US Pharm.
2007;1:42-55.
Vitamins
and minerals serve crucial functions in almost all bodily processes and must
be obtained from foods or supplements, as our bodies are unable to make them.
According to results of the National Health and Nutrition Examination Survey,
52% of adults take dietary supplements.1 Supplement use is
typically a safe and effective method of maintaining a healthy body; however,
supplements have the potential to interact with prescribed medications. In
this article, common drug interactions with vitamins and minerals are
discussed, along with suggestions on how the pharmacist should manage these
interactions.
It is important for
pharmacists to be able to identify patients who are most at risk. Risk factors
for poor outcomes from drug interactions include use of multiple medications
and/or supplements, older age, inadequate kidney or liver function, and the
use of medications with narrow therapeutic indexes. Patients with these risk
factors should be targeted for interventions to prevent drug interactions with
vitamins and minerals.
When managing interactions
between medications and vitamin/mineral supplements, the pharmacist has
several factors to keep in mind. First, he or she needs to determine the need
for the supplement during the time the medication is to be taken. For a short
course of medication, it is usually possible to discontinue the supplement
until the therapy is completed. Second, the pharmacist should identify
alternatives. For example, if the interacting substance comes from an antacid
that the patient needs to treat symptoms of gastroesophageal reflux disease,
an alternative such as an H2-blocker or proton pump inhibitor can
be used during the course of the medication. If the affected medication is to
be taken chronically, and the supplement is deemed necessary, the pharmacist
should work with the patient and the prescriber to suggest alternatives that
would limit exposure to potentially dangerous drug interactions.
Vitamins and Drug
Interactions
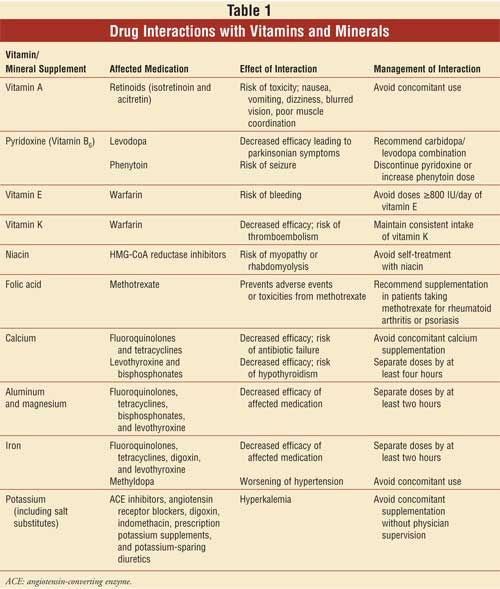
Vitamin A:
Vitamin A is a fat-soluble vitamin found in everyday foods, including liver,
yellow-orange fruits and vegetables such as carrots, margarine, milk, and dark
green, leafy vegetables such as spinach. It is not commonly available as a
supplement on its own; however, it can be found as a component of
multivitamins and combination supplements targeted towards improving skin,
hair, and nails. Vitamin A plays an important role in vision, bone growth,
cell differentiation, and the immune system. Vitamin A deficiency often leads
to problems with eyesight, a situation less common in the United States than
in underdeveloped countries where nutrition may be poor. Some conditions such
as celiac disease, Crohn's disease, and pancreatic disorders may lead to
malabsorption of vitamin A from the diet.
Interactions with vitamin A
become a concern with the use of products classified as retinoids--compounds
that are chemically similar to vitamin A. Retinoids such as isotretinoin
(Accutane) and acitretin (Soriatane) are indicated for the treatment of acne
and psoriasis, respectively. When retinoid products are prescribed, there is
concern of vitamin A toxicity.2 Pharmacists should alert patients
who are taking retinoid products to the importance of avoiding excess vitamin
A. They should also educate patients about the signs and symptoms of vitamin A
toxicity, such as nausea, vomiting, dizziness, blurred vision, and poor muscle
coordination.
Vitamin B6
(Pyridoxine): Vitamin
B6, or pyridoxine, is a water-soluble vitamin used to treat
deficiencies in vitamin B6 and some types of anemia. Foods rich in
pyridoxine include meats, whole grains, and certain fruits and vegetables.
