US Pharm. 2013;38(5):HS8-HS11.
ABSTRACT: Approximately 1 in 10 babies in the United States is born prematurely. Prematurity is the leading cause of death in newborns. Rates of morbidity and mortality are inversely related to gestational age or birthweight. Even infants born 2 or 3 weeks prematurely have an increased risk of death during their first year, compared with term infants. Premature infants suffer short-term and long-term consequences such as hypothermia, respiratory distress, cardiovascular disturbances, and increased hospitalizations, as well as visual, auditory, and learning disabilities. Drug therapy for common ailments affecting premature infants continues to evolve as new data become available.
Infants born before 37 weeks’ gestation are classified as premature. Prematurity is further delineated according to gestational age and birthweight (TABLE 1).1 Compared with term infants, premature infants have higher rates of morbidity and mortality, especially those who are extremely preterm (EPT) (<28 weeks) or extremely small (weight <1,000 g).1

Of the nearly 4 million births occurring in 2011 in the United States, 11.7% were premature.2 This represents a 2% decline from 2010 (11.99%) and an 8% decline from 2006 (12.8%). The drop in premature births is commendable, but still higher than that reported in 1990 (10.62%). The percentage of EPT or very preterm (VPT) infants remained constant, whereas the percentage of infants born between 34 and 36 weeks’ gestation increased from 7.3% in 1990 to 8.3% in 2011.2,3
Preterm births are classified as iatrogenic (medically necessary), spontaneous, or secondary to premature rupture of membranes (PROM). From 1995 to 1996, spontaneous births accounted for 53% of preterm deliveries; iatrogenic births accounted for 33%.4 Recent data indicate a shift: From 2004 to 2005, spontaneous births accounted for 44% of preterm deliveries and iatrogenic births accounted for 47%. The rise in iatrogenic preterm births is attributed to an increase in late preterm births, although evidence suggests that not all are truly medically necessary.5
RISK FACTORS FOR PRETERM BIRTH
There are many risk factors for preterm birth, including socioeconomic factors, psychosocial factors, and maternal and fetal attributes.6 Other factors include congenital malformations, history of substance abuse or smoking, increased maternal age, low socioeconomic status, maternal infection, hypertension, preeclampsia or eclampsia, diabetes, cardiovascular disorders, multiple gestations, prior preterm delivery, race or ethnicity, stress, obesity or poor diet, and use of artificial reproductive technology with subsequent multiple births.7,8
MORTALITY AND MORBIDITY
From 1995 to 2004, mortality in premature infants decreased by 14.9%.9 However, prematurity-related deaths are the number-one cause of infant mortality. The lower the birthweight or gestational age, the higher the mortality rate. Infants born at less than 32 weeks’ gestation are 73 times more likely than term infants to die within their first year.9,10 Even infants born at 34 to 36 weeks’ gestation are three times more likely to die.9,10
COMPLICATIONS
Preterm infants are susceptible to a variety of complications that manifest immediately after birth (short-term) and into childhood (long-term). Short-term complications due to disruption of the safe uterine environment result from immature development and growth. Long-term complications include visual, auditory, and learning disabilities.
Short-Term Complications
Hypothermia: Compared with term infants, preterm infants are more susceptible to cold intolerance because of their large surface area relative to their weight and lack of subcutaneous fat.11 A lower body temperature at birth correlates with lower birthweight and/or younger gestational age.12 An analysis of 5,277 premature infants revealed that 46.9% had a body temperature higher than 36°C, which was associated with a greater risk of late-onset sepsis (11% increase per 1°C decrease) and increased mortality (28% increase per 1°C decrease). Symptoms of cold intolerance include apnea, bradycardia, lethargy, hypotonia, metabolic acidosis, poor feeding, and pallor.4 To prevent hypothermia and maintain a normal body temperature of 36.5°C to 37.4°C (97.7°F-99.3°F), the infant should be dried immediately after birth and wrapped in a prewarmed blanket.13 In addition, a warm delivery-room temperature should be maintained.
Respiratory Distress: Lung development is a complex process, with alveolar-sac maturation, capillary oxygen exchange, and surfactant production peaking just before birth. Upon delivery, the newborn’s lungs adapt, starting out fluid-filled and becoming capable of gas exchange.14 Respiratory distress may occur secondary to transient tachypnea of the newborn (TTN) or apnea of prematurity (AOP). Additionally, disruption of lung development may manifest as respiratory distress syndrome (RDS) and/or bronchopulmonary dysplasia (BPD).
