US Pharm. 2016;41(5):27-30.
ABSTRACT: Depression in pediatric patients with type 1 diabetes (T1D) is a growing area of discussion. A diagnosis of T1D requires pediatric patients and their families to make drastic lifestyle changes. Improper coping skills can negatively affect the patient’s life and jeopardize overall health, as signaled by poor glycemic control and A1C readings. The mainstay of treatment for depression in pediatric patients with T1D is psychotherapy. Medication is warranted in certain cases, and currently only two agents have FDA indications for pediatric depression: fluoxetine and escitalopram. The use of antidepressants in pediatric patients with T1D requires thorough counseling by a pharmacist to ensure optimal outcomes.
The management of type 1 diabetes (T1D) in pediatric patients is a lifelong commitment that places a large burden on patients and their families. T1D forces patients to modify their daily schedule to accommodate new, complex, and demanding regimens. It can be difficult for young patients to fully grasp the importance of these changes. These alterations signal the need for behavioral changes and increased responsibility on the part of the entire family to ensure proper care.
One issue that can be overlooked is the mental health of the patient. It is important to address the patient’s emotional well-being because disconnects can make management increasingly problematic and lead to serious short-term and long-term complications.1 Research indicates that nearly one in seven youths with T1D meets the clinical cutoff for depression; this rate is nearly double that for youths without diabetes.2 Additionally, mothers of children with T1D have been shown to have increased levels of both depression and anxiety.3 Mental-health screenings are an important part of ongoing care. The implications of a diabetes diagnosis can negatively affect T1D patients’ quality of life.4 Diabetes-specific concerns include fear of hypoglycemia or hyperglycemia, anxiety, and disordered eating. Chronic psychological stress has been associated with higher A1C and can lead to difficulties in diabetes treatment.5
ETIOLOGY
The connection between depression and poor health outcomes in T1D patients is not fully understood. The chronicity of the disease, as well as the physical and emotional drain of disease management, places a great deal of stress on the patient. The SEARCH for Diabetes in Youth study demonstrated that the first 6 years following diagnosis are marked by a significant decline in glycemic control based on a change in A1C.6 The study authors concluded that the psychosocial burden, especially in a patient with poor diabetes-specific quality-of-life measures, is a major contributor to poor glycemic control. Overall, the prevalence of depression has a positive correlation with age.3
CLINICAL CHARACTERISTICS
Signs of depression can vary from patient to patient, but the theme of change is most prevalent. Characteristics include loss of interest in enjoyed activities, changes in sleep patterns, appetite changes, concentration problems, decreased energy, guilt, nervousness, and trouble in school. Some manifestations that could signal depression in T1D pediatric patients include a history of increasing A1C, inconsistent blood sugars, diabetic ketoacidosis, and recurrent hospital admissions.2,6 Typically, adolescent girls are at greater risk for depression than adolescent boys, but this does not always hold true.7
DIAGNOSIS
Since 2005, the American Diabetes Association (ADA) has recommended that patients receive mental-health screenings starting at 10 years of age and on a yearly basis thereafter.8 Depression should be assessed and diagnosed according to the criteria detailed in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Information should be gathered from various sources in addition to the initial patient interview. Parents or caregivers, social workers, and teachers could be interviewed in order to obtain a clearer picture. It is imperative to perform a suicide ideation/crisis evaluation during the initial interview. During the meeting, it is important to follow the ADA’s recommendations for the psychiatric care of T1D patients8:
• Family involvement: Confirm that there is developmentally appropriate parent/family involvement in the management of the patient’s diabetes care in order to avoid an early transfer of sole responsibility for diabetes management to the patient. (Recommendation Grade: B)
• Family conflict/stress: Ask openly about diabetes-related family conflict or stress and negotiate an acceptable resolution with the child or adolescent and the parent(s). If family conflict is deeply rooted and cannot be resolved by the diabetes team, the family should be referred to a mental-health specialist who is knowledgeable about T1D in youths and family functioning. (Recommendation Grade: C)
TREATMENT
The treatment of depression in the pediatric population is founded on the initiation of psychotherapy. Pharmacotherapy may be used if a patient meets certain criteria, which are discussed below.
