US Pharm. 2015;40(7):21-24.
Sleep-disordered breathing (SDB) refers to a spectrum of periodic respiratory events that occur during sleep. These events range from mild snoring to partial cessation of airflow (hypopnea) to complete cessation of airflow (apnea).1 Obstructive sleep apnea (OSA), constituting 90% of sleep apnea cases, is airflow cessation as a result of airway closure in spite of adequate respiratory muscle effort.2 Central sleep apnea (CSA) encompasses a heterogeneous group of sleep-related breathing disorders in which respiratory effort is diminished or absent during sleep and occurs in an intermittent or cyclical manner.3,4
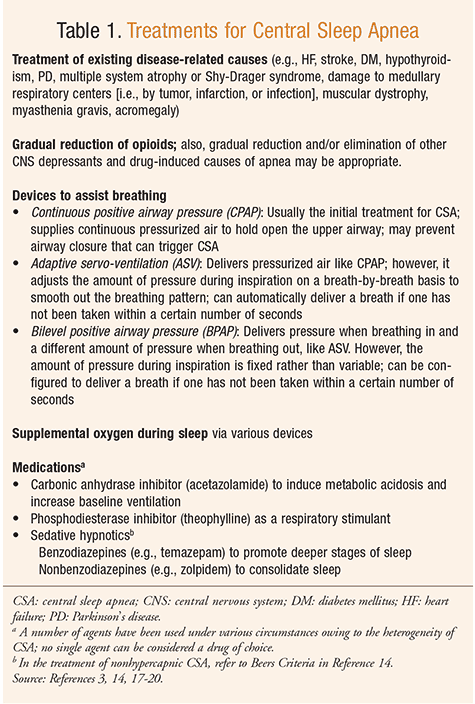
While CSA is most often associated with obstructive sleep apnea syndromes, it is also associated with or caused by an underlying medical condition (TABLE 1), a recent ascent to high altitude, drugs, or substance abuse.3 Primary CSA, which is considered a rare condition, is not entirely understood with regard to etiology.3 Individuals with cardiac, respiratory, or neurologic disease may be at greater risk of CSA.5
The use of drugs that cause central nervous system (CNS) depression, respiratory muscle dysfunction, or myopathy (TABLE 2) can place vulnerable individuals, such as the elderly and the chronically debilitated, at risk for drug-induced apnea (DIA).6 Medications such as long-acting benzodiazepines and narcotics should be avoided, if possible, since they have been shown to increase the frequency and duration of apneas.1 While the prevalence of CSA is less than 1% in the general population, Wang et al reported the prevalence rate of CSA at 30% in a population of patients in a stable methadone maintenance program.7,8

CSA syndromes are caused by two types of pathophysiological phenomena: 1) ventilator instability or 2) depression of the brainstem respiratory centers or chemoreceptors.3
CSA-hypoventilation syndromes, as seen with narcotic use, are secondary to disturbances of the central respiratory pattern center or peripheral chemoreceptors, or both, that may become more evident during sleep because of the suppression of wakefulness or behavior drive.3 Most often, CSA is seen during non-REM sleep followed by REM sleep; it is least likely to manifest in the fully awake individual. SDB is associated with nocturnal cardiac arrhythmias.9 Different types of SDB have been shown to be associated with different types of arrhythmias; CSA has been strongly associated with atrial fibrillation and atrial flutter.9 Patients with heart failure and Cheyne-Stokes respiration CSA (CSR-CSA) have a higher mortality rate than those without it.3
Symptoms
Symptoms of CSA most commonly reported are insomnia, excessive daytime sleepiness, and fatigue; additional frequent presentations also include nighttime awakenings and nocturnal hypoxia.10 Other symptoms reported may pertain to an underlying disease-related cause such as heart failure, stroke, renal failure, Parkinson’s disease, or multiple system atrophy. In the case of CSR-CSA, dyspnea, orthopnea, paroxysmal nocturnal dyspnea, and other heart failure symptoms can be seen.3
Measuring Apnea and Daytime Sleepiness
A central apneic event, as seen on polysomnography (PSG), is conventionally defined as cessation of airflow for 10 seconds or longer without an identifiable respiratory effort; this is in contrast to an OSA event that has a discernible ventilator effort during the airflow cessation period.3 The severity of SDB is measured by the apnea hypopnea index (AHI), which is the number of episodes of apneas and hypopneas per hour of sleep.1,2 To make a diagnosis of CSA due to drugs or substance abuse, an AHI of >5 in the absence of periodic breathing and an AHI of >10 in the presence of periodic breathing is required.3 The AHI is used for Medicare reimbursement for continuous positive airway pressure (CPAP; TABLE 1) treatment using a threshold based on a minimum of 2 hours of sleep as measured by PSG. The Epworth Sleepiness Scale is useful for documenting and monitoring daytime sleepiness.2
