US Pharm. 2022;47(3):38-42.
ABSTRACT: Preexposure prophylaxis (PrEP) is a medication therapy regimen provided to HIV-negative persons at high risk of acquiring HIV. Current FDA-approved oral therapies are emtricitabine-tenofovir disoproxil fumarate (FTC/TDF) and emtricitabine-tenofovir alafenamide (FTC/TAF). Recently, the FDA approved the first injectable therapy, cabotegravir long-acting injectable (CAB-LA), which could provide an advantage to certain patient populations. When PrEP therapy is taken consistently, it is very effective at preventing HIV. This article will discuss who is a candidate for PrEP, available therapy options, and additional therapies that are actively being studied. Pharmacists can help provide education to these patients to help promote adherence within high-risk populations.
In 2019, 36,801 individuals received a new diagnosis of HIV in the United States.1 Approximately 1,189,700 people over the age of 13 years are living with HIV infection, and up to 13% of these individuals may be undiagnosed.1 Occurrences are highest in men who have sex with men (MSM), Black, and Hispanic patients.1 At this time, there is no vaccine available to prevent HIV transmission. Nonpharmacologic and pharmacologic interventions are needed to successfully reduce HIV acquisition. Nonpharmacologic methods include, but are not limited to, counseling regarding safe sex practices, access to harm-reduction strategies such as syringe service programs to decrease sharing syringes, and circumcision in some situations.2 Pharmacologic methods for HIV prevention focus on postexposure prophylaxis (PEP), pre-exposure prophylaxis (PrEP), and treatment as prevention (TasP).3 PEP is starting antiretroviral therapy (ART) within 72 hours of potential exposure to HIV.3,4 PrEP is utilizing ART to prevent HIV infection in HIV-negative individuals who are at a greater risk of HIV.3,5 TasP is effective at preventing HIV transmission when the infected person takes ART as prescribed to reduce viral loads to nondetectable levels.3 The focus of this article will be to discuss who is a candidate for PrEP, available treatment options, recommended monitoring, and pipeline therapies.
The use and consistent adherence of PrEP have been estimated to reduce the occurrence of HIV by 99% for MSM, 90% or more for women, and 74% among individuals who inject drugs.5,6 Currently, there are two FDA-approved, once-daily oral tablet PrEP therapies available to prevent HIV infection in adolescents and adults weighing at least 35 kilograms: emtricitabine-tenofovir disoproxil fumarate (FTC/TDF) and emtricitabine-tenofovir alafenamide (FTC/TAF).5,7,8 FTC/TDF is recommended to prevent HIV infection in all individuals at risk through injection drug use or sex.7 In clinical trials, FTC/TDF showed a 92% to 100% risk reduction in acquiring HIV among adherent individuals.9,10 The DISCOVER trial showed that FTC/TAF was noninferior to FTC/TDF in reducing the risk of acquiring HIV among men and transgender women who have sex with men.11 FTC/TAF is FDA-approved to prevent HIV infection among individuals at risk through sex, excluding individuals at risk through receptive vaginal sex due to the lack of available clinical data.5,8
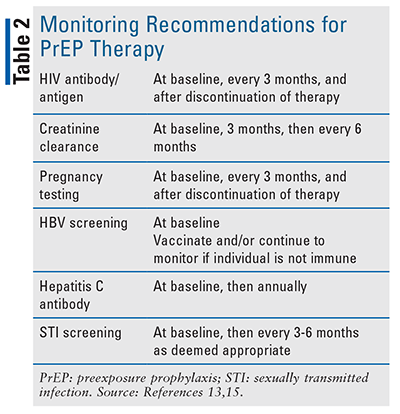
