US Pharm.
2008;33(1):22-26.
Cerebral palsy is the leading
cause of childhood disability.1 Current estimates suggest that
764,000 children and adults in the United States have cerebral palsy.2
The occurrence is 1 to 2.3 cases per 1,000 births.3 The extent of
disability in cerebral palsy can vary from a mild motor disorder to major
cognitive defects and wheelchair dependence. Cerebral palsy is defined as a
primary abnormality of movement and posture secondary to a nonprogressive
lesion in the developing brain. The motor disorder is often accompanied by
epilepsy and disturbances of sight, hearing, cognition, communication, and
behavior.1 This article will review pharmacologic treatments of the
most common movement disorder associated with cerebral palsy--spasticity.
Etiology, Classification,
and Diagnosis
The precise cause
of cerebral palsy is still unknown. It results from the interaction of
multiple risk factors before, during, and after birth.4 While
prematurity and congenital malformations are considered risk factors, in many
cases no specific cause of cerebral palsy is identified.5
Spasticity is the most common
movement disorder of cerebral palsy, affecting 75% of patients. Spasticity is
defined as increased velocity-dependent resistance to movement. It is
characterized by hypertonicity of affected muscles, clonus, and hyperreflexia.
Other aspects of spasticity are decreased motor planning, loss of selective
motor control, weakness, and poor endurance.6 Other movement
disorders of cerebral palsy are less common and may have extrapyramidal
features such as athetosis, chorea, dystonia, and ataxia.
Treatment of Spasticity
Spasticity is
debilitating. It can impact a patient's mobility and activities of daily
living, and may lead to chronic musculoskeletal complications such as
contractures, joint dislocation, pain, and decreased growth.5
Treatment of spasticity involves physical and occupational therapy, orthopedic
interventions, and medications. Individualized care for a patient may involve
a combination of oral, intramuscular, and intrathecal treatments. Oral
treatment options for the control of spasticity in children include baclofen,
clonidine, diazepam, dantrolene, and tizanidine. Their dosages are summarized
in TABLE 1.
Baclofen
Baclofen (Lioresal) is first-line
oral therapy for the treatment of spasticity in children with cerebral palsy.
Baclofen is a structural analog of the inhibitory neurotransmitter gamma-amino
butyric acid (GABA). It acts on the GABA receptors in the central nervous
system (CNS), inhibiting the release of excitatory neurotransmitters that
cause spasticity. Baclofen is well absorbed orally, but it is 30% protein
bound and has low lipid solubility; therefore, it does not readily cross the
blood-brain barrier.7 Even though oral baclofen is considered
first-line therapy, there are few clinical trials published evaluating its use
in children with cerebral palsy.8 The information in pediatrics is
from open-label studies or older, controlled clinical trials.8,9 A
more recent double-blind, placebo-controlled study from 2006 demonstrated that
baclofen improved function in children with cerebral palsy.8
Oral baclofen is approved for
adults and children over 12 years of age. The recommended dosage is 5 mg three
times a day, titrated every three days to a maximum dose of 80 mg. For
children under 12 years, the recommendation is 10 to 15 mg/day, divided three
times a day to maximum dose of 40 mg for children between 2 and 7 years, and
to a maximum of 60 mg for children older than 8 years.10 A liquid
formulation is not commercially available, but there are data on
extemporaneous preparations.11,12
The adverse effects of
baclofen include sedation, confusion, memory loss, and attention deficits.