Psychotherapy
Psychotherapy, also known as talk therapy or simply counseling, teaches patients and their families about the nature of depression and effective tools for dealing with the condition. This is the treatment of choice for mild-to-moderate depression. More severe depression should be treated with psychotherapy in combination with medication.9
Two evidence-based types of psychotherapy are used to treat depression in pediatric T1D patients: cognitive-behavioral therapy (CBT) and interpersonal therapy for adolescents (IPT-A). Counseling sessions may be conducted by a variety of methods, including individual, family, and group. Family-based psychotherapy correlates well with the psychosocial adaptation of diabetic adolescents and should be considered.7 Parents may be more aware of symptoms signaling problematic emotional functioning owing to the high level of involvement required for effective disease management.2 The length of time that counseling is conducted varies on a case-by-case basis but varies from 8 weeks to 20 weeks or more, and sessions are 30 to 50 minutes long. Improvement may be seen after the first couple of sessions, with the greatest benefit around the 8- to 10-week mark.10 The inclusion of a mental-health professional on the multidisciplinary team highlights the importance of attending to the psychosocial issues of diabetes.4 A patient’s psychotherapy is as integral to his or her overall health as diabetes management.
CBT: This form of therapy is based on the concept that thoughts influence behaviors and feelings, and vice versa. Treatment focuses on the patient’s thoughts and behaviors to reframe his or her perspective. CBT reduces feelings of hopelessness by increasing enjoyable activities, reducing negative thoughts, and improving assertiveness and problem-solving skills.5,9 These interventions should incorporate diabetes-related self-monitoring, lifestyle-related behaviors, and worry/stress management. Baseline improvements in A1C, well-being, diabetes-related stress, perceived stress, anxiety, and depression may occur and are especially noteworthy in patients who are poorly controlled.5
IPT-A: This therapy targets a patient’s interpersonal problems, which can cause or exacerbate depression in four distinct areas: grief, role disputes, role transitions, and interpersonal deficits.11 Problems are identified in order to improve problem-solving skills and change communication patterns. The purpose of IPT-A is to improve interpersonal functioning and the patient’s mood.9 IPT-A was adapted to address the developmental and interpersonal needs of adolescent patients and their families.12 In a randomized, controlled trial comparing IPT-A with an unspecified psychological school-based clinic, patients in the IPT-A arm fared much better than the other patients.11 Compared with supportive-counseling patients, IPT-A patients had fewer reported symptoms on the Hamilton Depression Rating Scale, increased functioning on the Global Assessment of Functioning scale, and improved social functioning on the Social Adjustment Scale Self-Report.11 Family participation in IPT-A is highly encouraged, but not mandatory. It is important to note that IPT-A has been approved only for patients aged 12 years and older, and it may not be an option for all patients.9
Pharmacotherapy
If psychotherapy is not feasible or the patient’s depression has not noticeably improved in 6 to 12 weeks, then medication may be added.9 Medications used to treat these patients are not without risks. In 2004, a black box warning was applied to all antidepressants regarding the increased risk of of suicidal thinking and behavior in young patients. The FDA mandated this warning to ensure that all providers are aware of the severity of this potential problem. A meta-analysis of more than 300 clinical trials determined that the rate of suicidal thinking or behavior was 4% in patients taking antidepressants versus 2% in those taking a placebo.13 None of the patients studied ultimately committed suicide.9 In 2007, the warning was expanded to state that depression itself was associated with an increased risk of suicide. The medications work by restoring the normal balance of chemicals in the brain. Selective serotonin reuptake inhibitors (SSRIs) are the class of choice, but atypical antidepressants and tricyclic antidepressants (TCAs) are sometimes used.
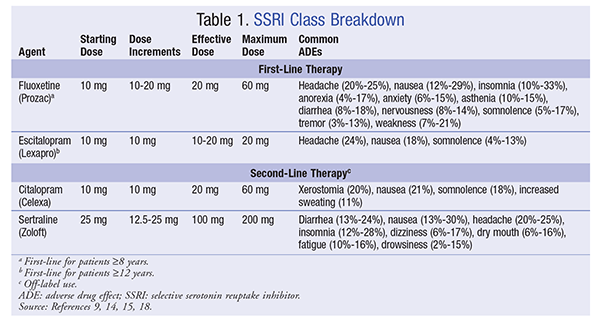
SSRIs: Fluoxetine (Prozac) and escitalopram (Lexapro) are the only agents that have received FDA approval, although the entire SSRI class is used. Fluoxetine is approved for major depressive disorder (MDD) in children aged 8 years and older initiated at 10 mg daily.14 Escitalopram is approved for the treatment of MDD in patients aged 12 years and older at a dosage of 10 mg daily. Age must be considered in choosing appropriate medication for these patients.15 This drug class can cause a wide range of adverse effects. Common adverse drug effects (ADEs) are headaches, gastrointestinal upset, and decreased blood pressure. Some side effects, including agitation, irritability, decreased sleep, and weight loss, warrant further investigation and may lead to decreasing or discontinuing the drug. These serious ADEs typically occur in fewer than 1% of patients taking SSRIs. TABLE 1 lists dosing parameters for the SSRI class.
