consistency, sputum purulence, breathlessness and or exercise tolerance, fatigue and/or malaise, hemoptysis, and the need for a change in bronchiectasis treatment as determined by a clinician.8,9
Chalmers et al reviewed 2,572 patients to determine if a prior history of exacerbations could be a predictor of future exacerbations, and as a result develop a clinical phenotype for patients with bronchiectasis.10 They were able to demonstrate that frequent exacerbations were the strongest predictor of future exacerbation frequency. The incident rate ratios for future exacerbations were 1.73 for one exacerbation per year, 3.14 for two exacerbations, and 5.97 for patients with three or more exacerbations per year at baseline.10 Without appropriate interventions, patients with frequent exacerbations will likely continue to exacerbate and have serious effects to quality of life, increased hospitalizations, and a potentially increased risk of mortality.
Treatment of Bronchiectasis
The goals of bronchiectasis treatment include reducing the airway inflammation cascade, elimination of infections, and strengthening of the mucociliary escalator.
Airway Clearance Therapy
The goal of airway clearance therapy is to mobilize bronchopulmonary secretions and interrupt the vicious cycle of inflammation and infection. Airway clearance employs an inhaled agent (mucolytics) in conjunction with chest physiotherapy. This method is used in conjunction with oscillatory positive pressure devices, high frequency chest wall oscillation devices, autogenic drainage, and active cycle breathing with huff coughs or manual chest percussions.4 Hyperosmotic agents, such as nebulized 7% hypertonic saline and mannitol inhaled as a dry powder, are intended to reduce sputum viscosity and osmolality, and as a result aid in the expulsion of sputum from the bronchial tree.
Bronchodilators
Bronchodilators improve ciliary motility and facilitate clearance of secretions, and they are sometimes used before physiotherapy. There is, however, a lack of data supporting the use of bronchodilators in the treatment of bronchiectasis.
Exercise
In a retrospective study of the effects of a pulmonary exercise program in patients with chronic lung disease, patients showed significant improvements not only in exercise capacity, but also required fewer emergency room and outpatient visits, as well as a decreased need for short-acting bronchodilators.11
Anti-inflammatory Therapy
Corticosteroids
The European Respiratory Society guidelines for the management of adult bronchiectasis recommend not offering inhaled corticosteroids for the treatment of bronchiectasis. They can, however, continue to be used in patients being treated with other comorbid conditions such as asthma and COPD.12
Macrolides
Azithromycin has been well documented for its antibacterial bacteriostatic activity. However, it also has a robust immunomodulatory effect that has proven beneficial in a variety of chronic lung illnesses including bronchiectasis. This effect results in decreased production of pro-inflammatory cytokines in the acute phase of inhibition of inflammation and promotes resolution of chronic inflammation in the later phases. Specifically, azithromycin has direct activity on airway epithelial cells to maintain their function and reduce mucus secretion.13
Dosing of azithromycin for use as an anti-inflammatory has been studied in two large trials, specifically the EMBRACE study and the BAT study. Dosing ranged from 500 mg three times a week for 6 months to 250 mg once daily for 12 months. Compared to the control arm, azithromycin demonstrated decreases in exacerbations and improvement in lung function as measured by the forced expiratory volume (FEV1). There was also a significant difference in inflammatory biomarkers like C-reactive protein, peripheral white blood cells, and peripheral neutrophils.4
When using long-term azithromycin therapy as an anti-inflammatory, clinicians must be aware of the true and valid possibility of the development of antibiotic resistance. Azithromycin also has the potential for QTC prolongation, gastrointestinal adverse effects, and the development of tinnitus. Clinicians must weigh the risk versus the benefits on an individual patient basis.
Antibiotics
Antibiotics used to treat bronchiectasis fall under three clinical treatment settings. The first setting is the attempt to eradicate Pseudomonas and/or methicillin-resistant Staphylococcus aureus infections, as these are the most common pathogens encountered. The second setting is to suppress the burden of chronic bacterial colonization. The third setting is to treat acute exacerbations.
Eradication
The goal is to eradicate Pseudomonas infections on first identification with a course of antibiotics that can interrupt the cycle of infection, inflammation, and airway damage. The European Respiratory Society guidelines recommend that adult patients with a new isolation of Pseudomonas be offered eradication treatment. Patients should not be offered eradication following new isolation of pathogens other than Pseudomonas.12
Suppression of Burden
The goal of suppressive antibiotic therapy is to reduce the bacterial burden for patients in whom eradication of the organism is not successful, to improve symptoms, and to reduce the frequency of exacerbations.
