Something like this happens to nearly every pharmacist at one time or another during a professional career: Someone asks you what to do with drugs that are no longer being used. This issue is of little consequence for most noncontrolled drugs because record-keeping requirements are minimal. When patients brought back these medications, we used to flush them down the toilet. This is no longer politically correct or environmentally favored. However, it is a major headache for nearly all health care workers when it comes to the return or destruction of controlled substances. Affected most acutely are long-term care facilities, such as nursing homes, where controlled substances are left over or no longer necessary because a patient has passed away or transferred to another medical institution.
Imagine the following situation: A middle-aged woman shows up at your pharmacy with a half-filled bottle of Demerol and a label indicating it came from your pharmacy. Imagine further that she tells you her mother passed away and she wants to get rid of the drugs because there are young children and teenagers in the house. She has seen the commercials and print media advising parents that their medicine cabinets can be a source for the diversion of dangerous drugs by their own children.1
As a pharmacist, what do you do or say? What do you think the law should be about returning controlled substances? Wouldn’t it be better to let you take management of the unwanted drugs in a manner that permits you to efficiently and economically get rid of them safely?
Just about every state has a law banning the return of pharmaceuticals to a pharmacy inventory after the drugs have left the control of a pharmacist. That policy, however, does not address the more important issue of dealing with unwanted controlled drugs. In point of fact, several states have recently passed laws allowing or encouraging the donation of unused pharmaceutical drugs to health care facilities, nursing homes, and pharmacies.2 These efforts also generally do not address the return of controlled substances. In one state, a single pharmacy collected enough drugs from 41 individuals, both controlled and noncontrolled, to fill a 30-gallon barrel in just four hours.3 Try to imagine the quantity of unused, out-of-date, or unwanted drugs there are in the households of America. Is there not some professional role for pharmacists to address these issues in every community and institutional pharmacy across the nation?
The Controlled Substances Act
How to properly dispose of unused controlled drugs has been a gray area for the almost four decades since the Comprehensive Drug Abuse Prevention and Control Act of 1970 (most often referred to as the Controlled Substances Act or CSA) and its Drug Enforcement Administration (DEA)–promulgated regulations were enacted.4 The problem is exacerbated by the fact that none of the statutes or regulations makes any explicit provision for patients or caregivers to give their excess drugs to DEA-registered facilities or individuals. Unfortunately, the DEA has interpreted the total silence addressing this issue to mean that it is illegal for a DEA registrant to accept returned controlled substances from just about anybody.5
On July 9, 2008, the American Pharmacists Association (APhA) posted a notice on its Web site entitled “DEA to patients: Don’t return unused controlled substance prescription medications to pharmacy.”6 The subtitle is just as compelling: “No provisions in Controlled Substances Act or Code of Federal Regulations for pharmacy take back of controlled substances from patients.” There are two noteworthy exceptions. If the drug is recalled by the manufacturer or the pharmacy makes a dispensing error, the drugs can be returned to the pharmacy (but not to the pharmacy inventory).
Current law yields an unwieldy and seldom-used method to let patients return controlled drugs to the DEA directly or to local police agencies that have been authorized by the DEA to accept the medications. The direct DEA way of doing things like this is to have a patient or caregiver write a letter to the local DEA office addressed to the Special Agent in Charge.7 That DEA agent is supposed to authorize and instruct the applicant seeking the authorization to dispose of the controlled substance through any of the following methods: “1) Transfer of the substance to a person registered under the CSA and authorized to possess the substance; delivery to an agent of the Administration or to the nearest office of the Administration; 2) by destruction in the presence of an agent of the Administration or other authorized person; or 3) by such other means as the Special Agent in Charge may determine to ensure that the substance does not become available to unauthorized persons.”8
The DEA acknowledges that while this option is available, “it is used in extremely limited circumstances.”9 In another more recent development, the DEA has granted “temporary permission to law enforcement agencies that have requested authorization to accept for disposal controlled substances that have been dispensed to ultimate users. In granting such temporary authorization, the DEA has imposed certain conditions to ensure that the controlled substances do not become available to unauthorized persons… and to promote consistency with the structure of the CSA.”10 This, of course, could be problematic for individuals who, for a variety of reasons, might not relish the idea of talking to the police about where the controlled substances came from, who was taking them, for how long, or why. It should also be noted that the so-called “reverse distributors,” who are authorized to take back unused, unwanted, or out-of-date controlled substances from other DEA registrants, are not empowered to accept controlled substances from nonregistrants such as patients and caregivers.
Options for Proper Disposal
This, of course, creates quite the conundrum. If individuals can’t make a good faith effort to return unwanted controlled drugs without a lot of bureaucratic hassle, what should they do with them? Recall the news last year that traces of prescription drugs have been found in the drinking water of an estimated 41 million Americans.11 An Associated Press investigation showed the presence of such drugs as antibiotics, anticonvulsants, mood stabilizers, and sex hormones. In the course of a five-month inquiry, the investigators discovered that pharmaceuticals had been detected in the tap water of 24 major metropolitan areas, including Southern California, Northern New Jersey, Detroit, Michigan, and Louisville, Kentucky. That study did not specifically address the issue of whether controlled substances were detected. However, it does not present any intellectual hurdle to understand that if drugs are being flushed or poured down the drain, then there will certainly be some controlled drugs found in the mix.