During labor, fluid is removed from the infant’s lungs and drained into the pulmonary circulation and lymph vessels, a process normally completed within 2 hours of birth.14 Premature infants who cannot clear lung fluid will develop TTN, which is generally self-limiting and treated with supportive measures such as oxygen.14 AOP (disruption of breathing >15-20 seconds, plus oxygen desaturation and/or bradycardia), which develops because of an immature respiratory response in the central nervous system, often resolves spontaneously between 36 and 40 weeks’ gestation.15 RDS is characterized by decreased lung surfactant, which reduces alveoli surface tension, preventing alveoli collapse (atelectasis).14 Deficiency of lung surfactant causes further lung injury and may progress to pulmonary edema and impact circulation.
Caffeine citrate, which has been used to treat AOP for more than 30 years, reduces the need for oxygen.15 The recommended loading dose (expressed as caffeine citrate salt) of 20 mg/kg is followed by a maintenance dose of 5 to 10 mg/kg.15 In the Caffeine for Apnea of Prematurity Trial, which included 1,640 children born prematurely (weight 500-1,250 g), subjects who received caffeine (n = 833) showed no improvement in survival or disability at age 5 years versus those who received placebo (n = 807).16 However, subjects treated with caffeine had reduced incidences of BPD at week 36 and neurodevelopmental disability at age 18 to 21 months.17
The American College of Obstetricians and Gynecologists recommends antenatal corticosteroids to decrease the risk of RDS and facilitate fetal lung maturation, as well as reduce infant mortality and morbidity.18 Women at 24 to 34 weeks’ gestation who are in jeopardy of delivering within 7 days and whose fetus has unknown lung maturity should receive antenatal corticosteroids. Beyond 34 weeks’ gestation, neonatal morbidity does not differ statistically from that reported for term infants.19 Studies showing a benefit for antenatal corticosteroid administration were conducted using the intramuscular (IM) route; therefore, the IV route is not recommended.
For women with PROM, a single-course corticosteroid is recommended before 32 weeks’ gestation.19 Therapy may be beneficial for women with PROM at 32 to 33 weeks’ gestation whose fetus who has documented pulmonary immaturity.18 For a single course of antenatal corticosteroids, betamethasone 12 mg IM every 24 hours for two doses or dexamethasone 6 mg IM every 12 hours for four doses is recommended.18
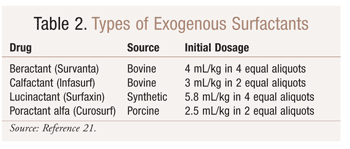
Prophylactic administration of exogenous surfactant within 10 to 30 minutes after birth decreases morbidity and mortality in preterm infants. Clinical trials indicate that infants of less than 30 weeks’ gestation or weighing less than 1,250 g benefit the most.20 Administration within 1 to 2 hours after birth in infants of less than 30 weeks’ gestation with low antenatal corticosteroid exposure and established RDS resulted in decreased occurrence of pneumothorax, pulmonary interstitial emphysema, and the combined outcome of death or BPD compared with surfactant given 2 or more hours after birth.20 Surfactants are administered intratracheally by trained personnel, such as respiratory therapists. Currently available surfactants and usual dosages are listed in TABLE 2.21
BPD: BPD, also known as chronic neonatal lung disease (CNLD), results in significant morbidity and mortality.22 BPD is the most common pulmonary complication in VPT or very-low-birthweight (VLBW) infants, developing in up to 97% of neonates weighing less than 1,250 g.22 In BPD, lung development ceases prematurely, and the alveoli and corresponding vasculature are impaired.14 Because of immature lung function, these infants often require prolonged ventilation.
According to the current definition, which includes specific diagnostic criteria and classifies BPD by severity, BPD is a CNLD in premature infants who require oxygen supplementation for at least 28 days after birth, with gradation of severity according to oxygen concentration and continuous positive airway pressure.22-24 Short-term use of IV furosemide reduces the fluid load when fluid restriction is inadequate.24 Additionally, in a small study, lung function improved in intubated preterm infants older than 3 weeks who received 4 weeks of thiazide diuretic and spironolactone therapy.25 Diuretic use does not impact mortality, however, and it predisposes the infant to serious adverse events (AEs), including electrolyte abnormalities.26 For infants with established BPD, administration of inhaled beta-agonists provides symptomatic treatment.24 The American Academy of Pediatrics issued a policy statement against the routine use of corticosteroids to prevent or treat BPD in VLBW infants.27 The potential benefits of corticosteroid therapy do not outweigh short-term and long-term AEs.
