US Pharm.
2007;32(3):52-65.
In
the past, cardiovascular disorders were a greater concern in the adult
population than in the pediatric population. However, the rising prevalence of
childhood obesity and the narrowing knowledge gap concerning the relationship
between adult obesity and cardiovascular complications has heightened public
awareness that noncongenital cardiovascular diseases are possible in children.
It is becoming clearer that childhood obesity increases cardiovascular risk in
adulthood;1 adult cardiovascular diseases, such as hypertension and
peripheral vascular disease, originate in the fetal and childhood environment.
From gestation to adolescence, the cardiovascular system seems vulnerable to
injury from maternal factors and dietary habits.2 One study found
that a relationship between risk factors for cardiovascular disease and
obesity is present at as young as age 9 years.1
Most medications that are used
to treat pediatric patients are not FDA approved for use in children, although
literature offering appropriate dosage recommendations exists. Pharmacists
should become more familiar with common cardiovascular diseases that occur in
children and with their management, in order to optimize the care of this
unique patient population. More important, pharmacists can help ensure that
these children become healthier adults. This article reviews congenital heart
defects (CHDs), arrhythmias, and hypertension in children.
Congenital Heart Defects
Approximately 30%
of birth defects are heart related.3 The prevalence is four in
1,000 live births.4 Heart defects are one of the most common causes
of infant death from a birth defect.3,5 CHDs are structural
problems due to the abnormal formation of the heart or major blood vessels.
They are categorized into 15 distinct types. The most common are ventricular
septal defects (14% to 16%), tetralogy of Fallot (9% to 14%), transposition of
the great arteries (10% to 11%), atrioventricular septal defects (4% to 10%),
coarctation of the aorta (8% to 11%), patent ductus arteriosus (PDA; 5% to
10%), and hypoplastic left heart syndrome (4% to 8%).3
In 2000, there were more than
130,000 hospitalizations of infants or children with CHDs, incurring $6.5
billion in hospital charges. Mortality from CHD has declined by 25% from 1993
to 2003; the actual number of deaths has declined by 26%. Mortality rates vary
depending on the weight of the newborn and the type of defect.3
CHDs are more common in
low-birth-weight babies (weight <2,500 g).4 Thirty-six thousand
(nine per 1,000) defects are expected in the United States annually, and of
these, 9,200 (2.3 per 1,000) live births will require invasive treatment or
result in mortality during the first year of life.3 Although a
significant number of cases still require invasive surgery, many CHDs are
small and close spontaneously during the child's first year of life.
3 For example, as many as 5% of newborns (about 200,000 per year) are
born with tiny muscular ventricular septal defects that close spontaneously
and do not require immediate surgical intervention.3
Another example of spontaneous
closure is PDA, where the defect lies in the vascular structure connecting the
proximal descending aorta to the roof of the pulmonary artery.4,6
Small PDAs, called silent PDAs, may close spontaneously within the first year
of life or remain open, but the patient is asymptomatic. When left untreated,
larger PDAs result in complications, such as respiratory distress,
hypotension, and pulmonary hypertension. PDAs are closed with indomethacin,
6,7 ibuprofen,8,9 or surgical ligation.9 Moderate-
to large-sized PDAs can decrease lung compliance and increase the work of
breathing. Large PDAs can also result in secondary pulmonary hypertension.
Moreover, left heart failure due to hypertrophy or ischemic heart disease can
present in adulthood after many years of insult.6
If surgery is required to
close the PDA, the procedure is delayed, if possible, until the pediatric
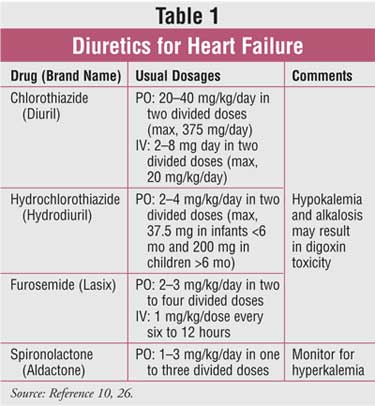
patient is stable. Treatment with a combination of diuretics (see TABLE 1
) and digoxin may also be necessary to maintain heart function. In addition,
angiotensin-converting enzyme (ACE) inhibitors may be initiated for afterload
reduction. Patients with atrial fibrillation may require antiarrhythmic
medications and anticoagulants. Upon closure of the PDA, endocarditis
prophylaxis precautions, such as prophylaxis with a short course of oral
antibiotics prior to dental procedures, should be observed until six months
after closure.6 However, patients with a PDA and pulmonary vascular
disease are not candidates for closure. Instead, management with pulmonary
vasodilators, such as chronic oxygen, prostaglandins, calcium channel
blockers, endothelin antagonists, and phosphodiesterase type V inhibitors, is
initiated. Partial closure is sometimes considered.6
In general, treatment with
medications such as digoxin, diuretics, ACE inhibitors, beta-blockers,
inotropes, or prostaglandin E1 (in the case of congestive heart
failure) has been successful in improving the prognosis for children with CHD.
