US Pharm. 2014;39(10):33-37.
ABSTRACT: Diabetes mellitus affects millions of people, causing many complications. Recent studies have shown a link between diabetes and mild cognitive impairment (CI) in persons with earlier onset and greater severity of diabetes. Although the pathophysiology of CI is multifactorial, there is stronger evidence that lack of glycemic control resulting in hypo- or hyperglycemia leads to CI among diabetic patients. With a growing geriatric population and increasing obesity trends in America, the prevalence of diabetes and the related CI, caregiving could be a challenge. Understanding the predisposing factors and the link between diabetes and CI or dementia can help improve the quality of life of diabetic individuals.
Millions of people around the globe are diagnosed with diabetes, and its incidence is estimated to double by 2030.1 The American Diabetes Association (ADA) estimates that 1.7 million Americans are diagnosed with diabetes every year, which means 4,660 adults newly diagnosed daily.2 There are two primary forms of diabetes, type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM).
Of the 30 million people who currently have diabetes in the United States, 90% to 95% have T2DM.2 T2DM is a group of metabolic disorders resulting in insulin resistance leading to reduced insulin sensitivity and therefore relative insulin insufficiency.1 This chronic disease process is accompanied by complications in various vital organs and can be associated with a higher risk for cardiovascular events, neurologic changes, and kidney impairment.
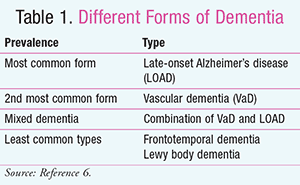
Additionally, recent studies have shown a link between T2DM and mild cognitive impairment (CI), Alzheimer’s disease, and vascular dementia (VaD).3,4 CI refers to changes in memory, mood swings, perception, reaction times, attention, and concentration.5 Although the pathophysiology of CI in diabetic persons is not clear and is believed to be multifactorial, a strong link between lack of glycemic control resulting in hypo- or hyperglycemia is strongly associated with CI.1 Micro- and macrovascular changes associated with T2DM are also linked to the development of dementia.1 With a growing geriatric population and the prevalence of diabetes increasing with obesity trends in America, healthcare professionals need to be alert. TABLE 1 shows the different forms of dementia.6
Since diabetes is a chronic disease with many complications, patients must be involved in their own treatment; but with CI on the rise, clinicians need to ensure that patients be compliant with treatment. Advocating for specific pharmacologic intervention can help decrease the risk of CI. Employing healthcare modalities from a multifaceted team approach is necessary to improve the quality of life of individuals with diabetes and CI.
PATHOGENESIS OF COGNITIVE IMPAIRMENT IN T2DM
Diabetes and diabetes-related CI continue to grow due to the increase in obesity and longevity of the modern-day human population, with significant effect on society. In this light, it is essential to understand the pathophysiological alterations that cause the development and progression of diabetes-related CI and also to devise treatments to reverse or prevent these complications.
Hypoglycemia vs. Hyperglycemia
Hypoglycemia is the number-one reason for temporary or short-term CI.7 A normal and controlled glycemic level is defined as 4 to 7 mmol/L or 72 to 126 mg/dL. Levels can fluctuate in this range, and the diabetic patient can function adequately; but when levels fall below 4 mmol/L, cognitive deficits can be seen. When the brain does not have a sufficient amount of glucose to function, typically defined as <3.5 mmol/L or 63 mg/dL, certain symptoms can present and should be immediately corrected (TABLE 2).6,7 It is possible for these short-term hypoglycemic states to result in permanent brain damage. Even repeated bouts of mild hypoglycemia have been shown to cause CI. Marked decreases in IQ were measured over a 6-year time period in one particular study.8

Many studies have been done on the effects of hypoglycemia and CI.9 The Edinburgh Type 2 Diabetes Study showed that severe hypoglycemia was linked to cognitive impairment later in life. The Fremantle Diabetes Study was able to document dementia and cognitive decline. A recent 7-year follow-up study done in Taiwan used 1 million random subjects as the largest sample size to date. The study found that many different parameters were statistically significant predictors of dementia. Among the parameters studied, female gender, older age, insulin use, and previous episodes of hypoglycemia all put the patients in the study at high risk for dementia. Cases with previous episodes of hospitalized hypoglycemia resulted in a three-fold increase in dementia by the end of the study.9
Hyperglycemia, when prolonged, can also have negative effects such as neuropathy, retinopathy, nephropathy, and CI.1 Brain imaging using MRI of patients with hyperglycemia for long periods of time can show visible lesions. These lesions represent damage to the neurons, which in turn translates to CI in that patient. A decrease in white matter volume in particular has been linked to reduced processing of information and loss of executive function.7 A tighter glycemic control can help to prolong or even to some extent prevent the CI that can occur with T2DM.