Other reported adverse effects include weakness, ataxia, and orthostatic
hypotension. Baclofen's effect on seizure activity is still unclear. Baclofen
has been reported to increase, decrease, or have no effect on the incidence of
seizures.13
It is very important for
pharmacists to be aware of a potentially serious complication of baclofen
therapy. An abrupt discontinuation of baclofen may result in a rebound
increase in spasticity, rhabdomyolysis, disorientation, hallucinations, and
seizures.10,14,15
Diazepam
The benzodiazepine
diazepam (Valium) is one of the oldest treatments for spasticity due to
cerebral palsy.14 Benzodiazepines increase the affinity of GABA for
its receptor. Studies comparing baclofen and diazepam found both agents to be
equally effective in treating spasticity. In the comparison trials, patients
receiving diazepam had a higher incidence of sedation than patients receiving
baclofen.6 The sedative properties of diazepam limit its clinical
use. Other common side effects of diazepam are impaired memory, decreased
attention, ataxia, weakness, constipation, and urinary retention.15
The incidence of sedation and impaired memory and attention are particularly
concerning in the school-aged child.
If diazepam is discontinued
abruptly or tapered too quickly, patients will experience a withdrawal
syndrome. Symptoms may include anxiety, agitation, restlessness, irritability,
tremor, nausea, hyperpyrexia, and seizures.15
Dantrolene Sodium
Dantrolene
(Dantrium) prevents full muscle contraction and therefore spasticity by
inhibiting the release of calcium from the sarcoplasmic reticulum of skeletal
muscle cells.15 The prevention of full muscle contraction results
in generalized muscle weakness. It does not affect smooth muscle or cardiac
muscle. Clinical trials determined that dantrolene is superior to placebo in
the treatment of children with spasticity due to cerebral palsy.15
Dantrolene is metabolized
extensively by the liver. It is involved in the following drug interactions:
concomitant use with verapamil can result in hyperkalemia; use of dantrolene
and estrogens may increase hepatotoxicity; and use with other CNS depressants
may increase sedation. The toxicity of dantrolene may also be increased when
used with monoamine oxidase inhibitors.16
Dantrolene's role in the
treatment of spasticity is limited because of its association with
hepatotoxicity. Fatal and nonfatal hepatitis have been reported. One series
reported an incidence of hepatotoxicity of 2% and the incidence of fatal
hepatitis at 0.3%.15 Baseline liver function tests should be
obtained before initiating therapy and regularly thereafter.
Centrally Acting Alpha2
-Agonists
The centrally acting alpha2
-agonists clonidine (Catapres) and tizanidine (Zanaflex) work in the spinal
cord to reduce spasticity by hyperpolarizing motor neurons and reducing
excitatory amino acid release.15
Tizanidine does not have
published dosing recommendations in children and is only available in tablet
form.16 Clonidine has the advantage of many different dosage forms.
It is available as a tablet, a patch, and an extemporaneously compounded
suspension.10 Clonidine is usually used as adjunctive therapy for
the treatment of spasticity.16
Adverse effects of the
centrally acting alpha2-agonists include sedation, hypotension, and
gastrointestinal upset. Not surprisingly, the adverse effect of hypotension is
more common with the use of clonidine. Elevated liver enzymes are associated
with tizanidine therapy.15
Intrathecal Baclofen
For many patients, oral medications
will not adequately control spasticity. For such patients, baclofen delivered
intrathecally could be an option. Baclofen, delivered directly to the spinal
cord through an implantable device, provides improved control of spasticity
with reduced adverse effects. The intrathecal administration of baclofen
results in concentrations in the lumbar cerebrospinal fluid 30 times greater
than can be achieved with oral dosing.17
The goals of intrathecal
baclofen (ITB) therapy are to slow or prevent contractures, improve comfort
and positioning, and ease the burden of care in nonfunctional patients.