In some cases, pulmonary hypertension has been successfully managed with
sildenafil, although it is used off label.10,11 Further studies are
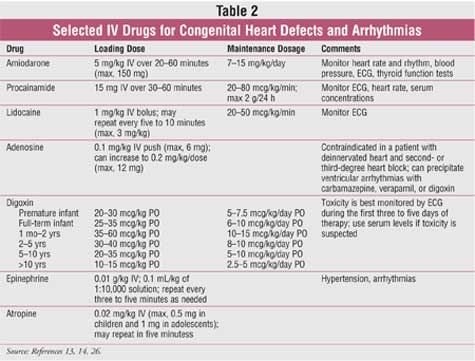
being conducted in both children and adults. Selected pharmacologic treatments
for children are summarized in TABLE 2.
While major defects (tetralogy
of Fallot, transposition of the great arteries, coarctation of the aorta, and
hypoplastic left heart syndrome) are usually detected during the neonatal
period because the infants are critically ill within the first few days of
life, minor defects may not be detected until adulthood.3,12 The
diagnosis is made via physical exam, echocardiograph, electrocardiograph, and
chest radiograph. Patients may present with a murmur, acute worsening of
underlying manifestations of premature birth (i.e., apnea of prematurity),
exercise intolerance, or atrial fibrillation, or they may have been previously
diagnosed with reactive airway disease. Surgery, including open-heart surgery
or catheterization, is sometimes performed at the bedside in the intensive
care unit, out of necessity. About one million adults have been told by a
physician that they have congenital heart disease.3 Although life
expectancy for these adults has improved, it is still lower than that of the
general population.5 Sudden death, progressive heart failure, and
perioperative death are common causes of mortality in adults.5
Pharmacists have an important
role in educating caregivers and patients about the rationale for taking
medications and their potential side effects. More important, some of the oral
liquid medications require extemporaneous compounding. Recipes can be found in
references such as the Pediatric Dosage Handbook and the literature.
Pharmacists should also demonstrate how to measure liquid medications
accurately with calibrated devices and offer tips to improve palatability.
Arrhythmias
One epidemiologic
study found that the overall incidence of arrhythmias in children was 55.1 per
100,000 emergency department visits. The diagnosis and treatment of
arrhythmias is essential for the prevention of cardiopulmonary compromise and
arrest. The most common arrhythmias include sinus tachycardia (50%),
supraventricular tachycardia (SVT; 13%), bradycardia (6%), and atrial
fibrillation (4.6%).13 It is important to note that children's
heart rates vary by age.1
Pharmacists can assist in
determining whether arrhythmias are drug induced by taking an accurate
medication history. For example, SVT can be caused by exposure to stimulants,
beta-agonists, anticholinergics, salicylates, theophylline, tricyclics, and
phenothiazines, while beta-blockers can contribute to bradycardia.14
Many pharmacists are also trained to participate in rescue efforts during
cardiac arrests.
