US Pharm. 2007;32(8):HS3-HS12.
Methicillin-resistant Staphylococcus aureus (MRSA) has been a predominant pathogen in health care settings for more than 40 years. Historically, this microorganism was associated with nosocomial rather than community-acquired infections. Reports of MRSA from within the community began to emerge in 1982; however, these reports were linked to IV drug use or recent contact with a health care setting.1-4 Nevertheless, in the early 1990s, cases of community-associated MRSA (CA-MRSA) emerged sporadically in the United States and throughout the world in various populations without an identifiable link to the health care setting or to previously associated risk factors.1-7 Development of these spontaneous outbreaks has become an ever-increasing concern among health care professionals.3,8,9 Consequently, it is imperative for clinicians to be aware that MRSA is not merely an infection acquired from a health care setting, but that it has uniquely transformed itself to disperse within the community.
Microbiology
S aureus presents as coagulase-positive, gram-positive cocci in clusters
on initial Gram stain and is reported as methicillin-resistant in a large
number of final isolate sensitivity panels. This beta-lactam resistance is
induced via the mecA gene, which alters the penicillin-binding protein
2 (PBP2) by a conformational change in the antibiotic's binding site to form
PBP2a. This results in the reduction of the S aureus affinity
for beta-lactam antibiotics, including penicillins and cephalosporins.
1,3,10
HA-MRSA Versus CA-MRSA
Hospital-acquired MRSA (HA-MRSA) has been defined by the CDC as an MRSA
infection occurring in individuals who have been hospitalized or received
surgery within the last year, who have a permanent indwelling medical device,
who reside in a long-term care facility, or who have recently received
dialysis.11 HA-MRSA strains are genetically and phenotypically
different than CA-MRSA strains. CA-MRSA has been associated with a smaller
composition, a higher incidence of virulence, and a lack of multidrug
resistance (i.e., retaining susceptibility to tetracyclines,
trimethoprim-sulfamethoxazole [TMP-SMX], rifampin, clindamycin, and
fluoroquinolones).1-3,6,12,13
CA-MRSA strains typically contain exotoxins, namely the Panton-Valentine Leukocidin (PVL) toxin, and often occur in immunocompetent patients without identifiable risk factors.2,3,12-15 CA-MRSA strains that contain PVL have the ability to cause severe tissue destruction and leukocyte damage through the formation of pores in the cell membrane, resulting in lesions on the skin and in the respiratory mucosa. 1-3,6,9,13-16
Risk factors that have been linked to the spread of CA-MRSA include openings in the skin, crowded living conditions (such as homeless shelters), frequent skin-to-skin contact (e.g., contact sports), poor hygienic practices, child care attendance, and shared items, including athletic equipment, razors, hair trimming devices, and towels. 1-3,6,11,14,15 Additional risk factors include IV drug use, recent antibiotic use, and exposure to someone who has recently been hospitalized, who resides in a long-term care facility, or who has recently been diagnosed with MRSA.4,11,14,17-20 Outbreaks associated with CA-MRSA have been documented in several settings, such as day care centers and prisons, as well as in military personnel, homosexual men, professional athletes, Native Americans, Native Alaskans, and Pacific Islanders.3,4,7,11,12,19,21 CA-MRSA has been found to disproportionately affect both children and young adults.4,15 Cases of CA-MRSA with no substantial link to these identified risk factors have also been observed.