Microvascular Changes
The common microvascular complications in diabetes include diabetic nephropathy, neuropathy, and retinopathy. Cerebral microvascular changes as seen in diabetic retinopathy can be the most common complication of T2DM. Predominantly, microvascular disease has been shown to affect the thalamus, basal ganglia, and white matter.10 An increased severity of microvascular changes shows a worse prognosis with regard to mental flexibility, verbal fluency, and processing speed. This was seen more in men than in women. The decline in processing speed was also found with hypertensive retinopathy.11 Executive-functioning deficits are also a concern because of the involvement of frontal subcortical atrophy.10
Macrovascular Changes
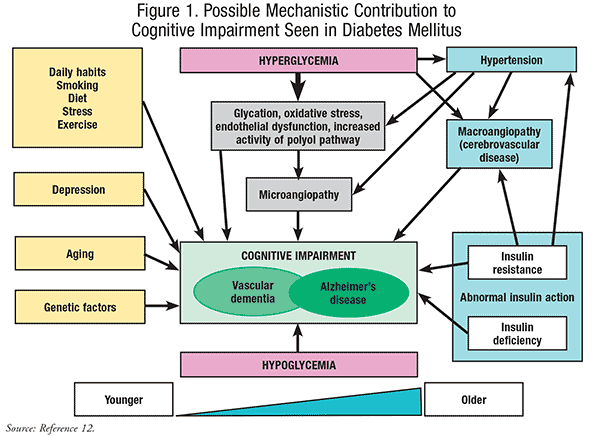
The most noted macrovascular complications in diabetes include coronary artery disease, peripheral arterial disease, and stroke. Atherosclerosis is a leading cause of acute coronary syndrome as well as cerebrovascular accident and is accelerated by hyperglycemia secondary to T2DM. Macrovascular changes can affect heart and brain function by occlusion and ischemia. Cognitive impairment related to stroke alone could be directly linked to prolonged periods of hyperglycemia.7 In addition, diabetes promotes hyperglycemia when not properly controlled, which combined with hypertension and hyperlipidemia, can result in cognitive decline from brain structure atrophy due to large deposits of amyloid and other fatty products.10,11 FIGURE 1 shows the probable mechanisms leading to CI during diabetes.12
Inflammatory Changes
Obesity and T2DM can be associated with some systemic inflammation; this inflammation is believed to increase the risk for vascular disease. It is also thought that the inflammation is directly related to CI because inflammation of the brain itself is much like inflammatory changes seen in dementia. Although most studies have not focused on this link between inflammatory cytokines and CI, some information has been documented. Increases in interleukin (IL)-6, tumor necrosis factor (TNF), and C-reactive protein (CRP) have all been linked to lower cognitive functions.11
DIABETES AND COGNITIVE IMPAIRMENT
Mild Cognitive Impairment
Mild CI is defined as the predementia state that is associated with an increased risk for developing dementia. Mild CI helps to identify individuals who may progress further to CI.11 Mild CI is characterized as memory complaints without a severe effect on activities of daily life.6 Diabetes has been linked to a 1.5-fold increase in the development of mild CI, thus putting diabetic patients at greater risk for dementia. Multiple longitudinal studies have demonstrated that T2DM is directly linked to mild CI and dementia and affects numerous cognitive domains. Mild CI can lead to a decrease in memory, language, verbal memory, executive functioning, abstract reasoning, and information-processing speed.11
Dementia and Alzheimer’s Disease
Dementia is associated with a decrease in memory, communication, and reasoning. There are different kinds of dementia (TABLE 1).6 In 2014, more than 5 million people in the U.S. had Alzheimer’s disease, with 1 in 9 persons aged 65 years suffering from the disease.13 The most common type of dementia is late-onset Alzheimer’s disease (LOAD), followed by VaD. LOAD accounts for 70% to 90% of all cases of dementia.6 There is a 50% to 100% increase in the development of LOAD and VaD in patients with T2DM compared to those without.6 LOAD can be specifically linked to the unregulated glycemic control caused by T2DM. Regions of the brain that are linked to LOAD are particularly high in insulin receptors.14 There can also be an increase of protein deposition in the brain in LOAD and T2DM that can be seen in the pancreas as well.7 Overall, there is increased incidence of LOAD with T2DM.