Treatment
The treatment of CSA is more difficult than the treatment of OSA, and varies according to the specific syndrome. While TABLE 1 outlines the treatment modalities for CSA, it is important to recognize that DIA is just one type of CSA. Primary treatment of DIA is discontinuation of the offending agent and supportive care.6 Furthermore, various agents have been used to treat CSA under a variety of circumstances owing to the heterogeneity of CSA, such that no single agent can be considered a drug of choice.3,4
Benzodiazepines, Opioids, and CYP3A4 Inhibitors
Pathologic changes are non-specific in most drug-induced diseases; with particular regard to drug-induced pulmonary diseases, selected populations may be more susceptible to toxicities associated with specific agents.6 For example, patients with chronic obstructive airway disease, alveolar hypoventilation, and chronic carbon dioxide retention have an exaggerated respiratory depressant response to narcotic analgesics and sedatives.6 Additional comments regarding benzodiazepines, opioids, and CYP 3A4 enzyme inhibitors are discussed below.
Benzodiazepines: Use of a benzodiazepine with another respiratory depressant may produce a profound additive or synergistic effect6:
• IV diazepam use with phenobarbital to stop seizures may result in an ICU admission and assisted mechanical ventilation (regardless of the administration rate)
• Too-rapid administration of a benzodiazepine (e.g., diazepam, lorazepam, midazolam) may cause apnea, even in the absence of other respiratory depressants
• Rapid IV administration of midazolam, when used for conscious sedation prior to medical procedures, has caused respiratory depression and arrests resulting in death and hypoxic encephalopathy. These occurrences have been more frequent in the elderly and the chronically debilitated, or in combination with opioid analgesics.
Opioids: Narcotics such as heroin, morphine, and methadone cause respiratory depression via stimulation of the mu opioid receptors on neurons in the medullary respiratory complex.3 Studies have shown that with chronic narcotic use, abnormal hypercapnic and hypoxic ventilatory responses are reported despite tolerance of many CNS effects of opioids, and CSA is common.3,8,11,12 The FDA has reviewed reports of deaths and life-threatening adverse events such as respiratory depression and cardiac arrhythmias in patients receiving methadone. These adverse events are the possible result of unintentional methadone overdoses, drug interactions, and methadone’s cardiac toxicities (QT prolongation and torsades de pointes).13 Patients need to be thoroughly counseled when this agent is prescribed (see Resources). Because of its long half-life and risk of accumulation, methadone is difficult to titrate and is not considered a drug of first choice, especially in the elderly.14

CYP3A4 Inhibitors: Importantly, CYP enzymes may be responsible for the metabolism (at least partially) of approximately 75% of all drugs, with the CYP3A subfamily responsible for nearly half of this activity.14 CYP inhibition occurs via several potential mechanisms; most commonly, competitively (and reversibly) binding to the active site on the enzyme, thus preventing the substrate from binding to the same site and preventing the substrate from being metabolized.14 Of note, concurrent use of CYP3A4 inhibitors (TABLE 3) with benzodiazepines is likely to lead to greater risk of respiratory depression.6 Examples of clinically important CYP3A4 inhibitors include macrolide antibiotics (e.g., clarithromycin, erythromycin), anti-HIV agents (e.g., ritonavir, delavirdine), and calcium channel blockers (e.g., verapamil, diltiazem).15

Conclusion
The use of drugs that cause CNS depression, respiratory muscle dysfunction, or myopathy can place vulnerable individuals, such as the elderly and the chronically debilitated, at risk for DIA.