The European Respiratory Society guidelines suggest offering long term treatment for adults with bronchiectasis who have three or more exacerbations per year. They suggest 1) long-term treatment with an inhaled antibiotic for adults with bronchiectasis and chronic P aeruginosa infection; 2) long-term treatment with macrolides (azithromycin, erythromycin) for adults with bronchiectasis and chronic P aeruginosainfection in whom an inhaled antibiotic is contraindicated, not tolerated, or not feasible; 3) long-term treatment with macrolides (azithromycin, erythromycin) in addition to or in place of an inhaled antibiotic, for adults with bronchiectasis and chronic P aeruginosa infection who have a high exacerbation frequency despite taking an inhaled antibiotic; 4) long-term treatment with macrolides (azithromycin, erythromycin) for adults with bronchiectasis not infected with P aeruginosa (conditional recommendation, moderate quality evidence); 5) long-term treatment with an oral antibiotic (choice based on antibiotic susceptibility and patient tolerance) for adults with bronchiectasis not infected with P aeruginosa in whom macrolides are contraindicated, not tolerated, or ineffective; 6) long-term treatment with an inhaled antibiotic for adults with bronchiectasis not infected with P aeruginosa in whom oral antibiotic prophylaxis is contraindicated, not tolerated, or ineffective.12
Inhaled antibiotics have a theoretical advantage over oral therapies by delivering higher concentrations of drug to the airway. They may reduce systemic absorption and the associated side effects that are commonly associated with the intravenous form. However, inhaled antibiotics are associated with their own potential side effects. The most common side effects seen with inhaled antibiotics are bronchospasm, dyspnea, cough, increased sputum, ototoxicity, and nephrotoxicity. Common inhaled antibiotics used in suppressive bronchiectasis therapy are tobramycin, gentamicin, and colistin. Inhaled ciprofloxacin and aztreonam have been studied but failed to provide clinical benefit.
Treatment of Exacerbations
One goal of using antibiotics for acute exacerbations is to lower the rate of future exacerbations. Treatment of exacerbations in bronchiectasis patients should be guided by culture results from lower respiratory secretions. Mild-to-moderate exacerbations can usually be treated with an oral antibiotic, but if the patient is infected with a drug-resistant P aeruginosa or other similar pathogens, then intravenous antibiotic may be required. The European Respiratory Society guidelines recommend that acute exacerbations of bronchiectasis be treated with 14 days of antibiotics.12
Surgery
The indications for surgical resection in bronchiectasis include persistent symptoms despite up to a year of comprehensive medical treatment, exacerbations that are either severe or frequent and interfere with social/professional life, recurrent refractory or massive hemoptysis, post obstruction bronchiectasis distal to tumors, and localized severely damaged lobe/segment that may be a source of sepsis.5
Role of the Pharmacist
Pharmacists can add to the collective efforts of other healthcare providers and patient advocacy groups because of their general understanding of the disease and expertise in the proper use of medications, durable medical equipment, and therapeutic options.
Smoking Cessation
Patients with respiratory disease have a greater and more urgent need to stop smoking than the average smoker. They should be encouraged to stop, but many often find it more difficult to do so.14 Pharmacists are keenly positioned to employ Screening, Brief Intervention, and Referral to Treatment (SBIRT) to patients with lung disease. Screening quickly assesses the patient’s severity of substance use and identifies the appropriate level of treatment. Brief intervention focuses on increasing the patient’s awareness regarding substance use and motivation toward behavioral change. Referral to treatment provides those identified as needing more extensive treatment with access to specialty care. Community and specialty retail pharmacists are best poised to conduct these conversations with patients. Their goal would be to provide early intervention and alternative treatment options for consuming nicotine (e.g., nicotine patches, lozenges, and gums) and ultimately change behavior patterns leading to complete smoking cessation.
Nutrition
Poor nutritional status can influence immunity, lung function, and strength, while overweight individuals may develop other conditions such as diabetes and heart disease secondary to decreased mobility. Pharmacists can counsel bronchiectasis patients on proper nutritional considerations. Include probiotic foods in the diet to help rebalance the gut bacteria after antibiotic treatment. Starchy high-fiber grains and vegetables should be included at each meal. Good-quality fats can be included daily. Sources include olive oil, vegetable oil, butter, nuts, nut butters, and seeds.
Underweight individuals should increase the nutrient density of foods and consume foods that are higher in fat and protein. Fluid intake is essential. Lung secretions are thick and viscous, increasing the risk for infection and dehydration. Consider removing dairy from the diet as that can increase phlegm in the system. It is now becoming relatively well established that vitamin D supplementation could help to attenuate respiratory tract infections. Vitamin D has been found to enhance neutrophil killing in patients with bacterial respiratory tract infections and to lower pro-inflammatory cytokine production induced by infected neutrophils.15
Inhaler Usage
The Global Initiative for Chronic Obstructive Lung Disease strategy document highlights the importance of regularly checking treatment adherence and assessing inhaler technique.16 Mastering the proper technique for inhaler devices, which are an important component of lung disease treatment, can be challenging for patients. Often, patients will be prescribed multiple medication inhalers and therefore required to learn a variety of techniques. Patients need to be assessed and reassessed on how they use their medication inhalers and educated on the proper maintenance and cleaning of the delivery devices.