Sinus Tachycardia:
Sinus tachycardia can be associated with hypoxia, anemia, hypovolemia, shock,
myocardial infarction, pulmonary edema, hyperthyroidism, medications,
hypocalcemia, and illicit drug use in older children. Normal heart rate can be
up to 160 beats per minute (bpm) in neonates, 180 bpm in a 1-month-old, 165
bpm in a 1-year-old, and 130 bpm in a 12-year-old, but a persistently elevated
heart rate is abnormal. In children, the most common causes are dehydration
and hypovolemia. Sinus tachycardia is usually benign, and treatment of the
underlying disorder is key.14
Supraventricular
Tachycardia: This
arrhythmia is characterized by a heart rate greater than 220 bpm in newborns
and infants and greater than 180 bpm in older children, with ECG changes
revealing a narrow complex tachycardia. Three types of SVTs exist:
atrioventricular (AV) nodal reentrant tachycardia phenomenon, AV nodal
tachycardia (also called junctional tachycardia), and ectopic atrial
tachycardia.13,14 The majority of infants with SVT present at age 4
months; half of the cases are idiopathic, whereas 24% are associated with
fever and drug exposure, 23% are caused by congenital heart disease, and 10%
to 20% are the result of Wolff-Parkinson-White syndrome. Among older children,
the cause of SVTs is most likely the last. SVTs are tolerable for 24 hours,
but within 48 hours, heart failure can develop in 50% of patients.3,13,14
Treatment involves maintaining
airway, breathing, and cardiovascular status; oxygen supplementation; and
cardioversion. Nonpharmacologic measures, such as vagal maneuvers (ice to the
face in an infant), can be attempted for patients with asymptomatic SVT or
with mild heart failure. If this fails, medications, such as adenosine,
procainamide, and amiodarone, can be initiated. Maintenance therapy consists
of a beta-blocker, procainamide, sotalol, amiodarone, or flecainide. Failure
to manage SVTs with medications requires radiofrequency catheter ablation to
prevent recurrence. SVTs without heart failure can be managed with digoxin,
with or without diuretics.13,14
Bradycardia:
Bradycardia is defined as a heart rate below the lower limit of normal for the
child's age (i.e., 120 bpm for a 1-month-old, 90 bpm for a 1-year-old, and 65
bpm for a 12-year-old) and may be attributed to hypoxemia, vagal stimulation,
acidosis, or an acute elevation of intracranial pressure. It is crucial to
correct the underlying cause of the bradycardia prior to increasing the heart
rate; cardiopulmonary resuscitation may be necessary if the bradycardia is
prolonged. Persistent bradycardia after resuscitation warrants therapy with
epinephrine or atropine.13,14
Atrial Fibrillation:
Atrial fibrillation describes disorganized, rapid atrial activity
("irregularly irregular") with atrial rates of 350 to 600 bpm and varying
ventricular rates.13,14 An underlying structural heart defect or
recent intra-arterial surgery is a risk factor for developing atrial
fibrillation. Decreased cardiac output secondary to the discord between the
atria and ventricles can result in congestive heart failure. Management
involves cardioversion if the patient is hemodynamically unstable. Once normal
sinus rhythm is achieved, amiodarone, procainamide, quinidine, or a
beta-blocker is prescribed to maintain the rhythm. In hemodynamically stable
patients, digoxin can be initiated for ventricular rate control. If after a
24-hour trial digoxin alone appears ineffective, propranolol, esmolol, or
procainamide may be added.13,14
Ventricular Tachycardia:
Although rare in children, this type of arrhythmia should be recognized and
treated promptly with amiodarone, procainamide, or lidocaine to avoid
decompensation to ventricular fibrillation. The goal of therapy is to maintain
the heart rate at less than 150 bpm in infants and less than 130 bpm in
children.14
Ventricular
Fibrillation: This
arrhythmia is an uncommon cause of cardiac arrest in children younger than 1
year but is considered life threatening; it is observed in 5% to 15% of
children with out-of-hospital cardiac arrests.3,14 It is usually
preceded by ventricular tachycardia and caused by postoperative states, severe
hypoxia, hyperkalemia, myocarditis, myocardial infarction, and drugs.
Defibrillation, followed by amiodarone or lidocaine if refractory to
defibrillation, is recommended.14
Hypertension
As childhood
obesity becomes more of a problem, the prevalence of hypertension continues to
rise in children.2,15 Obesity appears to be a strong risk factor
for hypertension, based on reports associating large body mass index (BMI) and
high blood pressure.16,17 It is unclear what role race or ethnicity
plays in childhood hypertension.18 Childhood hypertension can lead
to adult hypertension, which is a known risk factor for coronary artery
disease (CAD) in adults.19,20 The presence of hypertension in
children may contribute to the early development of CAD.21
End-organ damage in children includes left ventricular hypertrophy (most
common), hypertensive encephalopathy, seizures, cerebrovascular accidents, and
congestive heart failure.15
Childhood hypertension has a
heritability rate of about 50%.2,21 One retrospective analysis of
medical records in a pediatric clinic found that 49% (n = 192) of children
(ages 21 years and younger) with primary hypertension had parents with primary
hypertension, and 46% (n = 84) of children (ages 21 years and younger) with
secondary hypertension had parents with secondary hypertension.22
Flynn and Alderman found that, overall, adolescents with primary hypertension
had an 86% positive family history of hypertension.23 Body size
(height percentile), age, and gender are used to determine if a child's
systolic and diastolic blood pressures (SBP and DBP, respectively) are within
normal limits (defined as SBP and DBP less than the 90th percentile). For
example, the SBP and DBP at the 90th percentile for a 5-year-old boy whose
height is in the 75th percentile are 110 and 69 mmHg, respectively. Similarly,
the SBP and DBP at the 90th percentile for a 5-year-old girl whose height is
in the 75th percentile are 107 and 69 mmHg, respectively. These data are
available in most pediatric drug references and in the literature.15,20
As with adults, in children younger than 18, hypertension is classified as:
15,20
• Prehypertension: SBP or DBP
greater than or equal to 90th percentile but less than 95th percentile
• Hypertension: SBP or
DBP greater than 95th percentile
• Stage 1 hypertension:
SBP or DBP from 95th percentile to 5 mmHg above 99th percentile
• Stage 2 hypertension:
SBP or DBP 5 mmHg above 99th percentile.