Clinical Presentation
CA-MRSA strains that contain PVL commonly present as skin and soft tissue
infections (SSTIs), such as boils, pimples, furuncles (boils or superficial
skin abscesses), and cutaneous abscesses, but increasing numbers of invasive
infections have also been reported, including necrotizing pneumonia and
fasciitis, osteomyelitis, septic arthritis, a toxic shock–type syndrome,
bacteremia, lymphadenitis, and myocitis.2,6,9,11,14,21,22
Additionally, it has been noted that PVL-containing MRSA strains may
preferentially bind to damaged respiratory epithelium, which may explain the
link to necrotizing pneumonia following an influenza-like syndrome.2
Any lesions appearing as "spider bites" should include CA-MRSA in the differential diagnosis due to observations of a high correlation of furuncles with CA-MRSA in several studies.2,6,11
Although infrequent, CA-MRSA has also been associated with community-acquired pneumonia, which is most commonly seen after an influenza-type illness.2 One possible way to differentiate pneumonia caused by CA-MRSA is by presentation of hemoptysis after an influenza-like syndrome. In the event of this presentation, the addition of empiric therapy against MRSA may be included in the initial therapy regimen until cultures and sensitivities are determined. At that time, therapy may be deescalated toward the causative organism.2,8,9,11,15,23
Treatment Options
Although beta-lactam antibiotics are the pharmacotherapy of choice for SSTIs,
risk factors for CA-MRSA (e.g., homelessness, high local prevalence) should be
considered during the treatment selection process.2,10 In addition,
since MRSA is ever evolving and has progressed to the community setting--even
in patients without identifiable risk factors--it is crucial to include CA-MRSA
in the differential diagnosis of any patient who presents to a medical
facility with an SSTI.15 It is also important to keep in mind that
few antimicrobial agents have been studied for the treatment of CA-MRSA; thus,
their true efficacy has not yet been proven. Because randomized, prospective
data on the treatment of CA-MRSA is lacking, no single antibiotic regimen is
indicated over others for primary treatment.15 Moreover, resistance
to these agents has been observed at varying rates throughout different
regions.3
Before initiating antimicrobial treatment, incision and drainage or debridement should be conducted (when indicated) for purulent skin lesions, abscesses, or necrotic lesions.4,14,15 Furthermore, the addition of an agent with activity against group A streptococcus should be considered for treating SSTIs--even in areas where CA-MRSA is endemic--since group A streptococcus is commonly identified in these infections.
Adult and pediatric dosages of agents that may be used for the treatment of CA-MRSA are listed in Table 1.
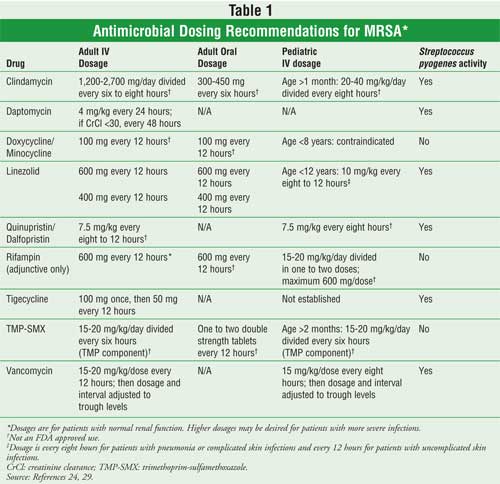
TMP-SMX: TMP-SMX is a potentially useful bactericidal antibiotic used for the treatment of CA-MRSA infections. Although this agent is not FDA approved for the general treatment of staphylococcal infections, several successful cases of its use as MRSA treatment have been reported.10,15 TMP-SMX is a two-component antibiotic that works through inhibition of folic acid synthesis at two separate pathways, thereby inhibiting bacterial DNA synthesis.24
Although large, randomized studies have yet to be conducted to directly evaluate TMP-SMX's efficacy in CA-MRSA, a recent literary analysis of reported data proposed its potential use in high dosages for susceptible MRSA isolates. The analysis concluded that TMP-SMX was useful for a majority of patients and, therefore may therefore be especially beneficial as an oral option for patients with susceptible isolates.10 Conversely, another small retrospective analysis that reviewed 39 outpatients with MRSA infections found a higher rate of recurrence in the group of patients treated with TMP-SMX monotherapy, compared with those who received combination treatment with TMP-SMX and rifampin.4
Major concerns with the use of this agent include treatment failure or disease relapse, development of resistance, adverse reactions, and risk of hypersensitivity reactions to sulfa agents.3 Increases in resistance have been correlated with increased use of TMP-SMX as prophylaxis against pneumocystis pneumonia in patients with HIV.10 Adverse reactions reported with TMP-SMX primarily include gastrointestinal complaints. More serious reactions include bone marrow suppression, nephrotoxicity, and rhabdomyolysis.24 Since group A streptococcus , acommon organism cultured in SSTIs, is not effectively treated by TMP-SMX, the addition of an agent with coverage--such as clindamycin or beta-lactam--should be considered upon initial empiric therapy.10
