US Pharm. 2016;41(3)21-23.
Pain experienced after a stroke is one of the most challenging consequences following this life-changing event. A syndrome known as central post-stroke pain (CPSP) is a central neuropathic pain condition characterized by pain and sensory abnormalities that manifest in the body parts that correspond to the area of the brain that has been injured by the cerebrovascular lesion.1,2 CPSP is a disabling condition with a low prevalence (1%- 8%) in stroke patients.1,3 However, some believe this figure may be misleading in light of the enormous burden of stroke in the population; more than half of the estimated 55 million worldwide survivors of stroke are expected to have some form of stroke-related disability.3-6 Stroke burden will likely escalate rapidly in the near future largely owing to an increasing life expectancy; this is because of a strong positive relationship between age and stroke incidence.6,7
The reason the exact prevalence of CPSP is not known partly owes to the difficulty in differentiating this condition from other pain types that can occur following a stroke, including shoulder pain, painful spasticity, persistent headache, and other musculoskeletal pain conditions.2 At present there is no clear relationship between patient age and the risk of CPSP development.4 One study did find that early evoked pain, or dysesthesia—an unpleasant abnormal sensation produced by normal stimuli—is a predictor for CPSP.1 It is important for pharmacists to note that CPSP often goes unrecognized, is underappreciated, and can be difficult to manage. Management may be particularly challenging in the elderly, in whom certain drugs may not be well tolerated or are potentially inappropriate.
While the exact pathogenesis of CPSP is currently unknown, it is suggested that underlying causes include hyperexcitation in the damaged sensory pathways, damage to the central inhibitory pathways, or a combination of the two.3 After a stroke, local pain is usually experienced in the joints, secondary to mechanical use; shoulder pain is especially common among stroke survivors.8 The central pain that is identified as CPSP may be described as constant, intermittent, moderate, or severe, and is caused by the brain lesion itself; as mentioned above, it corresponds to the brain territory that has been injured by the cerebrovascular lesion. Normal pain messages from the body, such as touch, warmth, cold, and other stimuli, are misinterpreted; even slight sensations may register as painful on the skin.8 CPSP is associated with sensory abnormalities, particularly of thermal sensation.4 A significant proportion of stroke patients experience a latency period before the onset of CPSP symptoms.4
PHARMACOLOGIC TREATMENT
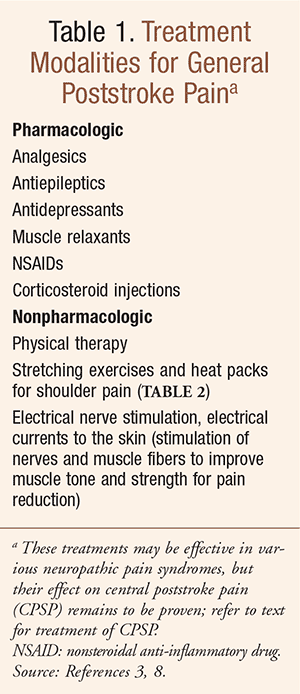
Pharmacologic and nonpharmacologic treatment of poststroke pain in general is outlined in TABLE 1 and may be effective in various neuropathic pain syndromes; however, owing to the dearth of published data from large, well-designed clinical trials involving patients with CPSP, the effect of these modalities on CPSP remains to be proven. More focused treatment of CPSP is discussed below.3,4 Tips for patients with poststroke pain may be found in TABLE 2, and information about a patient fact sheet is available in the Patient Resource sidebar.

Amitriptyline and lamotrigine are effective treatments for CPSP.9 Efficacy reported with antidepressants in reducing CPSP in some patients seems to implicate multiple chemical mechanisms. The effectiveness of antiepileptics and antiarrhythmics implicates calcium and sodium channels; the effectiveness of opioids implicates opioid receptors.4 Limited available data seem to suggest multiple mechanisms, owing to the apparent lack of universal effectiveness of any drug class and the ineffectiveness of all therapeutic approaches in the vast majority of patients; neuronal damage secondary to a stroke suggests involvement of local neurotoxicity, which is often attributed to glutamate, an excitatory amino acid.4 While treatment of CPSP is difficult, the most effective approaches are those that target the increased neuronal hyperexcitability.2
Antidepressants
Antidepressants such as amitriptyline and nortriptyline can be used as first-line treatment.10
Amitriptyline, an adrenergic antidepressant, is currently the first-line drug for CPSP. However, its effect is frequently incomplete, and a high dose is commonly not tolerated in stroke patients.3 Further, amitriptyline is considered potentially inappropriate in older adults because of its highly anticholinergic and sedating effects and pronounced effects on the cardiovascular system, including hypotension.10
Nortriptyline is the least likely of the tricyclic antidepressants (TCAs) to cause orthostatic hypotension and one of the least anticholinergic and sedating TCAs; it is a preferred agent when a TCA is indicated.11
Antiepileptics
Antiepileptics, including lamotri-gine, gabapentin, pregabalin, and carbamazepine, can be used as first-line treatment.10
Lamotrigine, a triazine derivative antiepileptic, inhibits the release of glutamate and inhibits voltage-sensitive sodium channels, which stabilizes neuronal membranes.11,12 This agent was found to be effective in a controlled trial and can be used as an alternative or additive therapy.3 There are no pharmacokinetic differences noted between young adults and the elderly with regard to this agent. Lamotrigine should be used with caution, however, in older adults with significant renal decline.11 Of note, antiepileptics are associated with an increased risk of suicidal behavior or thoughts with use, regardless of indication; patients should be monitored for signs and symptoms of depression, suicidal tendencies, and other behavioral changes during therapy.11