Blood pressure monitoring at
each well-child visit begins at age 3 years and is done by auscultation of the
right arm with a standard sphygmomanometer and a cuff of the correct size.
15,20 Children younger than 3 years should have their blood pressure
measured if they were born prematurely or have congenital heart disease,
recurrent urinary tract infections, hematuria, or proteinuria; known renal
disease or urologic malformations; family history of congenital renal disease;
solid organ transplant; malignancy or bone marrow transplant; treatment with
medications known to elevate blood pressure; and other systemic illnesses
associated with hypertension.15,20
Essential (primary)
hypertension is rarer in children younger than 10 years than in adolescents,
but it can occur in younger children as a diagnosis of exclusion. Most pre
adolescent children with hypertension have secondary hypertension from renal
parenchymal disease.24,25 Other causes include renal vascular
disease, endocrine disease (e.g., pheochromocytoma and hyperthyroidism),
coarctation of the aorta, and drugs (e.g., sympathomimetics, oral
contraceptives, and nutritional supplements). Transient hypertension is
sometimes mistaken for secondary hypertension from caffeine consumption or
anxiety disorders.15
Risk factors for the
development of essential hypertension in adolescents include family history
and increasing BMI.2,15,20 Approximately 9.2 million children and
adolescents between ages 6 and 19 are considered overweight or obese; in this
age group, obesity is defined as a BMI at or above the 95th percentile.3
Other risk factors for metabolic syndrome, such as low plasma high-density
lipoprotein levels, elevated plasma triglyceride levels, abdominal obesity,
and insulin resistance/hyperinsulin emia are important to consider as well.
2,15,20 Among children and adolescents ages 4 to 19 years, the mean
total blood cholesterol level is 165 mg/dL (163 mg/dL for boys and 167 mg/dL
for girls). Ten percent of adolescents ages 12 to 19 have total cholesterol
levels above 200 mg/dL.3 More worrisome is that one in 10 (9.2%)
adolescents between ages 12 and 19 has metabolic syndrome (three or more of
the following abnormalities: serum triglyceride level ?110 mg/dL;
high-density lipoprotein cholesterol level ?40 mg/dL; elevated fasting
glucose level ?110 mg/dL; or blood pressure ?90th percentile for
age, gender, and height). The prevalence is higher for boys than for girls
(9.5% vs. 8.9%, respectively). One in three overweight or obese adolescents
has metabolic syndrome, and two in three have at least one metabolic
abnormality. In moderately and severely obese adolescents, 38.7% and 49.7%,
respectively, have metabolic syndrome. Over 13,000 children are diagnosed with
type 1 diabetes each year, and the prevalence of type 2 diabetes is increasing.
3
Role of the Pharmacist
Pharmacists can
suggest therapeutic lifestyle modifications involving weight control, regular
exercise, and a low-fat, low-sodium diet for children and adolescents with
prehypertension and stage 1 hypertension. Smoking cessation and alcohol
abstinence are also recommended, when necessary. Approaches should be family
centered to ensure optimal success.15,20 According to data from
1999 to 2000, only 20% of children between ages 2 and 6 had a "good" diet; 74%
had a diet needing improvement; and 6% had a "poor" diet. In children between
ages 7 and 12, 8% had a good diet, 79% had a diet needing improvement, and 13%
had a poor diet.3 An example of a good diet would include fiber,
fruits and vegetables, and foods with low fat, salt, and cholesterol content.