The CDC determined that more than 1.2 million individuals met eligibility criteria for PrEP in 2015.12,13 TABLE 1 provides indications for PrEP for patients at high risk of acquiring HIV.13 The U.S. Preventive Services Task Force has given PrEP a “grade A recommendation” that prescribers offer PrEP to patients at high risk of acquiring HIV.14 A specialization in infectious diseases or HIV medicine is not required for prescribing, which means primary care providers can prescribe PrEP.5 During the visit, a thorough past medical history should be conducted, and a complete medication review, risk assessment for acquiring HIV, risk assessment for drug or alcohol abuse, and resources and education should be provided to aid in reducing HIV acquisition.5,13 Prior to initiating either therapy, individuals should be tested and confirmed HIV negative to prevent resistant HIV. If an individual is HIV positive, additional ART is needed for effective management, which is not discussed in this article. Laboratory work includes a complete metabolic panel, liver enzymes, renal function, hepatitis serology, and screening for sexually transmitted infections (STIs).13,15 TABLE 2 provides recommendations for laboratory monitoring at baseline and for subsequent follow-up visits.13 Follow-up screening for STIs is based on an individual’s risk of STIs.15 Patients should follow up every 3 months for laboratory monitoring, medication adherence, and screening for medication toxicity.13

When selecting a regimen, shared decision-making with patient and provider is important. Providers should discuss with patients regarding the need for frequent visits and expenses related to necessary monitoring. Common adverse effects for FTC/TDF and FTC/TAF include nausea, gastrointestinal upset, and headache (which usually resolves after a few weeks).7,8 Clinicians may consider prescribing supportive therapy, such as antiemetics, to help with adverse effects. Changes in bone mineral density and renal function are associated with FTC/TDF and are usually reversible after a certain time period following discontinuation.7 FTC/TDF is appropriate for patients with renal function >60 mL/min while FTC/TAF is approved for ≥30 mL/min based on the Cockcroft-Gault equation.7,8 In patients who have difficulty swallowing larger tablets, FTC/TAF may be preferred given its smaller size than FTC/TDF. Prescribers and clinicians can consult the National Clinician Consultation Center PrEP Line at 1-855-448-7737 for additional prescribing information.5
Alternative Dosing Strategy
On-Demand Oral PrEP: The only FDA-approved, oral once-daily therapies are FTC/TDF and FTC/TAF. On-demand oral PrEP is another strategy prescribers are utilizing against the adherence barrier prevalent in many individuals’ PrEP therapy. Although on-demand PrEP—also known as “nondaily PrEP,” “event-driven PrEP,” or “2-1-1 PrEP”—is not FDA-approved or CDC-recommended, it has been studied and is currently being utilized in clinics worldwide.16 This approach replaces daily oral PrEP therapy with PrEP therapy at times of a sexual encounter. This means that a patient would take two tablets of oral PrEP 2 to 24 hours prior to sex, followed by a third tablet 24 hours after the initial double dose, and then a fourth tablet 48 hours after the initial double dose.15,16 In a multisex scenario, the patient would continue to take one tablet every 24 hours until 48 hours after the last sexual encounter.15,16 If there are fewer than 7 days between sexual episodes, the patient would need to take one tablet to restart the cycle. If there are more than 7 days between sexual episodes, the patient would need to start again with the two-PrEP-tablet cycle.15,16
The IPERGAY trial compared on-demand PrEP with placebo and found an 86% reduction rate of HIV infection in the on-demand PrEP group.17 Study limitations included the low participant population and the median number of doses (15 tablets per month); this is very close to the four tablets per week that has been known to produce high levels of protection during attempted daily dosing.17 This brings to light the question of whether the high rate of dosing was a main contributor to the HIV-acquisition reduction rate.17 Current guidelines for on-demand PrEP are conflicting, and more data will be needed before we have a clear understanding of the implications of on-demand dosing in PrEP therapy.