Patent Ductus Arteriosus (PDA): During fetal development, a blood vessel known as the ductus arteriosus forms, connecting the pulmonary artery to the aorta and bypassing the lungs. PDA occurs when the ductus arteriosus fails to close normally, resulting in abnormal blood flow between the aorta and the pulmonary artery. Spontaneous closure occurs in term infants shortly after birth, and in the majority of VLBW infants by day 7.28 When the PDA fails to close, cerebral blood flow decreases, compromising hemodynamics in an already-fragile system. PDA incidence is inversely related to birthweight and gestational age.
The optimal management of PDA has yet to be defined. Prophylactic treatment with cyclo-oxygenase inhibitors or ibuprofen results in PDA closure. IV indomethacin reduces the incidence of symptomatic PDA, the need for surgical closure, and the risk of intravascular hemorrhage (IVH).29 However, long-term neurologic complications and mortality remain unchanged.29 Likewise, IV ibuprofen decreases the incidence of symptomatic PDA and the need for surgical closure, but does not reduce the risk of IVH, mortality, or CNLD.30 Additionally, its use was associated with renal and gastrointestinal (GI) AEs. The use of prophylactic IV indomethacin or ibuprofen is not routinely recommended because of the high likelihood of spontaneous closure and the added AEs.29-31 Indomethacin and ibuprofen have similar AEs, although ibuprofen exhibits less renal toxicity.31 When used to treat symptomatic PDA, these drugs yield a similar rate of closure.32 See TABLE 3 for dosage regimens.21

Long-Term Complications
Surviving preterm infants may struggle throughout life with visual, auditory, and learning disabilities. Greater morbidity occurs as gestational age decreases. Chronic medical conditions are more common in children born EPT compared with normal-weight counterparts.33 Compared with 8-year-olds who had been normal-weight term infants, those who weighed less than 1,000 g at birth were 8.6 times more likely to experience mental delay, 7 times more likely to experience visual disorders, and 6.5 times more likely to have hearing disorders.33
The risk of recurrent hospitalizations increases as gestational age decreases.34 In one study in the United Kingdom, infants were followed for 8 years. From age 9 months to 5 years, 13.9% of subjects born at less than 32 weeks’ gestation, 7.2% born between 32 and 36 weeks, and 3.8% born at term were hospitalized at least three times. Thus, preterm infants were 5.7 times more likely to be hospitalized at least three times. The most common causes of hospitalization during the first 9 months were respiratory disorders, GI disorders, and viral illness or fever. More infants born at less than 32 weeks’ gestation had long-standing illnesses, such as respiratory or GI disorders, at age 5 years (36%) versus those born at 32 to 36 weeks (26%) or 37 weeks (19%).34
CONCLUSION
Premature birth is associated with a variety of complications that affect the infant’s health shortly after birth and well into childhood. The EPT or extremely small infant requires extensive medical interventions, including pharmacotherapy, for which definitive evidence is not available. The pharmacist working with premature infants will need to keep current with evidence as it becomes available.
REFERENCES
1. Tucker J, McGuire W. Epidemiology of preterm birth. BMJ. 2004;329:
675-678.
2. Hamilton BE, Martin JA, Ventura SJ. Births: preliminary data for 2011. Natl Vital Stat Rep. 2012;61:1-19.
3. Martin JA, Hamilton BE, Ventura SJ, et al. Births: final data for 2010. Natl Vital Stat Rep. 2012;61:1-71.
4. Lisonkova S, Hutcheon JA, Joseph KS. Temporal trends in neonatal outcomes following iatrogenic preterm delivery. BMC Pregnancy Childbirth. 2011;11:39.
5. Gyamfi-Bannerman C, Fuchs KM, Young OM, Hoffman MK. Nonspontaneous late preterm birth: etiology and outcomes. Am J Obstet Gynecol. 2011;205:456.e1-e6.
6. McParland PC. Obstetric management of moderate and late preterm labour. Semin Fetal Neonatal Med. 2012;17:138-142.
7. Shapiro-Mendoza CK, Lackritz EM. Epidemiology of late and moderate preterm birth. Semin Fetal Neonatal Med. 2012;17:120-125.
8. Blencowe H, Cousens S, Oestergaard MZ, et al. National, regional,
and worldwide estimates of preterm birth rates in the year 2010 with
time trends since 1990 for selected countries: a systematic analysis and
implications. Lancet. 2012;379:2162-2172.
9. Mathews TJ, MacDorman MF. Infant mortality statistics from the 2009 period linked birth/infant death data set. Natl Vital Stat Rep. 2013;61:1-68.
10. Mathews TJ, MacDorman MF. Infant mortality statistics from the 2008 period linked birth/infant death data set. Natl Vital Stat Rep. 2012;60:1-27.
11. McCall EM, Alderdice F, Halliday HL, et al. Interventions to
prevent hypothermia at birth in preterm and/or low birthweight infants. Cochrane Database Syst Rev. 2010;(3):CD004210.