Contraindications are presence of infections at the time of screening, history
of allergy to baclofen, inability to implant the pump 2.5 cm from the skin
surface, and concerns about the patient's or family's ability to be compliant
with refill appointments.16,18 It is recommended that the child be
at least 15 kg in weight or 4 years of age for implantation.16,19
There are numerous studies describing the efficacy of ITB in treating
spasticity due to cerebral palsy in adults as well as children.20-23
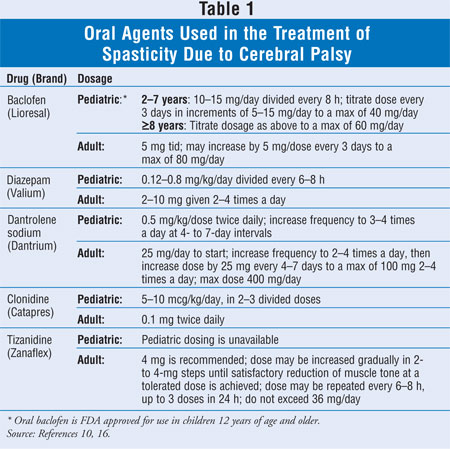
The battery-powered device
contains and delivers drug from the pump reservoir through the catheter to the
intrathecal space by peristaltic action (FIGURE 1). The life of the
battery is four to seven years. The titanium pump is the size of a hockey puck
with a reservoir capacity from 10 to 40 mL.16
If a patient is a candidate
for ITB, a test dose is administered by lumbar puncture. An initial dose of 50
mcg is administered and if no response is noted, subsequent doses of 75 mcg
and 100 mcg are administered 24 hours apart. If no response is seen at any
dose, the patient is not a good candidate for ITB. If a response is evident,
the patient may have a pump implanted. The initial daily dose is calculated by
doubling the screening dose and delivering it continuously over 24 hours.
16
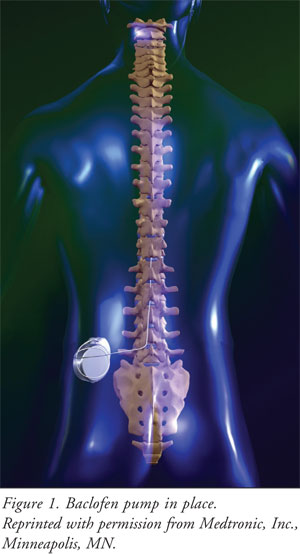
The pump is programmed from
outside the body using a telemetry wand.16 It can be programmed to
deliver a constant rate or a variable rate depending on the patient's needs
during the day.18 The pump does need to be refilled. A needle is
used to enter the device through the skin. Patients must be counseled to
recognize the sound of the alarm when the reservoir is low. Usual refill
interval is every two to three months depending on the dose of baclofen
required by the patient.18
The most common drug-related
adverse effects of ITB are chronic constipation, hypotonia, somnolence,
headache, vomiting, and paresthesias. ITB does have the potential for some
severe complications related to the catheter and the device such as
dislodgement, kinking disconnection, cerebrospinal fluid leaks, and infection.
16,18,20 Overdosage can occur after a large dose increase or an error in
programming. Overdose symptoms are hypotonia, decreased alertness, decreased
respirations, bradycardia, and coma. Another complication is the baclofen
withdrawal syndrome.18 Baclofen for intrathecal use was given a
black box warning from the FDA to alert clinicians that a very serious
withdrawal syndrome can occur with discontinuation of ITB therapy.16
Withdrawal is usually the result of pump or catheter malfunction or neglect
in refilling the reservoir. Mild withdrawal is characterized by pruritus,
agitation, diaphoresis, and increased tone. In moderate-to-severe withdrawal,
fever, tachycardia, and painful muscle spasms can occur. Withdrawal symptoms
can progress to seizures, hallucinations, delirium, rhabdomyolysis, and death.
All patients receiving ITB therapy must be educated about withdrawal symptoms
and should have oral baclofen available for emergency use.16
Local Intramuscular Therapy
Botulinum toxin is used as
adjunctive therapy in the treatment of spasticity. Botulinum toxin is produced
by the anaerobic bacterium Clostridium botulinum. There are seven toxin
subtypes (A, B, C, D, E, F, and G); only A and B are commercially available.