An example of a poor diet is one with a high consumption of fast foods.
Pharmacologic therapy, in
conjunction with therapeutic lifestyle modifications, is recommended when
children and adolescents have symptomatic hypertension, target-organ damage,
secondary hypertension, diabetes, persistent stage 1 hypertension unresponsive
to lifestyle changes, or stage 2 hypertension.20 The goal of
therapy is to reduce blood pressure to below the 95th percentile in the
absence of end-organ damage or comorbidities and to below the 90th percentile
for age, height, and gender in the presence of end-organ damage or
comorbidities.15,20 The impact of untreated hypertension or
long-term use of antihypertensive agents on growth and development is unknown;
thus, a step-up approach beginning with a single agent is considered prudent.
15,20 A low dosage that is ineffective can be slowly titrated upward
until side effects are experienced or the maximum dosage is reached but
without improvement. Second, or even third, agents with complementary
mechanisms of action are added in the latter case. ACE inhibitors,
angiotensin-receptor antagonists, beta-blockers, calcium channel blockers, and
diuretics are all in the armamentarium. Selection of the agent is by
prescriber's choice, as data pertaining to specific drugs and clinical end
points do not exist. Clinically, more than one agent is traditionally used to
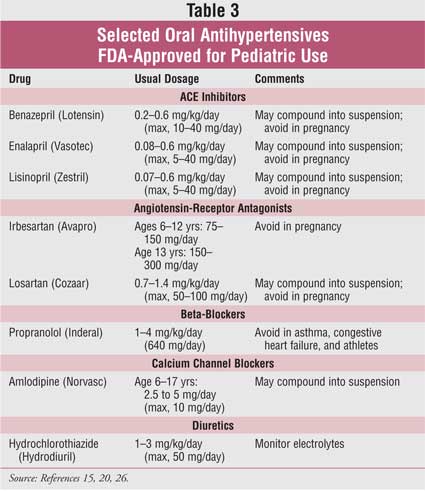
control blood pressure in children.15 Selected oral
antihypertensive agents used in children are listed in TABLE 3.
Pharmacists can assist in
optimizing therapy by ensuring that comorbidities are considered. For
instance, children or adolescents with diabetes or proteinuria should be
initiated on an ACE inhibitor; migraine sufferers should receive beta-blockers
or calcium channel blockers; and athletes should avoid diuretics.15,20
Additionally, pharmacists can help monitor blood pressure and prevent
target-organ damage by educating patients about symptoms and adherence to
therapy and by referring patients to a cardiologist and counseling about
nonpharmacologic therapies. The "step-down" approach toward discontinuation of
therapy is appropriate for children with uncomplicated essential hypertension
who are successful in weight control.15,20 It is important to note
that fixed-dose combination products are not routinely recommended due to the
lack of data to support their use in children.15
Severe, symptomatic
hypertension (blood pressure >99th percentile) requires prompt treatment.
15,20 Hypertensive emergencies are usually accompanied by signs of
hypertensive encephalopathy and seizures, while hypertensive urgencies are
accompanied by headache or vomiting. The treatment for hypertensive
emergencies is to reduce the blood pressure by 25% or less over the first
eight hours after presentation and then to normalize blood pressure over the
next 24 to 48 hours with intravenous antihypertensive therapy, such as
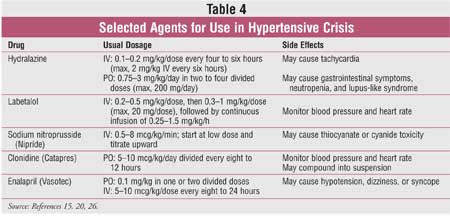
nicardipine, hydralazine, labetalol, or sodium nitroprusside. Several agents
used for hypertensive crisis are listed in TABLE 4.15,20
Summary
Cardiovascular
diseases are becoming increasingly prevalent in children, due at least in part
to the rise in childhood obesity. Public awareness is also increasing, and
treatment options are improving. Pharmacists have the responsibility of
counseling children and their caregivers about pharmacologic and
nonpharmacologic therapies and interventions that can improve long-term
prognosis. Adherence to complex medication regimens is key to optimal
management. In cases of CHDs, for example, pharmacists can provide education
about the rationale for drug therapy, and in cases of arrhythmia, they can
obtain a thorough medication history to assist in diagnosis or avoidance. More
important, pharmacists can stress the possibility of preventing the onset of
cardiovascular diseases and encourage lifestyle changes that can help reduce
the occurrence of hypertension and obesity.