Clindamycin: This agent is a bacteriostatic, time-dependent lincosamide antibiotic that works via inhibition of the 50S ribosomal subunit to inhibit protein synthesis. 24 It is indicated in serious S aureus infections, but it does not have FDA approval for treatment of MRSA. Clindamycin may be a viable therapeutic option for CA-MRSA, as successful outcomes with this agent have been reported.22
It is vital, however, for clinicians to recognize that inducible macrolide-lincosamide-streptogramin B (iMLSB) resistance has been observed in reportedly susceptible isolates.3 A key indicator for the potential of this inducible resistance is a displayed resistance to erythromycin on the culture's sensitivity panel. CA-MRSA isolates presenting in this manner may have the erm gene, which allows an inducible resistance mechanism that can be turned on upon exposure of the pathogen to clindamycin.1-3,5 To test for the isolate's ability to induce resistance in an erythromycin-resistant, clindamycin-susceptible strain, a double disk diffusion test, also known as a D test, can be performed. Clindamycin and erythromycin disks are placed on an agar plate 15 to 20 mm apart. The plate is streaked with a sample of the MRSA that is to be tested and is then incubated for 24 hours. Following the incubation period, if a flattened area that invades the zone of inhibition and mimics a "D" appearance is seen around the clindamycin disk on the side adjacent to the erythromycin disk, this D formation is considered a positive test.17,22 If the test is positive, another agent should be chosen for therapy. If the test is negative, clindamycin may be considered as a therapeutic option for CA-MRSA, pending the severity of the infection. Clindamycin is not recommended for treatment of CA-MRSA infections unless the D test is performed on the isolate being treated.3
A small study, conducted at Texas Children's Hospital in Houston, looked at the use of clindamycin for treatment of invasive CA-MRSA infections versus methicillin-susceptible S aureus (MSSA) infections in immunocompetent patients. Infections included in the study were bacteremia, pneumonia, septic arthritis, osteomyelitis, pyomyositis, abscesses, bursitis, and lymphadenitis. Children with a history of immunodeficiency were excluded from the study. Of the 46 infections caused by CA-MRSA, all isolates that tested resistant to erythromycin underwent the D test to screen for MLSB resistance. Only one isolate undergoing the D test was positive for the MLSB resistance phenotype. Thirty-nine patients in the MRSA group received clindamycin therapy and 35 (90%) of these patients received a surgical procedure in addition to their pharmacologic treatment. Dosing of clindamycin varied from 30 mg/kg/day to 40 mg/kg/day divided every eight hours. All patients in the analysis had either a significant improvement or were completely cured at discharge. This small study demonstrated the potential efficacy of clindamycin as a treatment option for patients seen in an area with demonstrated susceptibility patterns.22
Although clindamycin may be a potential option for invasive CA-MRSA treatment when the disease is proven completely susceptible, practitioners may consider the addition of a bactericidal agent for the treatment of life-threatening infections.22,25 Some concerns associated with clindamycin are the potential for increased resistance, the ability to induce resistance, and its adverse reaction profile. It is important to be aware that varying rates of clindamycin resistance have been reported, and with amplified use, the incidence of resistance will likely increase.5 Furthermore, to avoid the possibility of inducible resistance during therapy, clindamycin should not be used when the D test is not completed and the MLSB resistance phenotype is suspected. Finally, the primary side effects associated with clindamycin use include gastrointestinal disturbance, which manifests as diarrhea, nausea and vomiting, and Clostridium difficile colitis.24
Potential benefits of this agent include its activity against beta-hemolytic streptococci, another possible pathogen involved in SSTIs, and an excellent bioavailability profile that provides patients the benefit of being converted to oral therapy for completion of their antibiotic regimen.2
Rifampin: Rifampin acts by binding the beta subunit of DNA-dependent RNA polymerase, ultimately inhibiting bacterial RNA synthesis.24 Although a majority of CA-MRSA isolates are reportedly susceptible to rifampin, resistance emerges rapidly when it is used as monotherapy. Therefore, rifampin monotherapy is not recommended; however, this agent may be used in combination with TMP-SMX or clindamycin.3,15,17 Despite rifampin's potential drug interactions, it appears to be a safe and cost-effective treatment when used in combination therapy for CA-MRSA infections.
