US Pharm. 2010;35(4):HS-9-HS-11.
Worldwide, the leading cause of blindness is cataract. Globally, in 2002, there were more than 161 million people with visual impairment, of whom about 37 million were blind.1 Vision 2020 estimates that 18 million people are bilaterally blind from cataract.2 In North America, the amount of blindness due to cataract, as compared with all eye diseases, is about 5%. Poorer regions account for about 50% of cases of blindness as a result of cataract.1 In the United States, the Framingham Eye Study found a decrease in vision as a result of cataract in 15.5% of the population overall and in 45.9% of people over 75 years of age.3 Vision loss was defined as vision of 20/30 or worse. The Beaver Dam Eye Study, using a similar definition of vision loss due to cataract, found a frequency of 38.8% in men and 45.9% in women aged 75 years and older.4
Background
A cataract is an opacification of the lens of the eye. There are three specific types of cataract: nuclear, cortical, and posterior subcapsular. Cataract formation is mainly a result of aging; it is generally seen in both lenses and is caused by changes in the lens protein. Adult cataract is typically seen in patients 50 years and older.1 Patients aged over 50 years have a lower prevalence of cataract development than patients aged 60 years and older.5-7
A cataract may be symptomatic or asymptomatic. Symptoms include glare, decreased visual function or blurred vision, and blindness. The incidence of blindness from cataract is expected to increase owing to the increase in life expectancy.8 Age is considered a nonmodifiable risk factor. Other risk factors include genetics, suggesting a hereditary nature to cataract; oral and inhaled corticosteroid use; the use of oral and topical beta-blockers; dose- and drug-dependent use of phenothiazines such as chlorpromazine and thioridazine; and busulfan alkylating agents.9,10 Some data suggest that myopia may cause cataract. Modifiable risk factors may include elevated ocular exposure to ultraviolet B light, diabetes, and smoking.11,12
Vitamin supplementation has not been shown definitively to prevent or cause cataract. Likewise, a link between estrogen and cataract formation has not been substantiated. Interestingly, specific types of cataract have been associated with specific risk factors.9-11 Based on longitudinal studies, racial and ethnic groups also have shown specificity for certain cataract types.5-7
Economic Impact of Cataract
Delaying cataract by 10 years can have a significant impact on quality of life while decreasing the economic burden.13 Changing modifiable risk factors may delay cataract, but surgery is required to restore vision. In 1991, Medicare spent $3.4 billion on cataract surgery, at an average cost of $2,500 per procedure. This equates to about 1.5 million people who underwent cataract surgery in 1991.14 It would be expected that, owing to an aging population, the number of people undergoing cataract surgery and the cost in the U.S. would be much higher in 2009. Regardless, the cost of cataract surgery is relatively low when the cost of not treating cataract is taken into consideration. Vision loss related to cataract may affect employment opportunities and the ability to function independently.15 Patients who wait more than 6 months for cataract surgery could experience more negative outcomes during the waiting period than those whose waiting period was less than 6 months. Negative outcomes during the waiting period could include vision loss, an increased rate of falls, and reduced quality of life.16
When Surgery Should Be Performed
Nonsurgical options prior to surgery have a limited role, but include changing the patient’s refractive correction.17 Other nonsurgical options include magnifiers for reading, the use of tinted lenses, and papillary dilation for small central cataracts.
Typically, patients with cataract experience a decrease in vision over months to years. Assessing whether a decrease in vision has affected daily activities is vital for determining when surgery should be performed. The benefits of surgery may be much greater for a person who drives, works, or is active in sports versus a patient with dementia who lives in a nursing home. Generally, surgery is performed only when a significant decline in visual function is seen.15 If a patient presents with a sudden decrease or change in vision, he or she should immediately be referred to an ophthalmologist. Cataract surgery may be performed prior to a decline in visual function in a diabetic patient if the lens is too opaque for the ophthalmologist to clearly evaluate the retina.
During surgery, the clouded lens is removed and replaced with a synthetic monofocal or multifocal lens. The monofocal lens has a single focus length; the multifocal lens has multiple focus lengths, which makes it easier for the patient to see at a distance and close up without the aid of glasses.15 Although the multifocal lens may provide freedom from wearing glasses, more patients with multifocal lenses have reported halos around lights and reduced contrast sensitivity than patients who have monofocal lenses.18 Contrast sensitivity is the ability to distinguish an object from its background.