Serotonin syndrome, a serious ADE associated with SSRIs, is caused by a sharp increase in serotonin levels in the body. Symptoms include hyperthermia, profuse sweating, agitation, and muscle rigidity. Serotonin syndrome may be caused by a high SSRI dose or by an SSRI taken in conjunction with other agents that have serotonergic properties, such as serotonin-norepinephrine reuptake inhibitors, monoamine oxidase inhibitors, triptans, tramadol, and dextromethorphan.16 Patients experiencing these symptoms should be admitted to the hospital for observation; those with severe symptoms may be admitted to the ICU.16 Symptoms typically resolve within 24 hours of discontinuing the serotonergic agent and initiating care. Treatment is based on five key principles16:
1. All serotonergic agents should be discontinued.
2. Sedation with benzodiazepines should be initiated to eliminate agitation, tremor, and elevations in heart rate and blood pressure. Doses should be titrated to the desired effect. Lorazepam 0.02 to 0.04 mg/kg/dose may be given to pediatric patients.
3. Supportive care should be initiated to normalize vital signs. This includes oxygen-saturation levels >94%, administration of IV fluids, and continuous cardiac monitoring.
4. If benzodiazepines and supportive care fail, a serotonin antagonist such as cyproheptadine should be initiated. Children aged 7 to 14 years may be given 4 mg every 6 hours, if needed (not to exceed 16 mg/day).
5. Serotonergic agents should be assessed once the symptoms have resolved to determine whether they should be restarted.
Atypical Antidepressants: This class may be considered if SSRIs are ineffective or not tolerated. Venlafaxine seems to be effective for depression in adolescents and works about as well as SSRIs, although it is associated with more ADEs.10 The off-label total daily dosage for MDD is 37.5 mg for children and 37.5 to 75 mg for adolescents; the dosage may be increased to 75 to 150 mg in children and 150 to 300 mg in adolescents. Common ADEs are headache, nausea, insomnia, dry mouth, and diaphoresis.17
TCAs: This class is rarely used and should be given to pediatric patients only if SSRIs and other alternatives are inadequate. TCAs have an extremely undesirable ADE profile, and they do not appear to be effective in children and younger adolescents. ADEs include anticholinergic effects such as dry mouth, urinary retention, constipation, and blurred vision; other ADEs are weight gain and rapid heartbeat.10
PHARMACIST INTERVENTION
Education About Medications
It is imperative that patients and caregivers be fully educated about any agents prescribed for depression. The black box warning associated with all antidepressants signals how important it is for the pharmacist to verify the patient’s understanding of his or her antidepressant. The patient should not abruptly discontinue the antidepressant, since doing so can cause serious negative effects, especially in association with the suicidality noted in the black box warning. If the dose is to be decreased or discontinued, it should be tapered slowly to prevent ADEs. It typically takes 6 to 12 weeks to see improvement, and the medication should be continued for 6 to 12 months.19
Pharmacists must be proactive when a patient taking an antidepressant presents with a new prescription or inquires about a nonprescription product. Pharmacists also must be vigilant for the possibility of serotonin syndrome. The occurrence of serotonin syndrome is more likely when a patient first starts an SSRI or when the SSRI dose is increased. The addition of another serotonergic drug increases a patient’s risk of serotonin syndrome. This potentially serious ADE should be discussed with the patient and/or caregiver when the prescription is originally filled and at every subsequent refill for the duration of treatment.