Adverse Event Counseling
Bronchiectasis patients are often treated with long- term macrolides and inhaled antibiotics. Upon dispensing of these antibiotics, the pharmacist is best suited to talk about the potential side effects and drug interactions associated with these drugs. Macrolides, as a class, can cause gastrointestinal disturbances and tinnitus. Pharmacists can offer OTC options to mitigate the disturbances. Pharmacists can also reinforce the concept of having an annual auditory exam. Inhaled antibiotics can cause bronchospasms, dyspnea cough, and increased sputum. Explaining this to the patient and offering some holistic remedies (i.e., lozenges, drinking warm beverages, use of pretreatment inhaled osmotics) at the time of dispensing the drug can help alleviate unexpected anxiety, stress, and discomfort to the patient should the event occur. These measures could also drive adherence and compliance to the medication regimen. Pharmacists can also help patients with adherence issues through behavioral interventions that use cues, reminders, and reinforcement such as linking medication-taking with certain daily activities.
Vaccinations
Today, pharmacists can administer vaccines in all 50 states and are able to give most available vaccines. In fact, most patients choose to get vaccinated at pharmacies due to convenience. Pharmacists are in a prime position to educate patients with accurate and scientifically based clinical data supporting the use of vaccines to prevent and treat diseases.
In the U.S., vaccination with the 13-valent PCV followed by the PPV-23 is now recommended for all high-risk adults and children aged 6 to 18 years with immunocompromising conditions and those aged 65 years or older.17 There are no specific data on the efficacy or effectiveness of the influenza vaccine in patients with bronchiectasis. Despite the lack of data in bronchiectasis, there is general agreement that the annual seasonal influenza vaccine should continue to be recommended for at-risk populations, including those with chronic lung diseases.18
Summary
Bronchiectasis is an ever-present disease in the U.S. and is increasing in prevalence with significant associated comorbidities. The medical management of bronchiectasis includes a careful confirmation of the diagnosis based on clinical and radiographic findings. Patients should have a systematic evaluation of the potential heterogenous causes of bronchiectasis. Treatment should be approached in a stepwise fashion based on severity of disease and microbiologic findings. It should include airway clearance therapy, reduction in inflammation, and treatment of acute and chronic infections. Education of the patient regarding the disease is vitally important. A multidisciplinary care team, including pulmonologists, infectious disease physicians, respiratory care practitioners, and pharmacists, is crucial to a successful management of the disease.
REFERENCES
1. Keir HR, Chalmers JD. Pathophysiology of bronchiectasis. Semin Respir Crit Care Med. 2021;42:499-512.
2. Gramegna A, et al. In: Chalmers J, Polverino E, Aliberti S, eds. Bronchiectasis: the EMBARC Manual. Cham, Switzerland: Springer International Publishing. 2018:65-76.
3. Weycker D, Hansen GL, Seifer FD. Prevalence and incidence of noncystic fibrosis bronchiectasis among US adults in 2013. Chron Respir Dis. 2017;14:377-384.
4. McShane PJ, Naureckas ET, Tino G, Strek ME. Non-cystic fibrosis bronchiectasis. Am J Respir Crit Care Med. 2013;188:647-656.
5. Hill AT, et al. British Thoracic Society guidelines for bronchiectasis in adults. Thorax. 2019;74(suppl1):1-69.
6. Cole PJ. Inflammation: a two-edged sword–the model of bronchiectasis. Eur J Respir Dis Suppl. 1986;147:6-15.
7. Flume PA, Chalmers JD, Olivier KN. Advances in bronchiectasis: endotyping, genetics, microbiome, and disease heterogeneity. Lancet. 2018;392:880-890.
8. Amati F, Simonetta E, Gramegna A, et al. The biology of pulmonary exacerbations in bronchiectasis. Eur Respir Rev. 2019;28:190055.
9. Hill AT, Haworth CS, Aliberti S, et al. Pulmonary exacerbations in adults with bronchiectasis: a consensus definition for clinical research. Eur Respir J. 2017;49:1700051.
10. Chalmers JD, Aliberti S, Filonenko A, et al. Characterization of the “frequent exacerbator phenotype” in bronchiectasis. Am J Respir Crit Care Med. 2018;197:1410-1420.
11. Al Moamary MS. Impact of a pulmonary rehabilitation programme on respiratory parameters and health care utilization in patients with chronic lung diseases other than COPD. East Mediterr Health J. 2012;18:120-126.
12. Polverino E, Goeminne PC, McDonnell MJ, et al. European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J. 2017;50:1700629.
13. Cramer CL, Patterson A, Alchakaki A, Soubani AO. Immunomodulatory indications of azithromycin in respiratory disease: a concise review for the clinician. Postgrad Med. 2017;129:493-499.
14. Tønnesen P, Carrozzi L, Fagerström KO, et al. Smoking cessation in patients with respiratory diseases: a high priority, integral component of therapy. Eur Respir J. 2007;29:390-417.
15. Derbyshire EJ, Calder PC. Bronchiectasis–could immunonutrition have a role to play in future management? Front Nutr. 2021;8:652410.
16. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. 2021 report. www.goldcopd.org/wp-content/uploads/2020/11/GOLD-REPORT-2021-v1.1-25Nov20_WMV.pdf. Accessed June 18, 2022.
17. CDC. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Morb Mortal Wkly Rep. 2012;61:816-819.
18. Chang CC, Morris PS, Chang AB. Influenza vaccine for children and adults with bronchiectasis. Cochrane Database Syst Rev. 2007;2007(3): CD006218.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.