Types of Surgery and Intraoperative Medications
In the U.S., there are two main types of extraction surgeries for cataract: extracapsular cataract extraction (ECCE) and phacoemulsification (PHACO); PHACO is more commonly performed.15 PHACO’s advantages over ECCE include a smaller incision, lower intraoperative rates of vitreous loss and iris prolapse, shorter surgery time, and faster time to visual recovery. The ECCE procedure may be chosen for a dense cataract that is more advanced. A rapid recovery and significant improvement in visual acuity occur in about 90% of patients.11
Sedation with topical anesthetic eyedrops is the most frequently used anesthesia for cataract surgery. Lidocaine is the topical anesthetic of choice.19 Rarely, general anesthesia may be considered if a patient with advanced age cannot cooperate, has poor mental status, or is severely claustrophobic. Topical anesthesia with eyedrops results in fewer complications than retrobulbar (space behind the eyeball) and peribulbar (space outside the muscle cone) injections.15 Less sedation is required in patients receiving topical anesthesia; as a result, there is less nausea and vomiting postoperatively. In addition, topical anesthesia does not cause temporary paralysis of the eye or affect vision; therefore, many patients have improved vision immediately after surgery.
There is a small risk of intraocular infection or endophthalmitis (occurring at a rate of 0.08% to 0.1%). Prophylactic pre- or postoperative antibiotics administered to the eye are generally not given unless an intraoperative complication occurs or the surgeon has a greater number of endophthalmitis cases than is typical.17 If a patient develops pain and decreased vision 2 to 5 days after cataract surgery, immediate evaluation by an ophthalmologist should be performed. An intraocular infection can cause severe permanent vision loss.
Finally, approximately 25% of ECCE surgeries result in posterior capsular opacification, generally occurring within 5 years of surgery. This is due to a proliferation of epithelial cells in the lens. The risk is higher in younger patients and in patients with certain types of intraocular lenses.15
Preoperative and Postoperative Medication
Generally, it is the standard of care to use topical nonsteroidal anti-inflammatory drugs (NSAIDs) pre- and postoperatively; however, there is no consensus for the therapeutic use of topical NSAIDs.17 According to an article by O’Brien, surgeons dose NSAIDs 1 to 2 days prior to surgery and for only 1 week to 10 days after surgery.20 Since a major postoperative complication of cataract surgery is cystoid macular edema (CME), typically occurring 4 to 6 weeks postoperatively, O’Brien recommends a more robust pre- and postoperative topical NSAID regimen that also considers patient risk factors (FIGURE 1).20 Following cataract surgery, topical NSAIDs are used for the management of postoperative inflammation, to prevent and treat CME, and for pain.20 Topical NSAIDs are also used intraoperatively to prevent miosis.
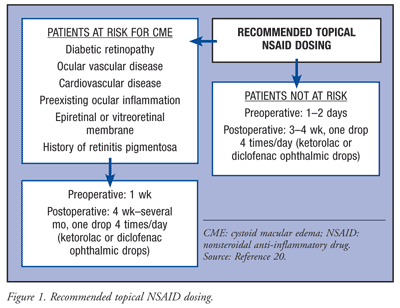
Topical corticosteroids have been used concomitantly with topical NSAIDs to treat CME in some patients. Data suggest that combination therapy is superior for treating CME versus either agent alone.20 Although the Canadian Ophthalmological Society recommends topical steroids, topical NSAIDs, or both in the perioperative period and for the treatment of CME, consensus on therapeutic use has not been established. Studies have shown that better visual outcomes and decreased angiographic CME were seen when topical NSAIDs were started 2 to 3 days before surgery and continued 3 to 4 times per day for 3 to 4 weeks.17 These recommendations, not based on consensus, differ somewhat from the recommendations made by O’Brien (FIGURE 1).20 The most common side effects of topical NSAIDs are stinging and irritation. Rarely, superficial punctate keratitis and epithelial defects have been seen. Delayed corneal wound healing and re-epithelialization may result.20
Aspirin or other anticoagulants may be taken without regard to cataract surgery; however, the international normalized ratio should be in the therapeutic range.15 Intraoperative floppy iris syndrome has been reported during cataract surgery in some patients who were receiving or had received tamsulosin. In most cases, tamsulosin had been stopped 2 to 14 days before surgery; however, a few cases were reported after the patient had been off the medication for 5 weeks to 9 months.21 Currently, there is no recommendation concerning discontinuing the use of tamsulosin prior to cataract surgery. In order for the ophthalmologist to be prepared for a modification in surgical technique, the patient must tell the surgeon whether he or she is currently taking, or previously took, tamsulosin.