Minocycline and Doxycycline: Tetracyclines, namely minocycline and doxycycline, are bacteriostatic, time-dependent antibiotics that inhibit bacterial protein synthesis by binding to the 30S ribosomal subunit.24 Although both agents are FDA approved for the treatment of S aureus infections, they are not indicated for the treatment of methicillin-resistant infections. However, it has been suggested that either agent may be an alternative choice for milder, tetracycline-susceptible SSTIs in nonpregnant females, as well as for persons older than 8.15 Adverse reactions commonly reported with these agents include gastrointestinal disturbance, photosensitivity, and tooth discoloration.24
Fluoroquinolones: Fluoroquinolones are bactericidal antibiotics that act through the inhibition of the DNA gyrase A subunits to block bacterial DNA replication and transcription.24 While newer fluoroquinolones have been approved by the FDA for uncomplicated skin infections secondary to S aureus, none are approved for treatment of MRSA infections. Fluoroquinolones should be reserved for second- or third-line therapy. Varying rates of susceptibility to fluoroquinolones have been reported for CA-MRSA isolates, yet rapid increases in fluoroquinolone resistance have also been reported. 14,17
Additionally, selection of MRSA (i.e., conversion of MSSA to MRSA) has been reported with the use of fluoroquinolones, leading to treatment failure and relapse.2,3,15 Treatment failures due to persistent infection or delayed response have also been reported in susceptible isolates. Because these higher rates of recurrence have been associated with fluoroquinolone use, initiating a higher than recommended dosage may help achieve a desired outcome, as fluoroquinolones are concentration-dependent antimicrobials. Regardless, clinicians must appreciate the risk for both MRSA selection and potential treatment failure with the use of this class of antimicrobials.
Common adverse reactions associated with fluoroquinolone use include gastrointestinal discomfort, glycemic instability, and headache. More severe reactions related to fluoroquinolone use include blood dyscrasias, central nervous system disturbances, and QTc interval prolongation.24
Vancomycin: Vancomycin has traditionally been the gold standard for treatment of resistant gram-positive infections, especially in severely ill patients. 10,14,15 It is a bactericidal antibiotic that inhibits bacterial cell wall synthesis through binding of the precursor D-ala-D-ala units of the cell wall. While vancomycin remains the treatment of choice in most cases of MRSA, it has been found that two forms of S aureus have arisen with reduced susceptibility to vancomycin.5,26
Vancomycin is also typically used as the treatment of choice in MRSA pneumonia, but achieving therapeutic concentrations in the alveolar tissue can be difficult due to tissue penetration. Because van comycin is a time-dependent antibiotic, treatment should be focused on maintaining continuous trough levels between 15 and 20 mcg/mL.27
Linezolid: Linezolid is an oxazolidinone antibiotic introduced to the market in 2000. Included in its indications are skin and skin structure infections and pneumonia caused by MRSA and infections caused by the vancomycin-resistant Enterococcus species. Linezolid binds to the 23S ribosomal RNA of the 50S subunit, preventing the formation of the 70S initiation complex and fundamentally inhibiting bacterial protein synthesis.24
Controversy exists regarding the superiority of linezolid over vancomycin for the treatment of pneumonia and skin and skin structure infections.28,29 However, no clear evidence has been established to support use of one agent over the other. Due to the fact that there has been a longstanding history of successful vancomycin use in MRSA infections and its cost is considerably less, linezolid remains second line to vancomycin in a majority of institutions.5 Furthermore, although controversial, one small analysis comparing treatment of CA-MRSA skin infections suggested that initial treatment with linezolid was beneficial when oral therapy was desired, since TMP-SMX and fluoroquinolones are associated with higher treatment failure rates.4 However, one must always be aware of the risk of resistance with newer agents when superiority remains unclear.