TRANSLATION TO CLINICAL PRACTICE
Screening
With T2DM being such a complex, chronic metabolic disorder, it requires intact cognition. Independent diabetic self-care is imperative for successful patient outcomes. Glycemic control relies on patient awareness of the condition even at home and is assisted by healthcare professionals with education on self-monitoring of blood glucose (SMBG), insulin dosing, and other pharmacologic interventions that depend upon compliance. Because the disease is associated with other comorbidities, it is part of diabetic care to have many annual screenings. For example, the presence of proteinuria is assessed to monitor kidney function, ophthalmoscopic examinations are performed to look for diabetic retinopathy, and monofilament tests are done to evaluate for diabetic neuropathy. In addition to the conditions already mentioned, as well as many others, CI is now on the rise as yet another commonly associated comorbidity. For clinicians, the Mini-Mental State Examination (MMSE) or another cognitive screening tool should be on the forefront of diabetic patient care. Some recommendations have been made to assist healthcare providers in treating the T2DM patient with regard to the increasing potential for cognitive impairment.
Sending written instructions and materials home for patients who are unable to remember them has been shown to be effective. For others, whose CI prevents reading comprehension, visual aids and even video depictions have been proven helpful for patient compliance with treatment.11
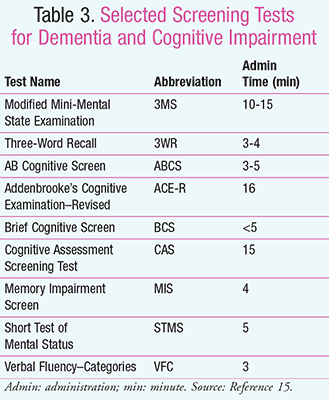
Mental Status Screenings: Different studies have shown trends between specific cognitive screening tools and forms of dementia due to the characteristics of each subtype of dementia. For example, Alzheimer’s disease in the initial stage can be characterized by loss of episodic memory, while VaD has a severe impairment of executive functioning. Frontotemporal dementia has early impairments on letter fluency, and Lewy body dementia has a loss of attention and abstract reasoning. A screening tool that could pick up Alzheimer’s disease might not detect frontotemporal dementia. Mental status screening tools should not only detect the CI, but also identify the most likely etiology. Although the MMSE is the most widely used, others, such as the Addenbrooke’s Cognitive Examination-Revised (ACE-R), help to distinguish Alzheimer’s disease specifically from other subtypes. Clinicians should take into account the many different types of screening tools to be performed (TABLE 3).15
Pharmacologic Management
For insulin-dependent diabetes mellitus (IDDM), strict glycemic control and SMBG are the preferred method of management, according to the ADA. When monitoring after an episode of hypoglycemia, a patient and provider should attempt to identify the cause and prevent any further hypoglycemic episodes due to their strong link to CI and dementia. Monitoring blood glucose before and after meals to prevent future events is recommended.16 Overall, well-controlled glycemic levels are what give the patient the best possibility for positive outcomes.
Some studies recently have argued that insulin significantly increases the prevalence of dementia and should be avoided, if possible. A documented decrease in the incidence of dementia is seen with the use of oral hypoglycemic agents, such as metformin. The use of HMG-CoA reductase inhibitors (statins) has been proven to significantly decrease the risk for dementia as well.12 Another drug class to consider is the peroxisome proliferator-activated receptors (PPARs) because of their efficacy in reducing inflammatory response, enhancing insulin sensitivity, and improving glucose metabolism.17 For example, rosiglitazone, a PPAR- agonist, has been shown to maintain performance on attention tasks and delayed recall. Other recent studies have shown negative effects of rosiglitazone in patients with Alzheimer’s disease with regard to objective cognitive performance.17 More studies and research would benefit the medical community on this dispute.