Recent FDA Approval
Cabotegravir Long-Acting Injectable (CAB-LA): CAB-LA was recently approved by the FDA in December 2021 for PrEP in at-risk adults and adolescents weighing at least 35 kilograms to reduce the risk of sexually acquired HIV.18 Prior to initiation and each subsequent injection, individuals must produce a negative HIV-1 test result.19 Shared decision-making with healthcare provider and patient should occur to determine the use of oral cabotegravir 30 mg once-daily lead-in for a month prior to administration of CAB-LA or to start CAB-LA without an oral lead-in.19
CAB-LA is a 600-mg injection given 1 month apart for 2 consecutive months and subsequent injections every 2 months thereafter administered by a healthcare provider via gluteal intramuscular injection. If an oral lead-in of cabotegravir is used, initiation injections should be administered on the last day or within 3 days of oral cabotegravir. CAB-LA can be administered 7 days before or after the scheduled date to receive the injection.19 If individuals miss subsequent injections, discussions with their healthcare provider are warranted to explore additional options. The most common adverse reactions with CAB-LA include injection site reactions, diarrhea, headache, fatigue, and pyrexia.19 Hepatoxicity has been reported with CAB-LA; regular monitoring of hepatic function is recommended, and if hepatotoxicity occurs, to discontinue use.19
CAB-LA was studied in two randomized, double-blind, controlled, multinational trials: HIV Prevention Trials Network (HPTN) 083 and HPTN 084. HPTN 083 compared CAB-LA to FTC/TDF. The primary outcome was the prevalence of HIV transmission in high-risk men who have sex with men and transgender women. Individuals were to be followed for 153 weeks and were randomly assigned to either CAB-LA or FTC/TDF. The study was stopped early due to the HIV infection rate being significantly lower in the CAB-LA group. In total, 52 of the 4,566 participants contracted HIV—39 participants in the FTC/TDF group and 13 in the CAB-LA group. This study was the first to show great promise for the use of an injection as an alternative to daily PrEP therapy.20
Furthermore, the HPTN 084 trial compared CAB-LA to FTC/TDF in adult women in sub-Saharan South Africa who were at high risk of being infected with HIV. This trial was also stopped early due to the HIV infection rate being significantly lower in the CAB-LA group. Thirty-eight of the 3,223 participants contracted HIV—34 participants in FTC/TDF group and four in the CAB-LA group.20 Primary analysis showed an 88% reduction in the risk of acquiring HIV-1 infection with CAB-LA.19
The availability of a long-acting injectable PrEP option will be an effective tool against current usage and adherence barriers in at-risk populations. Adjustments will need to be made in pharmacies and provider offices to establish workflow processes that are safe and efficient in billing and provide monthly and bi-monthly PrEP injections.
PrEP Therapies in the Pipeline
The protective effects of FTC/TDF and FTC/TAF rely on optimal adherence, which can be a barrier for at-risk patient populations. To combat this, clinical trials are in progress exploring alternative treatment options to daily oral PrEP therapy.
Islatravir (ISL) Once-Monthly Oral Tablet: Impower-022 and Impower-024 are both phase III trials comparing ISL with FTC/TDF and FTC/TAF in different patient populations. The primary end point will be the incidence of HIV infections. Both are estimated to be completed in 2024.22,23
Dapavirine Long-Acting Vaginal Ring (DPR-VR): DPR-VR is a long-acting vaginal ring system dosed once-monthly that is in clinical trials to offer PrEP therapy for women in developing countries who are at substantial risk of acquiring HIV.
Two separate phase III clinical trials showed that DPR-VR significantly reduced the risk of HIV transmission compared with placebo. The Ring Study showed a risk reduction of 31%, and the ASPIRE trial showed a risk reduction of 27%.24,25
DPR-VR received positive scientific opinion from the European Medicines Agency in 2020 for use in adult women in developing countries, recommendations from the World Health Organization (WHO) in 2021, and approval in Zimbabwe in July 2021.26,27 DPR-VR also has regulatory reviews pending in additional countries in Africa and is currently under review by the FDA.26
Although these results are promising and trials are still in process, approval of DPR-VR in developing countries would need to act as a supplement to existing HIV-prevention practices. The ring could be another expansion in our “PrEP toolbox” to fight the global rate of HIV infection.
Other delivery systems in the pipeline include additional topical products, implants, and transdermal devices. The goal of these new delivery systems is to expand the availability of PrEP and to appeal to a wide variety of populations globally, therefore increasing the percentage of eligible patients on PrEP therapy and ultimately decreasing the rate of HIV infections.28
Medication Affordability
The average cost of oral PrEP therapy is approximately $1,845 per month; cost can be a barrier to getting at-risk patients on therapy.15 There are several forms of assistance to help combat this barrier. Most health insurance and state-funded Medicaid plans cover PrEP therapy for little or no charge.29 To assist with high copays, Gilead, the manufacturer of FTC/TDF and FTC/TAF, offers copay cards to bring the patient’s cost down even further. For those who do not have insurance, most states have patient-assistance programs.29 Pending patient qualification, these programs assist in getting PrEP for free or at a reduced cost to the patient.
Many insurance companies require patients to use specialty pharmacies to fill these medications. Specialty pharmacies have resources dedicated to cost assistance and education for these medications. Physician offices that specialize in PrEP therapy can assist patients with these enrollment processes as well.