Clinicians should be aware of the adverse effect profile of linezolid, in addition to the potential for S aureus resistance. The most commonly reported side effects include diarrhea, nausea, and headache. Reversible myelosuppression, which is dose- and duration-dependent, is a rare, more serious side effect that is frequently observed in patients receiving treatment for more than two weeks; however, this side effect has been found to occur after shorter durations of treatment. Therefore, weekly monitoring of complete blood count panels is strongly recommended.15 Additional serious adverse reactions reported with linezolid use include lactic acidosis and optic and peripheral neuropathy. 24
A significant advantage of linezolid therapy over other antibiotics that are reserved for use with resistant infections is its availability in an oral dosage form. Linezolid treatment is more conducive to long-term outpatient therapy, which may allow for an abbreviated hospital stay when indicated.
Tigecycline: Tigecycline, a derivative of minocycline, is a new glycylcycline antibiotic that received FDA approval in 2005 for the treatment of intra-abdominal and skin and skin structure infections caused by several microorganisms, including MRSA. Tigecycline binds to the 30S ribosomal subunit, ultimately inhibiting protein synthesis. The most commonly reported adverse events include gastrointestinal disturbances, headache, fever, and elevated hepatic enzymes. 24
Since tigecycline is a new antibiotic that shows promise for a broad range of pathogens, reservation of its use for more serious and resistant infections is essential to maintain its full spectrum of activity.
Daptomycin: The FDA approved daptomycin in 2003 for the treatment of skin and skin structure infections caused by MSSA, MRSA, and Enterococcus and Streptococcus species. Daptomycin is a rapidly bactericidal lipopeptide antibiotic that works by calcium-dependent binding to the bacterial cell membrane, which causes rapid membrane depolarization and potassium ion efflux through channel formation. This action leads to bacterial cell death by arresting DNA, RNA, and protein synthesis.24 A potential benefit of daptomycin is that its mechanism of action allows bacteria to remain intact, thus inhibiting their ability to release any toxins.5
If daptomycin is chosen for treatment, it is important to recognize that it should not be used for the treatment of pneumonia due to inactivation of the drug by pulmonary surfactant.2,5 Additional concerns with daptomycin include the risk of headache, diarrhea, and elevated creatine kinase.5,24
Quinupristin/Dalfopristin: Quinupristin/dalfopristin was approved in 1999 for the treatment of severe skin and skin structure infections. Although it is not approved for use against MRSA, it has demonstrated activity against this microorganism. This combination streptogramin antibiotic binds to the 50S ribosomal subunit at two different sites to disrupt both early and late stages of protein synthesis. Major adverse events associated with this agent are infusion site reactions, including pain and inflammation. Therefore, a central line for quinupristin/dalfopristin administration is recommended.24
Preventing Transmission
Basic self-care practices to help
prevent CA-MRSA outbreaks include daily showers with hot water; use of
antibacterial cleansers; regular hand washing; proper fingernail maintenance;
bathing prior to entering communal areas (e.g., whirlpools or saunas); and
prohibiting the shared use of personal items, such as razors, trimming
devices, or towels.2-4,6,12 In addition, proper cleansing of
athletic equipment with diluted bleach or commercial disinfectants and
laundering of shared linens in hot water may reduce transmission.2,3,14
Finally, if CA-MRSA skin infection is suspected, open wounds or purulent
lesions should be identified, and the wound should be appropriately covered.