Patient Support
Patients and their parents may be more comfortable speaking to their pharmacist about the ongoing treatment of depression. See SIDEBAR 1 for some resources to which patients can be directed for more information. For a pharmacist to be an effective communicator and source of support, there are a few points that should discussed with patients and possibly their families9:
• Destigmatize the experience of being depressed. Express empathy in conversation and present yourself as a resource as well as an advocate for the patient’s health.
• Educate the patient and family about the origins, time course, and treatment options for depression. Emphasize that depression will not be resolved overnight, but rather will take time and commitment to overcome.
• Empower the patient and family to get the help they need. Stress the importance of seeking treatment and that adherence to both psychotherapy and pharmacotherapy is critical.

CONCLUSION
T1D is a major burden on both the pediatric patient and the family, especially if the patient is also experiencing depression. It has been well documented that this patient population is much more susceptible to the manifestations of depression compared with their non-T1D counterparts. As knowledgeable sources of information, pharmacists can play an integral role in the care of these pediatric patients with T1D and their families.
REFERENCES
1. Ducat L, Rubenstein A, Philipson LH, Anderson BJ. A review of the Mental Health Issues of Diabetes Conference. Diabetes Care. 2015;38:333-338.
2. Hood KK, Huestis S, Maher A, et al. Depressive symptoms in children and adolescents with type 1 diabetes: association with diabetes-specific characteristics. Diabetes Care. 2006;29:1389-1391.
3. Duru NS, Civilibal M, Elevli M. Quality of life and psychological screening in children with type 1 diabetes and their mothers. Exp Clin Endocrinol Diabetes. 2016;124:105-110.
4. American Diabetes Association. Children and adolescents. Diabetes Care. 2015;38(suppl 1):S70-S76.
5. Plack K, Herpertz S, Petrak F. Behavioral medicine interventions in diabetes. Curr Opin Psychiatry. 2010;23:131-138.
6. Hood KK, Beavers DP, Yi-Frazier J, et al. Psychological burden and glycemic control during the first 6 years of diabetes: results from the SEARCH for Diabetes in Youth study. J Adolesc Health. 2014;55:498-504.
7. Adal E, Önal Z, Ersen A, et al. Recognizing the psychosocial aspects of type 1 diabetes in adolescents. J Clin Res Pediatr Endocrinol. 2015;7:57-62.
8. Chiang JL, Kirkman MS, Laffel LM, et al. Type 1 diabetes through the life span: a positional statement of the American Diabetes Association. Diabetes Care. 2014;37:2034-2054.
9. Cheung AH, Zuckerbrot RA, Jensen PS, et al. Guidelines for Adolescent Depression in Primary Care (GLAD-PC): II. Treatment and ongoing management. Pediatrics. 2007;120:e1313-e1326.
10. Moreland CS, Bonin L. Patient information: depression treatment options for children and adolescents (Beyond the Basics). UptoDate. www.uptodate.com/contents/depression-treatment-options-for-children-and-adolescents-beyond-the-basics#H5. Accessed September 4, 2015.
11. Stein RE, Zitner LE, Jensen PS. Interventions for adolescent depression in primary care. Pediatrics. 2006;118:669-682.
12. Interpersonal Psychotherapy for Depressed Adolescents (IPT-A). Rockville, MD: Substance Abuse and Mental Health Services Administration; 2014.
13. Friedman RA. Antidepressants’ black-box warning—10 years later. N Engl J Med. 2014;371;18:1666-1668.
14. Prozac (fluoxetine) product information. Indianapolis, IN: Eli Lilly and Co; July 2014.
15. Karesh A. Pediatric focused safety review: escitalopram oxalate (Lexapro®). www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/PediatricAdvisoryCommittee/UCM255406.pdf. Accessed April 7, 2016.
16. Boyer EW. Serotonin syndrome (serotonin toxicity). UptoDate. www.uptodate.com/contents/serotonin-syndrome-serotonin-toxicity. Accessed April 7, 2016.
17. Medscape. Venlafaxine. http://reference.medscape.com/drug/effexor-venlafaxine-342963#0. Accessed September 14, 2015.
18. Halverson JL, Bhalla RN, Andrew LB. Depression medication. http://emedicine.medscape.com/article/286759-medication#2. Accessed September 22, 2015.
19. Kupfer DJ. Long-term treatment of depression. J Clin Psychiatry. 1991;52(suppl 5):28-34.
To comment on this article, contact rdavidson@uspharmacist.com.