References
1. Thompson DR,
Obarzanek E, Franco DL, et al. Childhood overweight and cardiovascular disease
risk factors: the National Heart, Lung, and Blood Institute Growth and Health
Study. J Pediatr. 2007;150:18-25.
2. Cohen MS. Fetal and childhood onset of adult cardiovascular diseases. Pediatr Clin N Am. 2004;51:1697-1719.
3. American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics – 2007 update. Circulation.2006;doi:10.1161/CIRCULATIONAHA.106.179918.
4. Ades A, Johnson BA, Berger S. Management of low birth weight infants with congenital heart disease. Clin Perinatol. 2005;32:999-1015.
5. Pelech AN, Neish SR. Sudden death in congenital heart disease. Pediatr Clin N Am.2004;51:1257-1271.
6. Takami T, Yoda M, Kawakami T, et al. Usefulness of indomethacin for patent ductus arteriosus in full-term infants. Pediatr Cardiol. 2007. [abstract;e-pub ahead of print].
7. Aranda JV, Thomas R. Systemic review: intravenous ibuprofen in preterm newborns. Semin Perinatol. 2006;30:114-120.
8. Thomas RL, Parker GC, Overmeire B, Aranda JV. A meta-analysis of ibuprofen versus indomethacin for closure of patent ductus arteriosus. Eur J Pediatr. 2005;164:135-140.
9. Schneider DJ, Moore JW. Patent ductus arteriosus. Circulation. 2006;114:1873-1882.
10. Schulze-Neick I, Hartenstein P, Li J, et al. Intravenous sildenafil is a potent pulmonary vasodilator in children with congenital heart disease. Circulation. 2003;108:II-167-173.
11. Buck M. Sildenafil for the treatment of pulmonary hypertension. Pediatric Pharmacotherapy.2004;10.
12. Park MK. Pediatric Cardiology for Practitioners. 4th Ed. Mosby; 2002.
13. Saccheti A, Moyer V, Baricella R, et al. Primary cardiac arrhythmias in children. Pediatr Emerg Care. 1999;15:95-98.
14. Doniger SJ, Sharieff GQ. Pediatric dysrhythmias. Pediatr Clin N Am. 2006;53:85-105.
15. Luma GB, Spiotta RT. Hypertension in children and adolescents. Am Fam Physician.2006;73:1158-1168.
16. Stabouli S, Kotsis V, Papamichael C, et al. Adolescent obesity is associated with high ambulatory blood pressure and increased carotid intimal-medial thickness. J Pediatr. 2005;147:651-656.
17. Muntner P, He J, Cutler JA, et al. Trends in blood pressure among children and adolescents. JAMA.2004;291:2107-2113.
18. Dekkers JC, Snieder H, Van Den Oord EJ, Treiber FA. Moderators of blood pressure development from childhood to adulthood, a 10-year longitudinal study. J Pediatr.2002;141:770-779.
19. Lauer RM, Clarke WR. Childhood risk factors for high adult blood pressure: the Muscarine Study. Pediatrics. 1989;84:633-641 [abstract].
20. U.S. Department of Health and Human Services. National Institute of Health. National Heart, Lung and Blood Institute. The Fourth Report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. NIH Publication No. 05-5267. Originally printed September 1996. Revised May 2005.
21. Jung FF, Ingelfinger JR. Hypertension in children and adolescence. Pediatr Rev.1993;14:169-179.
22. Robinson RF, Batisky DL, Hayes JR, et al. Significance of heritability in primary and secondary pediatric hypertension. Am J Hypertens. 2005;18:917-921.
23. Flynn JT, Alderman MH. Characteristics of children with primary hypertension seen at a referral center. Pediatr Nephrol. 2005;20:961-966.
24. Bartosh SM, Aronson AJ. Childhood hypertension. An update on etiology, diagnosis, and treatment. Pediatr Clin North Am. 1999;46:235-252.25. Flynn JT. Hypertension in adolescents. Adolesc Med Clin. 2005;16:11-29.
26. Taketomo CK, Hodding JH, Kraus DM.
Pediatric Dosage Handbook. 11th ed. Ohio: Lexi-Comp; 2005-2006.
To comment on this article, contact
editor@uspharmacist.com.