Individuals lacking the ability to adequately keep draining wounds covered
with dry dressings or maintain appropriate hygiene should refrain from contact
activities until the lesions are healed.2,15
Role of Colonization Eradication
Colonization rates within the
community range from 0.2% to 2.8%, with the highest rates among the homeless
and IV drug users.6 Although the association between CA-MRSA
colonization and infection is not fully understood--since minimal data are
available--attempts to eliminate colonization have been discussed.15
It is not recommended to eradicate colonization in all patients. Elimination of colonization may be considered for patients with recurrent infections despite adequate therapy, those experiencing continued transmission of CA-MRSA and living in close proximity, or those who have high person-to-person contact.17 When indicated, body washes with 4% chlorhexidine gluconate or 7.5% to 10% povidone-iodine, intranasal 2% mupirocin or 0.05% cetylpyridinium chloride, and combinations of systemic antibiotics have been suggested and tried.2,4,6,14
When considering colonization eradication, it is important to keep in mind that colonization occurs at extranasal sites, such as the axilla, rectum, and perineum. Due to varied sites of colonization, use of intranasal anti-MRSA ointment may not fully eliminate all MRSA colonization. Additionally, it is important to keep in mind that although eradication of colonization may be successful initially, future recolonization is commonly observed.15
Conclusion
Increasing resistance and various
susceptibility patterns have been noted in CA-MRSA strains. Though difficult
to predict, these strains may obtain resistance patterns similar to those of
their health care–related counterparts in the near future.2
Additional studies involving treatment and epidemiology, as well as
region-specific resistance monitoring, are crucial to understanding and
ensuring treatment efficacy of this endemic pathogen. Although several new
antimicrobials are available for the treatment of resistant gram-positive
infections, including MRSA, it is critical to reserve therapy with these novel
agents in order to prevent further emergence of resistance. Vancomycin remains
a first-line agent for MRSA treatment until clinically significant studies
evaluate superiority and new recommendations are formed.
Moreover, although several long-standing oral agents appear to be effective against this relatively new strain of MRSA, large, randomized clinical trials have yet to be conducted to assess the true efficacy of one agent over another. These antimicrobial options should therefore be evaluated in clinical studies before specific treatment protocols can be recommended.
In short, a myriad of evidence supports MRSA as an organism that now has the ability to cause disease sporadically throughout the community. For this reason, it is of the utmost importance that pharmacists recommend the most viable regimen against this virulent pathogen.
References
1. Dietrich DW,
Auld DB, Mermel LA. Community-acquired methicillin-resistant Staphylococcus
aureus in sourthern New England Children. Pediatrics.
2004;113:e347-e352.
2. Kowalski TJ, Berbari
EF, Osmon DR. Epidemiology, treatment, and prevention of community-acquired
methicillin-resistant Staphylococcus aureus infections. Mayo Clin
Proc. 2005;80:1201-1208.
3. Rybak MJ, LaPlante
KL. Community-associated methicillin-resistant Staphylococcus aureus: a
review. Pharmacotherapy. 2005;25:74-85.
4. Iyer S, Jones DH.
Community-acquired methicillin-resistant Staphylococcus aureus skin infection:
a retrospective analysis of clinical presentation and treatment of a local
outbreak. J Am Acad Dermatol. 2004;50:854-858.
5. Akins RL, Haase KK.
Gram-positive resistance: pathogens, implications, and treatment options:
insights from the Society of Infectious Diseases Pharmacists.
Pharmacotherapy. 2005;25:1001-1010.
6. Frazee BW, Lynn J,
Charlebois ED, et al. High prevalence of methicilin-resistant Staphylococcus
aureus in emergency department skin and soft tissue infections. Ann Emerg
Med. 2005;45:311-320.
7. Lee MC, Rios AM,
Aten MF, et al. Management and outcome of children with skin and soft tissue
abscesses caused by community-acquired methicillin-resistant Staphylococcus
aureus. Pediatr Infect Dis J. 2004;23:123-127.
8. Frazee BW, Salz TO,
LambertL, Perdreau-Remington F. Fatal community-associated
methicillin-resistant Staphylococcus aureus pneumonia in an immunocompetent
young adult. Ann Emerg Med. 2005;46:401-404.
9. Miller LG,
Perdreau-Remington F, Rieg G, et al. Necrotizing fasciitis caused by
community-associated methicillin-resistant Staphylococcus aureus in Los
Angeles. N Engl J Med. 2005;352:1445-1453.
10. Grim SA, Rapp RP,
Martin CA, Evans ME. Trimethoprim-sulfamethoxazole as a viable treatment
option for infections caused by methicillin-resistant Staphylococcus aureus.
Pharmacotherapy. 2005;25:253-264.
11. Centers for Disease
Control and Prevention. CA-MRSA Information for Clinicians. Available at:
www.cdc.gov/ncidod/dhqp/ar_mrsa_ca_clinicians.html. Accessed July 7, 2007.
12. Kazakova S, Hageman
JC, et al. A clone of methicillin-resistant Staphylococcus aureus among
professional football players. N Engl J Med. 2005;352:468-475.