Role of the Pharmacist
Pharmacists can complete comprehensive medication reviews, identify drug interactions with PrEP therapy, and educate patients to notify prescribing clinicians when starting a new medication—whether it is an herbal supplement, OTC medication, or prescription therapy—to avoid potential interactions. Pharmacists can also provide counseling on PrEP therapy to discuss potential adverse effects, as well as management, importance of optimal adherence, and need for routine follow-up for monitoring. Pharmacists may be able to identify repeat PrEP patients as candidates for PrEP therapy and recommend follow-up with a prescriber. Additionally, pharmacists may be able to assist with resources available to make PrEP therapy more affordable, including co-pay cards and patient-assistance programs.
Impact of COVID-19
COVID-19 has impacted healthcare and individuals’ access to PrEP. Some offices may have been able to expand telehealth services to patients, while other offices may have reduced their hours or closed. The CDC continues to recommend quarterly HIV testing for patient safety and prefers laboratory-only visits for assessment.29 However, if laboratory-only visits are not available, there are two options: home specimen collection kits for HIV and STI infection to test urine or blood, where the patient mails the kit back to the laboratory and test results are provided to the prescribing provider, or self-testing via an oral swab–based test. The latter is used if other options are not available, given its lower sensitivity to detect recent HIV infection.30 Once HIV-negative status is confirmed, the prescriber could consider writing 90-day prescriptions instead of 30-day prescriptions. If a clinic is changing services or closing, it should provide individuals with other services or clinics for continuity of care and access to PrEP.30
Some clinics may have created flexible care plans to extend laboratory work and utilized telemedicine when in-person visits were not possible. One study showed a 44% reduction in new PrEP starts and a 0% decrease in follow-up visits for PrEP.31 Another study showed a 191% increase in PrEP refill lapses and a 72.1% decrease in new PrEP starts. Continued emerging data will show the impact of COVID-19 on PrEP therapy, and additional research is needed to assess the full impact of COVID-19 and PrEP therapy.32
Conclusion
PrEP is effective at reducing HIV transmission when used consistently. Pharmacists can provide education to patients to help continue therapy. There are new therapies in development that may further prevent HIV. COVID-19 continues to impact healthcare, and additional research will be needed to be conclude the impact on PrEP usage.
REFERENCES
1. CDC. HIV Surveillance Report. www.cdc.gov/hiv/statistics/overview/index.html. Accessed November 1, 2021.
2. CDC. HIV: Protect yourself if you inject drugs. www.cdc.gov/hiv/basics/hiv-prevention/inject-drugs.html. Accessed November 3, 2021.
3. Heendeniya A, Bogoch I. Antiretroviral medications for the prevention of HIV infection: a clinical approach to preexposure prophylaxis, postexposure prophylaxis, and treatment as prevention. Infect Dis Clin North Am. 2019;33(3):629-646.
4. CDC. HIV: About PEP. www.cdc.gov/hiv/basics/pep/about-pep.html. Accessed November 3, 2021.
5. CDC. HIV: Pre-exposure prophylaxis (PrEP) www.cdc.gov/hiv/clinicians/prevention/prep.html. Accessed November 1, 2021.
6. Beymer M, Holloway I, Pulsipher C, et al. Current and future PrEP medications and modalities: on demand, injectables, and topicals. Curr HIV/AIDS Rep. 2019;16(4):349-358.
7. Truvada (emtricitabine and tenofovir disoproxil fumarate) package insert. Foster City, CA: Gilead Sciences, Inc; Revised June 2020.
8. Descovy (emtricitabine and tenofovir alafenamide) package insert. Foster City, CA: Gilead Sciences, Inc; Revised March 2021.
9. Grand RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363(27):2587-2599.
10. Van Damme L, Corneli A, Ahmed K, et al. Preexposure prophylaxis for HIV infection among African women. N Engl J Med. 2012;367(5):411-422.
11. Hare C, Coll J, Ruane P, et al. The phase 3 DISCOVER study: daily F/TAF or F/TDF for HIV pre-exposure prophylaxis. Presented at the Conference on Retroviruses and Opportunistic Infections, Seattle, WA. March 2019.
12. Smith DK, Van Handel M, Wolitski RJ, et al. Vital signs: estimated percentages and numbers of adults with indications for preexposure prophylaxis to prevent HIV acquisition–United States, 2015. MMWR Morb Mortal Wkly Rep. 2015;65(46):1291-1295.