Vigilance regarding the use of opioids and benzodiazepines, among others, in the elderly—and in the elderly and other individuals with cardiac, respiratory, or neurologic disease—is necessary to protect those at greater risk of CSA. The use of the resources presented above can assist pharmacists in identifying those patients at additional risk secondary to the use of drugs that cause additive effects or changes in drug metabolism, and are imperative for the delivery of quality pharmaceutical care and safe medication management.
REFERENCES
1. Ramsawh HJ, Bloom HG, Ancoli-Israel S. Sleep, aging and late-life insomnia. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:943-948.
2. Reuben DB, Herr KA, Pacala JT, et al. Geriatrics at Your Fingertips. 17th ed. New York, NY: American Geriatrics Society; 2015:310-312.
3. Becker K, Wallace JM. Central sleep apnea syndromes. Medscape. Updated: March 16, 2015. http://emedicine.medscape.com/article/304967-overview#showall. Accessed June 12, 2015.
4. Panossian LA, Avidan AY. Review of sleep disorders. Med Clin North Am. 2009;93(2):407-425, ix.
5. Anderson P. New guideline for sleep apnea diagnosis. Medscape. Aug 11, 2014. www.medscape.com/viewarticle/829717. Accessed June 15, 2015.
6. Raissy HH, Harkins M, Marshik PL. Drug-induced pulmonary diseases. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York, NY: McGraw-Hill Inc; 2011:511-512.
7. Bixler EO, Vgontzas AN, Ten Have T, et al. Effects of age on sleep apnea in men: I. Prevalence and severity. Am J Respir Crit Care Med. 1998;157(1):144-148.
8. Wang D, Teichtahl H, Drummer O, et al. Central sleep apnea in stable methadone maintenance treatment patients. Chest. 2005;128(3):1348-1356.
9. Mehra R, Stone KL, Varosy PD, et al. Nocturnal arrhythmias across a spectrum of obstructive and central sleep-disordered breathing in older men: Outcomes of Sleep Disorders in Older Men (MrOS sleep) study. Arch Intern Med. 2009;169(12):1147-1155.
10. Eckert DJ, Jordan AS, Merchia P, Malhotra A. Central sleep apnea: pathophysiology and treatment. Chest. 2007;131(2):595-607.
11. Teichtahl H, Wang D, Cunnington D, et al. Ventilatory responses to hypoxia and hypercapnia in stable methadone maintenance treatment patients. Chest. 2005;128(3):1339-1347.
12. Walker JM, Farney RJ, Rhondeau SM, et al. Chronic opioid use is a risk factor for the development of central sleep apnea and ataxic breathing. J Clin Sleep Med. 2007;3(5):455-461.
13. Information for Healthcare Professionals Methadone Hydrochloride text version. Last Updated: August 23, 2013. www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm142841.htm. Accessed June 16, 2015.
14. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 19th ed. Hudson, OH: Lexicomp; 2014:1566-1573.
15. Zhou SF. Drugs behave as substrates, inhibitors and inducers of human cytochrome P450 3A4. Curr Drug Metab. 2008;9(4):310-322.
16. Epocrates Essentials Version 15.5. Updated June 15, 2015. Epocrates.com. Accessed June 17, 2015.
17. Mayo Clinic. Central sleep apnea. June 28, 2013. www.mayoclinic.org/diseases-conditions/central-sleep-apnea/basics/causes/con-20030485?p=1. Accessed June 2, 2015.
18. Quadri S, Drake C, Hudgel DW. Improvement of idiopathic central sleep apnea with zolpidem. J Clin Sleep Med. 2009;5(2):122-129.
19. Bradley TD, Logan AG, Kimoff RJ, et al. Continuous positive airway pressure for central sleep apnea and heart failure. N Engl J Med. 2005;353(19):2025-2033.
20. Ruttanaumpawan P, Logan AG, Floras JS, Bradley TD. Effect of continuous positive airway pressure on sleep structure in heart failure patients with central sleep apnea. Sleep. 2009;32(1):91-98.
To comment on this article, contact rdavidson@uspharmacist.com.