13. Chambers HF.
Community-associated MRSA--resistance and virulence converge. N Engl J
Med. 2005;352:1485-1487.
14. Cohen PR, Grossman
ME. Management of cutaneous lesions associated with an emerging epidemic:
community-acquired methicillin-resistant Staphylococcus aureus skin infections
. J Am Acad Dermatol. 2004;51:132-135.
15. Gorwitz RJ,
Jernigan DB, Powers JH, Jernigan JA; Participants in the CDC Convened Experts'
Meeting on Management of MRSA in the Community. Strategies for Clinical
Management of MRSA in the Community: Summary of an experts' meeting convened
by the Centers for Disease Control and Prevention. 2006. Available at:
www.cdc.gov/ncidod/dhqp/ar_mrsa_ca.html. Accessed March 28, 2006.
16. Boubaker K, Diebold
P, Blanc DS, et al. Panton-valentine leukocidin and staphylococcal skin
infections in schoolchildren. Emerg Infect Dis. 2004;10:121-124.
17. Dellit T, Duchin J,
et al. Interim guidelines for evaluation and management of
community-associated methicillin-resistant Staphylococcus aureus skin
and soft tissue infections in outpatient settings. Infectious Disease Society
of Washington. 2004. Available at:
www.doh.wa.gov/topics/Antibiotics/MRSAInterim.htm. Accessed March 20, 2006.
18. Seybold U,
Kourbatova E, Johnson JG, et al. Emergence of community-associated
methicillin-resistant Staphylococcus aureus USA300 genotype as a major cause
of health care-associated blood stream infections. Clin Infect Dis.
2006;42:647-656.
19. Baum SE, Morris JT,
Dooley DP, Watson R. Methicillin-resistant Staphylococcus aureus in an adult
military beneficiary population lacking risk factors: susceptibility to orally
available agents. Military Med. 2003;168:126-130.
20. Baggett HC,
Hennessy TW, Rudolph K. Community-onset methicillin-resistant Staphylococcus
aureus associated with antibiotic use and the cytotoxin Panton-Valentine
leukocidin during a furunculosis outbreak in rural Alaska. J Infect Dis
. 2004;189:1565-1573.
21. Four pediatric
deaths from community-acquired methicillin-resistant Staphylococcus aureus
--Minnesota and North Dakota, 1997-1999. MMWR Morb Mortal Wkly Rep
. 1999;48(32):707-710.
22. Martínez-Aguilar G,
Hammerman WA, Mason EO, Kaplan SL. Clindamycin treatment of invasive
infections caused by community-acquired, methicillin-resistant and
methicillin-susceptible Staphylococcus aureus in children. Pediatr Infect
Dis J. 2003;22:593-598.
23. Gillet Y, Issartel
B, et al. Association between Staphylococcus aureus strains carrying gene for
Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young
immunocompetent patients. Lancet. 2002;359:753-759.
24. Lacy C, Armstrong
L, et al. Drug Information Handbook. 14th ed. Hudson, Ohio: Lexi-Comp;
2006.
25. Clinical
Pharmacology. Available at: cpip.gsm.com. Accessed April 17, 2006.
26. Martínez-Aguilar G,
Avalos-Mishaan A, Hulten K. Community-acquired, methicillin-resistant and
methicillin-susceptible Staphylococcus aureus musculoskeletal infections in
children. Pediatr Infect DisJ. 2004;23:701-706.
27. Rappa L,
Larose-Pierre M, et al. Vancomycin resistance: means of prevention, control,
and treatment revisited. Formulary. 2004;39:490-505.
28. American Thoracic
Society and Infectious Diseases Society of America. Guidelines for the
management of adults with hospital-acquired, ventilator-associated, and
healthcare-associated pneumonia. Am J Respir Crit Care Med.
2005;171:388-416.
29. Wunderink RG, Rello
J, Cammarata SK. Linezolid vs vancomycin: analysis of two double-blind studies
of patients with methicillin-resistant Staphylococcus aureus nosocomial
pneumonia. Chest. 2003;124:1789-1797.
To comment on this article, contact editor@uspharmacist.com.