GABA-ergic drugs with potential calcium channel-blocking effects, such as gabapentin or pregabalin, have recently emerged as a potentially useful therapy.3 While these drugs are effective in various neuropathic pain syndromes, their effect on CPSP remains to be proven.3
Pregabalin may improve pain-related anxiety and sleep disturbances.3 Peripheral edema, somnolence, ataxia, headache, and fatigue occur frequently with this agent; a decrease in the platelet count or prolongation of the PR interval may occur.11 Of note, pregabalin has been associated with discontinuation symptoms following abrupt cessation; dosage tapering over at least 1 week is recommended.11 Further, pregabalin should be used with caution in patients with renal impairment, and dosage adjustment is required.11 In terms of combination therapy, gabapentin or pregabalin with amitriptyline is not effective.10
Carbamazepine use in the elderly may increase the risk of symptoms mimicking syndrome of inappropriate antidiuretic hormone (SIADH).11 This patient population is also more susceptible to carbamazepine-induced confusion, agitation, atrioventricular block, and bradycardia.11
Other Agents
For patients refractory to the above-noted treatments, opioids such as morphine or levorphanol may be prescribed, although no large studies have directly examined their efficacy for CPSP.10 Fluvoxamine and mexiletine may be used adjunctively in some patients.3 Fluvoxamine is considered to be potentially inappropriate in the elderly; in light of the number of its drug interactions and the limited information available on its use in the elderly, it may be best to select an alternative agent.11,13 Further, older adults are more prone to hyponatremia induced by selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs).10
Agents to Avoid
NSAIDs such as ibuprofen and cyclooxygenase-2 (COX-2) inhibitors are not recommended for the treatment of CPSP.10 Local anesthetics such as lidocaine, ketamine, cannabinoids, and botulinum toxin A are also not recommended.10
NONPHARMACOLOGIC MODALITIES
Nonpharmacologic treatment modalities, such as motor cortex stimulation or deep brain stimulation, are used in some cases; however, they are not proven to be effective.3 Future prospective studies with clear diagnostic criteria are essential for the proper collection and processing of epidemiological data.2 Well-designed clinical trials are needed to further our current understanding of the pathophysiology of CPSP; the development of better treatments is also necessary and may require basic research to advance this cause.3
CONCLUSION
The persistent, painful sensations following stroke are a major problem for affected patients. The diagnostic challenge of CPSP is its similarity to other pain conditions such as frozen shoulder syndrome, complex regional pain syndrome, and musculoskeletal problems. CPSP is often treatment-resistant, probably due to the involvement of multiple mechanisms contributing to the disease.
Pharmacists may be sought to recommend an individualized pharmacotherapy plan that incorporates pharmacologic and nonpharmacologic measures based on the currently limited available data. Diligent medication management may provide the opportunity to adjust therapy, based on patient response and future studies, thus targeting symptoms in a tailored and responsive approach to this common, underappreciated, and troublesome consequence of stroke.
REFERENCES
1. Klit H, Hansen AP, Marcussen NS, et al. Early evoked pain or dysesthesia is a predictor of central poststroke pain. Pain. 2014;155(12):2699-2706.
2. Klit H, Finnerup NB, Jensen TS. Central post-stroke pain: clinical characteristics, pathophysiology, and management. Lancet Neurol. 2009; 8(9):857-868.
3. Kim JS. Pharmacological management of central post-stroke pain: a practical guide. CNS Drugs. 2014; 28(9):787-797.
4. Henry JL, Lalloo C, Yashpal K. Central poststroke pain: an abstruse outcome. Pain Res Manag. 2008;13(1):41-49.
5. Bonita R, Solomon N, Broad JB. Prevalence of stroke and stroke-related disability. Estimates from the Auckland stroke studies. Stroke. 1997;28:1898-1902.
6. Thrift AG, Shrikanth VK. Stroke epidemiology and pathology. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:478-483.
7. Hankey GJ. Vascular disease of the heart, brain and limbs: new insights into a looming epidemic. Lancet. 2005;(366):1753-1754.
8. National Stroke Association. We can help. Pain. www.stroke.org/we-can-help/survivors/stroke-recovery/post-stroke-conditions/physical/pain. Accessed January 25, 2016.
9. Kumar B, Kalita J, Kumar G, et al. Central poststroke pain: a review of pathophysiology and treatment. Anesth Analg. 2009;108(5):1645-1657.
10. Gordon A. Best practice guidelines for treatment of central pain after stroke. In: Henry JL, Panju A, Yashpal K, eds. Central Neuropathic Pain: Focus on Poststroke Pain. Seattle, WA: IASP Press; 2007.
11. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 20th ed. Hudson, OH: Lexicomp; 2015:215-219,816-820,1047-1049,1205-1207.
12. Epocrates.com. Epocrates Plus Version 15.12.1. Updated January 13, 2016.
13. Reuben DB, Herr KA, Pacala JT, et al. Geriatrics at Your Fingertips: 2015. 17th ed. New York, NY: American Geriatrics Society; 2015:70,82.
To comment on this article, contact rdavidson@uspharmacist.com.






