US Pharm. 2011;36(8):63-66.
Patients with diabetes have a 30-fold higher risk of lower-extremity amputation due to infection compared with patients without diabetes.1,2 Diabetic foot infections that are not appropriately treated because of delayed diagnosis or that are inadequately treated lead to lower-extremity amputation in approximately 10% of patients.3,4 Lower-extremity amputations may be debilitating and can dramatically affect the patient’s quality of life. Successful outcome depends upon prompt identification of the infection, followed by appropriate antibiotic therapy in conjunction with good wound care and judicious use of surgical procedures when warranted.4,5
Community pharmacists can play an integral role in educating patients about foot care and in recognizing ulcers that can lead to skin infections such as cellulitis, which involves the epidermis, dermis, and—in more complicated cases—subcutaneous tissue.6 Resulting from a cut, abrasion, trauma, or puncture, cellulitis may lead to diabetic foot infection. The presence of a foot wound does not necessarily signify infection; however, an existing infection must be treated. Infection is indicated by the presence of purulent secretions or at least two of the following symptoms: erythema, warmth, tenderness, pain, and induration. The clinician should also be alert for friable tissue, wound tenderness, and/or foul odor.3
Risk Factors
Patients with diabetes have a 12% to 25% risk of developing diabetic foot infections due to neuropathy—sensory, motor, and/or autonomic disturbances in which the patient loses the ability to recognize injury or excessive pressure, resulting in foot ulcerations that can develop into infection.3 In addition, peripheral arterial disease impairs blood flow and restricts the body’s ability to fight infection. Other risk factors include hyperglycemia, which can compromise the body’s immune response, and degenerative joint disease (e.g., Charcot’s disease).3
Patients and health care providers should routinely inspect the feet and recognize ulcers that could potentially become infected. The dorsal toes and the plantar surface are the most common sites for ulcers. Ulcers at these locations tend to be superficial and do not extend below the subcutaneous fascia. However, if not treated properly, these ulcers can eventually involve deep tissue.6-8
Classification
Many different classification systems exist for staging diabetic foot ulcers, but none are universally accepted. A classification system developed by the International Working Group on the Diabetic Foot is described by the acronym PEDIS: perfusion, extent/size, depth/tissue loss, infection, and sensation (TABLE 1).1,8,9 The keys to classification include the depth of the lesion (depth of tissue involvement), the presence of ischemia (pedal pulses or diminished blood pressure in the foot), and clinical signs of infection.7 Based on these findings, the infection is stratified as mild, moderate, or severe, and antibiotics are used to eradicate it.8-10

Microbiology
Cultures obtained by biopsy, ulcer curettage, or aspiration are preferable to those from wound swab specimens. Information obtained from cultures can help the clinician make the initial antibiotic choice.8,9 The goal of antimicrobial treatment is to cure the infection, not heal the wound.2 Antibiotics should be discontinued when signs and symptoms of infection have resolved, even if the wound has not healed.7
The most common pathogens that cause diabetic foot infection are aerobic gram-positive cocci, particularly Staphylococcus aureus, beta-hemolytic streptococci (especially group B), and coagulase-negative staphylococci. Patients with chronic foot ulcers and those who recently received antibiotic treatment have a combination of gram-negative bacilli, obligate anaerobes, and gram-positive cocci.3,7,9,10 The presence of methicillin-resistant S aureus (MRSA) should be part of the workup in these patients, as this virulent pathogen is associated with poor wound healing and increased risk of extremity amputation.5-7
Management
Initial management consists of wound cleaning, débridement of necrotic or gangrenous material, and probing the affected area for foreign bodies.3,7,8 Relief of pressure on the ulcer—also known as offloading—is important because it is central to wound healing.3,7
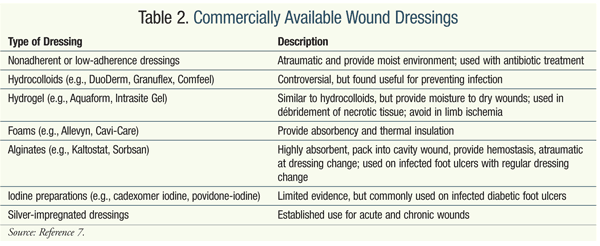
Wound dressings can help maintain a moist environment, thereby facilitating healing. Most clinicians use wound dressings to manage foot ulceration, and various types are available commercially (TABLE 2).7,10-12 Adequate studies evaluating the different dressings are necessary; currently, clinicians use dressings based on their experience. Both clinical and community pharmacists should familiarize themselves with these products in order to advise the health care team and the patient regarding their properties. Although clinical data regarding the use of topical antibiotics (e.g., silver sulfadiazine, neomycin, polymyxin B, gentamicin, mupirocin) are limited, clinicians may choose to employ dressings in conjunction with antibiotics.1,6,7
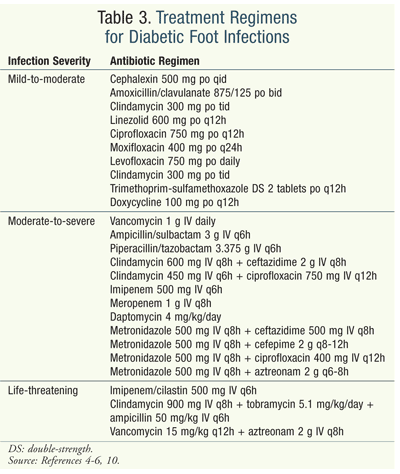