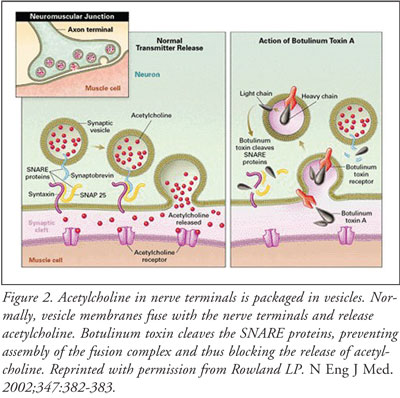
The toxin prevents acetylcholine from being released into the synaptic cleft,
preventing muscle contraction. Within the nerve cell, vesicles containing
acetylcholine fuse with the neuronal cell membrane in order for acetylcholine
to be released. The binding of the vesicle is facilitated by a synaptic fusion
apparatus called soluble NSF attachment receptor (SNARE) proteins. Botulinum
toxin cleaves the fusion proteins and the SNARE complex does not form. This
prevents the acetylcholine-filled vesicles from fusing with the cell membrane
and releasing acetylcholine (FIGURE 2). Neuromuscular transmission is
prevented, resulting in flaccid paralysis and muscle weakness.16
There are two available
preparations of botulinum toxin in the U.S.--subtype A (Botox) and subtype B
(Myobloc). Botox is approved for the treatment of strabismus and blepharospasm
associated with dystonia in patients 12 years and older.24 It is
also approved for hyperhidrosis.24 Botox is currently approved in
Europe for the treatment of spasticity due to cerebral palsy in children. It
does not currently have this indication in the U.S., although spasticity is a
common off-label use.25 Myobloc is approved for cervical dystonia.
26 It is used less frequently than Botox for the treatment of
spasticity.
Botulinum toxin types A and B
have similar onset of action. Declines in muscle action potentials begin one
to three days after injection, and the peak effect occurs in one to three
weeks. The clinical effects from type AÜ last three to four months. Effects
can vary from less than one month to up to six months. The clinical response
from type B is similar to that from type A; the maximum paralysis at two weeks
is less pronounced, and recovery occurs more quickly.16
The Spasticity Study Group has
developed dosing regimens for botulinum toxin based on units/kg of body
weight. The maximum dose for a large muscle is 3 to 6 units/kg and for a small
muscle, 1 to 2 units/kg. The maximum dose per injection site is 50 units.
27
Botulinum toxin has a mild
side-effect profile. Some patients experience pain on injection, muscle
soreness, bruising, excessive weakness in injected and nearby muscles, rash,
and fever. Falls may be due to greater-than-desired weakness and spread of
toxin to adjacent muscles. Diffusion to other muscles is dependent on the
dose, volume, dilution, and number of injections given. Patients should be
counseled to expect a change in gait to avoid accidental injury and falls.
16
Conclusion
Spasticity is the
most common movement disorder of cerebral palsy, exhibiting debilitating
effects on the patient. Pharmacologic therapy of spasticity may be composed of
oral, intrathecal, and intramuscular therapies. A comprehensive treatment plan
for the patient with spasticity involves setting appropriate goals, utilizing
physical therapy, orthopedic therapy, surgical therapy, and pharmacologic
therapy to achieve the desired response.
REFERENCES
1. Green LG, Hurvitz EA. Cerebral palsy. Phys Med Rehabil Clin N Am. 2007;18:859-882.
2. United Cerebral Palsy.
Cerebral palsyñfacts & figures. Available at: www.ucp.org/
ucp_channeldoc.cfm/1/11/10427/10427-10427/447. Accessed November 15, 2007.
3. Murphy N, Such-Neibar T. Cerebral palsy diagnosis and management: the state of the art. Curr Probl Pediatr Adolesc Health Care. 2003;33:146-169.
4. Ashwal S, Russman BS, Blasco PA, et al. Practice parameter: diagnostic assessment of the child with cerebral palsy. Neurology. 2004;62:851-863.