12. Laptook AR, Salhab W, Bhaskar B. Admission temperature of low birth weight infants: predictors and associated morbidities. Pediatrics. 2007;119:e643-e649.
13. Verklan T. So, he’s a little premature…what’s the big deal? Crit Care Nurs Clin North Am. 2009;21:149-161.
14. Rozance PJ, Rosenberg AA. The neonate. In: Gabbe SG, Niebyl JR, Simpson JL, et al, eds. Obstetrics: Normal and Problem Pregnancies. 6th ed. Philadelphia, PA: Saunders; 2012:481-516.
15. Picone S, Bedetta M, Paolillo P. Caffeine citrate: when and for how long. A literature review. J Matern Fetal Neonatal Med. 2012;25(suppl 3):S11-S14.
16. Schmidt B, Anderson PJ, Doyle LW, et al. Survival without
disability to age 5 years after neonatal caffeine therapy for apnea of
prematurity. JAMA. 2012;307:275-282.
17. Schmidt B, Roberts RS, Davis P, et al. Long-term effects of caffeine therapy for apnea of prematurity. N Engl J Med. 2007;357:1893-1902.
18. ACOG Committee on Obstetric Practice. ACOG Committee Opinion No.
475: antenatal corticosteroid therapy for fetal maturation. Obstet Gynecol. 2011;117:422-424.
19. Surbek D, Drack G, Irion O, et al. Antenatal corticosteroids for
fetal lung maturation in threatened preterm delivery: indications and
administration. Arch Gynecol Obstet. 2012;286:277-281.
20. Engle WA. Surfactant-replacement therapy for respiratory distress in the preterm and term neonate. Pediatrics. 2008;121:419-432.
21. Lexi-Comp Online. Pediatric & Neonatal Lexi-Drugs [online database]. Hudson, OH: Lexi-Comp, Inc; 2013.
22. Groothuis JR, Makari D. Definition and outpatient management of
the very low-birth-weight infant with bronchopulmonary dysplasia. Adv Ther. 2012;29:297-311.
23. Walsh MC, Szefler S, Davis J, et al. Summary proceedings from the Bronchopulmonary Dysplasia Group. Pediatrics. 2006;117(suppl 1):S52-S56.
24. Papoff P, Cerasaro C, Caresta E, et al. Current strategies for treating infants with severe bronchopulmonary dysplasia. J Matern Fetal Neonatal Med. 2012;25(suppl 3):15-20.
25. Stewart A, Brion LP, Ambrosio-Perez I. Diuretics acting on the
distal renal tubule for preterm infants with (or developing) chronic
lung disease. Cochrane Database Syst Rev. 2011;(9):CD001817.
26. Stewart A, Brion LP. Intravenous or enteral loop diuretics for preterm infants with (or developing) chronic lung disease. Cochrane Database Syst Rev. 2011;(9):CD001453.
27. Watterberg KL, American Academy of Pediatrics. Policy
statement—postnatal corticosteroids to prevent or treat bronchopulmonary
dysplasia. Pediatrics. 2010;126:800-808.
28. Nemerofsky SL, Parravicini E, Bateman D, et al. The ductus arteriosus rarely requires treatment in infants > 1000 grams. Am J Perinatol. 2008;25:661-666.
29. Fowlie PW, Davis PG, McGuire W. Prophylactic intravenous
indomethacin for preventing mortality and morbidity in preterm infants. Cochrane Database Syst Rev. 2010;(7):000174.
30. Ohlsson A, Shah SS. Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants. Cochrane Database Syst Rev. 2011;(7):CD004213.
31. Hammerman C, Bin-Nun A, Kaplan M. Managing the patent ductus
arteriosus in the premature neonate: a new look at what we thought we
knew. Semin Perinatol. 2012;36:130-138.
32. Mezu-Ndubuisi OJ, Agarwal G, Raghavan A, et al. Patent ductus arteriosus in premature neonates. Drugs. 2012;72:907-916.
33. Hack M, Schluchter M, Andreias L, et al. Change in prevalence of
chronic conditions between childhood and adolescence among extremely
low-birth-weight children. JAMA. 2011;306:394-401.
34. Boyle EM, Poulsen G, Field DJ, et al. Effects of gestational age
at birth on health outcomes at 3 and 5 years of age: population based
cohort study. BMJ. 2012;344:e896.
To comment on this article, contact rdavidson@uspharmacist.com.
ERRATUM: In the print version of this article, the ibuprofen dosage of 25 mg/kg listed in TABLE 3 is incorrect. The correct dosage is 5 mg/kg.