If patients cannot bring drugs that are controlled substances to the pharmacy or other health care facility and, in our efforts to keep a clean environment, we must find another method of disposal that does not contaminate surface water, just what should we advise patients and caregivers to do with these “waste” products? A unique public-private partnership between the U.S. Fish and Wildlife Service, the APhA, and the Pharmaceutical Research and Manufacturers of America has established SMARXT DISPOSAL initiatives to address the question of what to do with waste drugs.12 This group of organizations has cited the FDA as the source for 13 drugs that can be flushed or poured down the drain (Table 1).
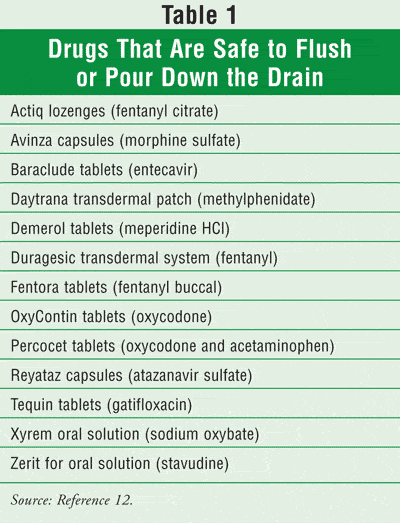
For drugs not on the list, whether or not they are controlled drugs, the SMARXT DISPOSAL coalition suggests putting unused drugs into the household trash with some precedent steps that include:
“a) Pour medication into a sealable plastic bag. If medication is a solid (pill, liquid capsule, etc.), crush it or add water to dissolve it.
b) Add kitty litter, sawdust, coffee grounds (or any material that mixes with the medication that makes it less appealing for pets and children to eat) to the plastic bag.
c) Seal the plastic bag and put it in the trash.
d) Remove and destroy ALL identifying personal information (prescription label) from all medication containers before recycling them or throwing them away.”12
The idea here is to prevent other adults, children, and pets or animals from consuming the drugs directly from the trash. You should also be aware that the coalition is recommending that people who have questions about these procedures consult a pharmacist.12
Proposed Rulemaking
On January 21, 2009, the DEA announced that it is seeking information and input from the public and stakeholders, including pharmacists, about what to do with controlled substances that are no longer being used.9 The DEA’s publication of this notice is in advance of any potential rulemaking. Instead, this notice asks patients (referred to in the law as “ultimate users”), state and local law enforcement agencies, publicly owned treatment works, concerned interest groups, long-term care facilities, hospices and in-home care groups, narcotic treatment programs, reverse distributors, state regulatory agencies, all interested parties, and pharmacies for input on the policies and procedures regarding taking care of no-longer-needed controlled substances. The document asks 117 questions directed at the various entities and individuals listed. The administration will accept comments and input until March 23, 2009. Typically, although not always, an “Advance Notice of Proposed Rulemaking” such as this one will be followed with the actual proposed rule, followed by a set time for public comment and, eventually, the final rule on the subject.
REFERENCES
1. Talking with kids about alcohol and drugs. www.talkingwithkids.org/drugs.
2. “Allowing or encouraging donation of unused pharmaceutical drugs is a popular idea—nine states passed such laws in 2006. For 2008 interest continues in 10 states, with some of the focus on cancer drug repository programs that allow an individual, health care facility or drug manufacturer to donate cancer drugs or supplies needed to administer cancer drugs for use by those who meet eligibility criteria.” 2008 Prescription Drug State Legislation. National Conference of State Legislatures. Updated December 22, 2008. www.ncsl.org/programs/health/
3. Kocian L. Getting rid of what the doctor ordered. Proper disposal of unwanted medications is daunting, costly. The Boston Globe. June 15, 2008. www.boston.com/news/local/
4. 21 USC §§801-971. These references also include the Controlled Substances Import and Export Act that was adopted as an amendment to the original CSA. The regulations are indexed at 21 CFR §§ 1300 to 1399.
5. At the risk of sounding like a grammatical snob, the DEA should have used the word “unlawful” instead of “illegal.” Grammar Girl. http://grammar.
6. DEA to patients: Don’t return unused controlled substance prescription medications to pharmacy. American Pharmacists Association. www.pharmacist.com/AM/
7. 21 CFR § 1307.21(a)(2).
8. 21 CFR § 1307.21(b).
9. Disposal of controlled substances by persons not registered with the Drug Enforcement Administration. Docket No. DEA-316. Published January 21, 2009. www.regulations.gov/
10. 21 CFR § 1307.21.
11. Donn J, Mendoza M, Pritchard J. AP: Drugs found in drinking water. USA Today. September 12, 2008. www.usatoday.com/news/
nation/2008-03-10-drugs-tap-
12. Responsible medication disposal safeguards lives and protects the environment. www.smarxtdisposal.net/. Accessed February 4, 2009.
To comment on this article, contact rdavidson@jobson.com.