Conclusion
Cataract is a prevalent condition in developed and developing countries. Adult cataract is typically seen in people aged 50 years and older, and prevalence increases with advancing age.1,5-7 In the U.S., approximately 40% to 46% of people over the age of 75 years will have vision loss of 20/30 or worse as a result of cataract.4 Delaying cataract by 10 years can have a significant impact on quality of life while decreasing the economic burden.13 Negative outcomes of unresolved cataract include vision loss, an increased rate of falls, fewer work opportunities, and the inability to live independently.15,16 Surgery, which is necessary to restore vision, results in rapid recovery and a significant improvement in visual acuity, generally seen in about 90% of patients.11 Identifying patients with risk factors may lead to referral to an ophthalmologist and ultimately to receiving the cataract surgery covered by Medicare.
There is no consensus on the use of topical NSAIDs and topical corticosteroids pre- and postoperatively.17 Generally, NSAIDs are used pre-, intra-, and postoperatively. The Canadian Ophthalmological Society recommends that topical NSAIDs be started 2 to 3 days before surgery and continued 3 to 4 times per day for 3 to 4 weeks.17 However, O’Brien recommends a longer treatment duration of 4 weeks to several months for patients at risk for CME.20
REFERENCES
1. Resnikoff S, Pascolini D, Etya’ale D, et al. Global data on visual impairment in the year 2002. Bull World Health Organ. 2004;82:844-851.
2. Vision 2020. What is avoidable blindness? Cataract. www.vision2020.org/main.cfm?type=WIBCATARACT.
3. Kahn HA, Leibowitz HM, Ganley JP. The Framingham Eye Study. I. Outline and major prevalence findings. Am J Epidemiol. 1977;106:17-32.
4. Klein BE, Klein R, Linton KL. Prevalence of age-related lens opacities in a population. The Beaver Dam Eye Study. Ophthalmology. 1992;99:546-552.
5. Varma R, Torres M. Prevalence of lens opacities in Latinos: The Los Angeles Latino Eye Study. Ophthalmology. 2004;111:1449-1456.
6. Xu L, Cui T, Zhang S, et al. Prevalence and risk factors of lens opacities in urban and rural Chinese in Beijing. Ophthalmology. 2006;113:747-755.
7. Murthy GVS, Gupta SK, Maraini G, et al. Prevalence of lens opacities in north India: the INDEYE feasibility study. Invest Ophthalmol Vis Sci. 2007; 48:88-95.
8. Riaz Y, Mehta JS, Wormald R, et al. Surgical interventions for age-related cataract. Cochrane Database Syst Rev. 2006;(4):CD001323.
9. Kanthan GL, Wang JJ, Rochtchina E, Mitchell P. Use of antihypertensive medications and topical beta-blockers and the long-term incidence of cataract and cataract surgery. Br J Ophthalmol. 2009;93:1210-1214.
10. Li J, Tripathi RC, Tripathi BJ. Drug-induced ocular disorders. Drug Saf. 2008;31:127-141.
11. Abraham AG, Condon NG, West Gower E. The new epidemiology of cataract. Ophthalmol Clin North Am. 2006;19:415-425.
12. Centers for Disease Control and Prevention. 2004 Surgeon General’s Report—The Health Consequences of Smoking. Chapter 6. Other effects. www.cdc.gov/tobacco/data_statistics/sgr/2004/pdfs/chapter6.pdf. Accessed November 15, 2009.
13. McCarty CA, Taylor HR. The genetics of cataract. Invest Ophthalmol Vis Sci. 2001;42:1677-1678.
14. Steinberg EP, Javitt JC, Sharkey PD, et al. The content and cost of cataract surgery. Arch Ophthalmol. 1993;111:1041-1049.
15. Bollinger KE, Langston RHS. What can patients expect from cataract surgery? Cleve Clin J Med.
16. Hodge W, Horsley T, Albiani D, et al. The consequences of waiting for cataract surgery: a systematic review. CMAJ. 2007;176:1285-1290.
17. Canadian Ophthalmological Society Cataract Surgery Clinical Practice Guideline Expert Committee. Canadian Ophthalmological Society evidence-based clinical practice guidelines for cataract surgery in the adult eye. Can J Ophthalmol. 2008;43:S7-S57.
18. Leyland M, Pringle E. Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane Database Syst Rev. 2006;(4):CD003169.
19. Navaleza JS, Pendse SJ, Blecher MH. Choosing anesthesia for cataract surgery. Ophthalmol Clin N Am. 2006;19:233-237.
20. O’Brien TP. Emerging guidelines for use of NSAID therapy to optimize cataract surgery patient care. Curr Med Res Opin. 2005;21:1131-1137.
21. Flomax (tamsulosin hydrochloride) package insert. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; December 2009.