5. Krigger KW. Cerebral palsy: an overview. Am Fam Physician. 2006;73:91-102.
6. Goldstein EM. Spasticity management. J Child Neurol. 2001;16:16-23.
7. Albright AL. Baclofen in the treatment of cerebral palsy. J Child Neurol. 1996;11:77-83.
8. Scheinberg A, Hall K, Lam LT, O'Flaherty S. Oral baclofen in children with cerebral palsy: a double-blind cross-over pilot study. J Paediatr Child Health. 2006;42:715-720.
9. Lubsch L, Habersang R, Haase M, Luedtke S. Oral baclofen and clonidine for treatment of spasticity in children. J Child Neurol. 2006;21:1090-1092.
10. Taketomo CK, Hodding JH,
Kraus DM. Pediatric Dosage Handbook. 13th ed.
Hudson, OH:
Lexi-Comp; 2006:183,347-348,395-396,432-435.
11. Allen LV, Erickson MA. Stability of baclofen, captopril, diltiazem hydrochloride, dipyridamole and flecainide acetate in extemporaneously compounded oral liquids. Am J Health Syst Pharm. 1996;53:2179-2184.
12. Johnson CE, Hart SM. Stability of an extemporaneously compounded baclofen oral liquid. Am J Hosp Pharm. 1993;50:2353-2355.
13. Vincenzo B, Scelsa B, Curci D, et al. Epilepsy and intrathecal baclofen therapy in children with cerebral palsy. Pediatr Neurol. 2005;33:110-113.
14. Verrotti A, Greco R, Spalice A, et al. Pharmacotherapy of spasticity in children with cerebral palsy. Pediatr Neurol. 2006;34:1-6.
15. Krach LE. Pharmacotherapy of spasticity: oral medications and intrathecal baclofen. J Child Neurol. 2001;16:31-36.
16. Haase MR. Cerebral palsy. In: Schmock G, Brundage D, Chapman M, et al, eds. Pharmacotherapy Self-Assessment Program. 5th ed. Kansas City, MO: American College of Clinical Pharmacy; 2006:213-240.
17. Goldstein EM. Spasticity management: an overview. J Child Neurol. 2001;16:16-23.
18. Albright AL, Ferson SS. Intrathecal baclofen therapy in children. Neurosurg Focus. 2006;21:e3.
19. Lioresal intrathecal
(baclofen injection) package insert. Available at: www.medtronic.
com/physician/itb/disclosure-package-insert.html#3. Accessed November 15, 2007.
20. Campbell WM, Ferrel A, McLaughlin JF. Long-term safety and efficacy of continuous intrathecal baclofen. Dev Med Child Neuro. 2002;44:660-665.
21. Butler C, Campbell S. Evidence of the effects of intrathecal baclofen for spastic and dystonic cerebral palsy. Dev Med Child Neuro. 2000;42:634-645.
22. Murphy NA, Irwin MC, Hoff C. Intrathecal baclofen therapy in children with cerebral palsy: efficacy and complications. Arch Phys Med Rehabil. 2002;83:1721-1725.
23. Aswaad Y, Tayem H, Munoz S, et al. Functional assessment following intrathecal baclofen therapy in children with spastic cerebral palsy. J Child Neurol. 2003;18:26-34.
24. Botox. Available at: www.botox.com. Accessed November 15, 2007.
25. Koman LA, Paterson Smith B, Balkrishnam R. Spasticity associated with cerebral palsy in children: guidelines for the use of botulinum A toxin. Paediatric Drugs. 2003;5:11-23.
26. Myobloc (botulinum toxin type B) injectable solution. Available at: www.myobloc.com. Accessed November 15, 2007.
27. Russman BS, Tilton A, Gormley ME. Cerebral palsy: a rational approach to a treatment protocol, and the role of botulinum toxin in treatment. Muscle Nerve Suppl. 1997;6:S181-S193.
To comment on this article, contact editor@uspharmacist.com.