It is generally accepted by clinicians that the majority of patients with mild (and sometimes moderate) infection can initially be treated on an outpatient basis with narrow-spectrum antibiotics that act against the Staphylococcus and Streptococcus species, with subsequent therapy based on clinical response (TABLE 3).4-10 Other factors to consider are recent antibiotic exposure and culture results. Treating mild infections with oral antibiotics for 7 to 14 days is preferred but not always possible, depending upon the patient’s response to therapy and the severity of the infection.1,7 The classification of moderate infections is challenging because the term moderate covers a broad spectrum of complicated and limb-threatening wounds.9 The patient should be started on an empiric antibiotic regimen, with therapy tailored based on clinical response.10,13
The patient should be hospitalized if the infection worsens and fails to respond to oral antibiotics, or if signs of systemic toxicity are present. Hospitalization is necessary in patients with barriers that prevent appropriate wound care or offloading and in those who are noncompliant with antibiotics, metabolically unstable, or in need of close monitoring.7-9
Severe infections should be treated intravenously with broad-spectrum antibiotics (e.g., imipenem/cilastatin), new fluoroquinolones (levofloxacin, ciprofloxacin), or third- or fourth-generation cephalosporins (ceftazidime, cefuroxime) and beta-lactam/beta-lactamase inhibitors (ampicillin/sulbactam, piperacillin/tazobactam) (TABLE 3).4-6,10 Patients in whom MRSA is suspected should be given a regimen that includes vancomycin or linezolid (TABLE 3).6-8,10,12 (Clinical data favoring any particular antibiotic regimen are lacking; more research is needed.) These patients may require 2 to 3 weeks of antibiotic treatment followed by 3 to 4 weeks of oral therapy. In more severe cases, the patient can be on a longer antibiotic regimen.10
A vascular surgeon should be consulted for an infection that fails to respond to antimicrobial therapy. Surgical interventions such as revascularization procedures may be necessary to improve therapeutic outcome.7,8 Additionally, in more severe cases, osteomyelitis—which is beyond the scope of this article—should be ruled out.
Patient Education
Patients should be encouraged to inspect their feet daily and to apply water-based moisturizer. Elastic socks and hosiery should be avoided, as they impair circulation. At home, pathways should be well lighted and free of clutter. Nails should be clipped with a safety clipper; patients with vision problems should have their nails clipped by a caretaker or physician. Proper footwear is extremely important. Shoes should be comfortable, and those that fit poorly or have caused ulcers in the past should be avoided. Smoking cessation should be emphasized, as should exercise and daily blood-sugar monitoring.14
Conclusion
The treatment of diabetes-related foot ulcers can be challenging, as various opinions exist regarding classification and treatment. Nonetheless, successful outcomes are possible with prompt diagnosis and effective treatment. Patients should be educated to routinely inspect their feet, and the importance of appropriate footwear should be emphasized. Patients who are actively involved in their care can improve their quality of life and have a better outcome.
REFERENCES
1. Richard JL, Sotto A, Lavigne JP. New insights in diabetic foot infection. World J Diabetes. 2011;2:24-32.
2. Lavery LA, Armstrong DG, Wunderlich RP, et al. Risk factors for foot infections in individuals with diabetes. Diabetes Care. 2006;29:1288-1293.
3. Cavanagh PR, Lipsky BA, Bradbury AW, Botek G. Treatment for diabetic foot ulcers. Lancet. 2005;36:1725-1735.
4. van Baal JG, Harding KG, Lipsky BA. Foot infections in diabetic patients: an overview of the problem. Clin Infect Dis. 2004;39:S71-S72.
5. Ulbrecht JS, Cavanagh PR, Caputo GM. Foot problems in diabetes: an overview. Clin Infect Dis. 2004;39:S73-S82.
6. Merlino JI, Malangoni MA. Complicated skin and soft-tissue infections: diagnostic approach and empiric treatment options. Cleve Clin J Med. 2007;74:S21-S28.
7. Lipsky BA. Medical treatment of diabetic foot infections. Clin Infect Dis. 2004;39:S104-S114.
8. Armstrong D, Lipsky BA. Diabetic foot infections: stepwise medical and surgical management. Int Wound J. 2004;1:123-132.
9. Lipsky BA, Berendt AR, Deery HG, et al. Diagnosis and treatment of diabetic foot infections. Clin Infect Dis. 2004;39:885-910.
10. Zgonis T, Roukis TS. A systematic approach to diabetic foot infections. Adv Ther. 2005;22:244-262.
11. Hilton JR, Williams DT, Beuker B, et al. Wound dressings in diabetic foot disease. Clin Infect Dis. 2004:39;S100-S103.
12. Weintrob AC, Sexton DJ. Overview of diabetic infections of the lower extremities. Waltham, MA: UpToDate; 2011.
13. Nicolau DP, Stein GE. Therapeutic options for diabetic foot infections: a review with an emphasis on tissue penetration characteristics. J Am Podiatr Med Assoc. 2010;100:52-63.
14. Ferry RJ Jr. Diabetic foot care. www.emedicinehealth.com/
To comment on this article, contact rdavidson@uspharmacist.com.