13. CDC. Preexposure prophylaxis for the prevention of HIV infection in the United States–2017 update: a clinical practice guideline. www.cdc.gov/hiv/pdf/risk/prep/cdc-hiv-prep-guidelines-2017.pdf. Accessed November 5, 2021.
14. U.S. Preventive Services Task Force. Preexposure prophylaxis for the prevention of HIV infection: US Preventive Services Task Force Recommendation Statement. JAMA. 2019;321(22):2203-2213.
15. Baker J, Rolls J. Update on HIV prevention and preexposure prophylaxis. JAAPA. 2020;33(6):12-17.
16. Saberi P, Scott H. On-demand oral pre-exposure prophylaxis with tenofovir/emtricitabine: what every clinician needs to know. J Gen Intern Med. 2020;35(4):1285-1288.
17. Beymer M, Holloway I, Pulsipher C, Landovitz RJ. Current and future PrEP medications and modalities: on demand, injectables, and topicals. Curr HIV/AIDS Rep. 2019;16(4):349-358.
18. FDA. FDA approves first injectable treatment for HIV preexposure prevention. December 20, 2021. www.fda.gov/news-events/press-announcements/fda-approves-first-injectable-treatment-hiv-pre-exposure-prevention. Accessed February 19, 2022.
19. Apretude (cabotegravir extended-release injectable suspension) package insert. Research Triangle Park, NC: GlaxoSmithKline. Published December 2021.
20. Landovitz RJ, Donnell D, Clement M, et al. Cabotegravir for HIV prevention in cisgender men and transgender women. N Engl J Med. 2021;385:595-608.
21. World Health Organization. Trial results reveal that long-acting injectable cabotegravir as PrEP is highly effective in preventing HIV acquisition in women. www.who.int/news/item/09-11-2020-trial-results-reveal-that-long-acting-injectable-cabotegravir-as-prep-is-highly-effective-in-preventing-hiv-acquisition-in-women. Accessed November 28, 2021.
22. Clinicaltrials.gov. Oral islatravir (MK-8591) once-monthly as preexposure prophylaxis (PrEP) in men and transgender women who have sex with men and are at high risk for HIV-1 infection (MK-8591-024) (Impower-024). www.clinicaltrials.gov/ct2/show/NCT04652700. Accessed November 27, 2021.
23. Clinicaltrials.gov. Oral ISL QM as PrEP in cisgender women at high risk for HIV-1 infection (MK-8591-022) (Impower-022). www.clinicaltrials.gov/ct2/show/NCT04644029. Accessed November 27, 2021.
24. Nel A, van Niekerk N, van Baelen B, et al. Safety, adherence, and HIV-1 seroconversion among women using the dapivirine vaginal ring (DREAM): an open-label, extension study. Lancet HIV. 2021;8(2):e77-e86.
25. Baeten J, Palanee-Phillips T, Brown E, et al. Use of a vaginal ring containing dapivirine for HIV-1 prevention in women. N Engl J Med. 2016;375:2121-2132.
26. International Partnership for Microbicides. A long-acting ring for women’s HIV prevention. www.ipmglobal.org/content/ring-backgrounder-0. Accessed November 28, 2021.
27. World Health Organization. Consolidated guidelines on HIV prevention, testing, treatment, service delivery and monitoring: recommendations for a public health approach. www.who.int/publications/i/item/9789240031593. Accessed November 28, 2021.
28. Coelho L, Torres T, Veloso V, et al. Preexposure prophylaxis 2.0: new drugs and technologies in the pipeline. Lancet HIV. 2019;6:e788-e799.
29. CDC. HIV: Paying for PrEP. www.cdc.gov/hiv/basics/prep/paying-for-prep/index.html. Accessed November 21, 2021.
30. CDC. PrEP during COVID-19. 2020. www.cdc.gov/nchhstp/dear_colleague/2020/dcl-051520-PrEP-during-COVID-19.html. Accessed December 1, 2021.
31. Rogers B, Tao J, Maynard M, et al. Characterizing the impact of COVID-19 on pre-exposure prophylaxis (PrEP) care. AIDS Behav. 2021;25:3754-3757.
32. Krakower DS, Solleveld P, Levine K, et al. Impact of COVID-19 on HIV preexposure prophylaxis care at a Boston community health center. NATAP. 2020. www.natap.org/2020/IAC/IAC_75.htm. Accessed December 1, 2021.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.