Investigational Therapies: Another pharmacologic intervention to consider is noninvasive intranasal insulin. The administration of intranasal insulin is argued to be more efficacious because it delivers drugs directly to the brain faster.17 It has also been demonstrated to improve memory in normal adults. Glucagon-like peptide-1 (GLP-1), a stimulator of insulin secretion via oral glucose, has been studied as well. A study in the animal-subject phase showed that cognitive deficits and insulin resistance had been improved by GLP-1. Two specific analogues were reviewed, extendin-4 and Val(8)-GLP-1(7-36). GLP-1 intranasal administration demonstrated improved glycemic control.17 Further studies would be needed to support these claims. Weighing the risks and benefits for all diabetic patients to achieve the best glycemic control while keeping pharmacologic management to an affordable cost for the patient is the preferred treatment on an individual basis.
CONCLUSION
With the rising number of diabetic patients in the U.S., the complications that accompany this metabolic disease process will play a key role in the future of healthcare, both for patients and providers. Providers need to be aware of the increasing prevalence of dementia as well and to recognize the link between two major ailments—diabetes and dementia. With the strong ties between the two with regard to hypo- and hyperglycemia, micro- and macrovascular changes, and other physiological changes in the body, clinicians need to be prepared to treat the whole patient. By using screening techniques to identify dementia early and detect cognitive impairment, patients can have a better chance at managing their diabetes at home with the assistance of medical providers. If clinicians can guide their patients to glycemic control in a healthy range, counsel them in pharmacologic interventions, and educate them on diabetes and all of its complications, then patients will have opportunities for better outcomes.
REFERENCES
1. McCrimmon RJ, Ryan CM, Frier BM. Diabetes 2: diabetes and cognitive dysfunction. Lancet Diabetes Endocrinol. 2012;379:2291-2299.
2. American Diabetes Association. Fast facts: data and statistics about diabetes. http://professional.diabetes.org/admin/UserFiles/0%20-%20Sean/14_fast_facts_june2014_final3.pdf. Accessed September 9, 2014.
3. Bruce D, Davis W, Casey G, et al. Predictors of cognitive decline in older people with diabetes. Diabetes Care. 2008;31:2103-2107.
4. Xu W, Qui C, Winblad B, Fratiglioni L. The effect of borderline diabetes on the risk of dementia and Alzheimer disease. Diabetes. 2007;56:211-216.
5. Cranston I. Diabetes and the brain. In: Shaw KM, Cummings MH, eds. Diabetes: Chronic Complications. 2nd ed. Chichester, England: John Wiley & Sons; 2005:145-168.
6. Luchsinger JA. Type 2 diabetes and cognitive impairment: linking mechanisms. Am J Alzheimers Dis. 2012;30:S185-S198.
7. Wilson V. Cognitive impairment in patients with diabetes. Nurs Stand. 2012;27:44-49.
8. Northam EA, Lin A. Hypoglycemia in childhood onset type 1 diabetes—part villain, but not the only one. Pediatr Diabetes. 2010;11:134-141.
9. Sheu WH, Lin CH. Hypoglycemic episodes and risk of dementia in diabetes mellitus: 7-year follow up study. J Intern Med. 2013;273:102-110.
10. Strachan MW. RD Lawrence Lecture 2010: The brain as a target organ in type 2 diabetes: exploring the links with cognitive impairment and dementia. Diabet Med. 2010;28:141-147.
11. Samaras K, Sachdev PS. Diabetes and the elderly brain: sweet memories? Ther Adv Endocrinol Metab. 2012;3(6):189-196.
12. Parikh NM, Morgan R, Kunik ME, et al. Risk factors for dementia in patients over 65 with diabetes. Int J Geriatr Psychiatry. 2010;26:749-757.
13. Alzheimer’s Association. 2014 Alzheimer’s disease facts and figures. www.alz.org/documents_custom/2014_facts_figures_fact_sheet.pdf. Accessed September 24, 2014.
14. Hugenschmidt C, Williamson JD. Solving the mystery of the diabetes-dementia connection: new clues—but far from “case closed.” J Diabetes Complications. 2014;28:8-9.
15. Cullen B, O’Neill B, Evans JJ, et al. A review of screening tests for cognitive impairment. J Neurol Neurosurg Psychiatry. 2007;78:790-799.
16. Holt P. Taking hypoglycaemia seriously: diabetes, dementia and heart disease. Br J Community Nurs. 2011;16(5):246-249.
17. Chen Z, Zhong C. Decoding Alzheimer’s disease from perturbed cerebral glucose metabolism: implications for diagnostic and therapeutic strategies. Prog Neurobiol. 2013;108:21-43.
To comment on this article, contact rdavidson@uspharmacist.com.