Pyridoxine has been shown to decrease the effects of both phenytoin and
levodopa. However, this effect is not seen when levodopa is prescribed in
combination with carbidopa, which prevents the interaction from occurring. In
the rare instance that patients are taking levodopa in absence of carbidopa,
pharmacists should advise their patients to avoid any products containing
pyridoxine, because even smaller doses, such as 10 to 25 mg of pyridoxine, may
be enough to inhibit levodopa.3 Pharmacists should strongly
encourage the switch to a levodopa/carbidopa combination product if a patient
is not already taking this combination therapy.
There is limited evidence that
high doses of pyridoxine reduce phenytoin serum concentrations, thereby
reducing the efficacy of phenytoin. One study of patients with seizure
disorders found an association between pyridoxine 200 mg/day and reductions in
phenytoin concentration of nearly 50%.4 Effects of pyridoxine doses
lower than 200 mg/day on phenytoin serum concentration have not been
established; however, lower doses can be considered if someone taking a
multivitamin presents with low phenytoin serum concentrations. In these
instances, it may be necessary to discontinue or lower the dosage of the
multivitamin or to increase the dosage of phenytoin.
Vitamin E:
Vitamin E is a fat-soluble vitamin that is used in an array of ailments,
including vitamin E deficiency, atherosclerosis, Alzheimer's disease, and
various cancers. It is also a common supplement taken by patients with
cardiovascular disease. Case reports have documented an increased risk of
bleeding in patients taking vitamin E and warfarin concomitantly. This adverse
effect is more likely to occur with larger doses of vitamin E (>800 IU) rather
than smaller doses found in multivitamins.5,6 Therefore,
pharmacists should advise patients taking warfarin to take a multivitamin for
their daily source of vitamin E rather than a supplement containing only
vitamin E.
There is controversy over
vitamin E and other antioxidants with respect to chemotherapy. A theoretical
interaction has been proposed whereby antioxidants interfere with the
oxidative mechanism of the chemotherapeutic agent, thereby reducing its
effectiveness. The clinical implications of this interaction is still unknown;
but it might be worthwhile, until more is known, to counsel patients about
avoiding antioxidant supplements while undergoing chemotherapy treatments that
rely on this mechanism. However, it is important to note that antioxidants are
sometimes used to prevent or lessen the toxic effects of specific agents.
Patients on chemotherapy should be counseled not to supplement on their own
and to inform their oncologist of all dietary supplements and alternative
therapies.7
Vitamin K:
Vitamin K is indicated to reverse supratherapeutic international normalized
ratio (INR) caused by warfarin. When warfarin and vitamin K–containing
products are taken together, the activity of warfarin is decreased and results
in a decreased prothrombin time and INR.8 This puts the patient at
risk for suboptimal anticoagulation, possibly leading to thromboembolic events
such as deep venous thrombosis, pulmonary embolism, myocardial infarction, or
stroke. Vitamin K is obtained from the diet through green, leafy vegetables
such as spinach and broccoli. Pharmacists should advise all patients taking
warfarin to eat consistent diets of green, leafy vegetables and avoid
inconsistent use of supplements that contain vitamin K (i.e., patients should
not routinely change their intake of vitamin K–containing supplements).
Niacin:
Niacin is a B-complex vitamin used for the treatment of hyperlipidemia and
pellagra. Patients may try to self-treat high cholesterol with a niacin
supplement. The combination of niacin and HMG-CoA reductase inhibitors
(statins) may increase the risk of myopathies or rhabdomyolysis.9,10
Pharmacists are likely to encounter this interaction, since statins are among
the most commonly prescribed medications. The use of niacin with statins
should be recommended only if the benefits of lipid-lowering outweigh the
risks of myopathies and rhabdomyolysis. Typically, the interaction occurs at
doses of 1 g/day or greater of niacin. Generally, over-the-counter niacin
supplements are not supplied in doses this high. Pharmacists should encourage
patients to take niacin supplements only under the supervision of a physician.
Folic Acid:
Folic acid is a B-complex vitamin used to treat and prevent folic acid
deficiency. Folic acid supplementation is commonly recommended during
methotrexate therapy as prophylaxis against toxicities in patients with
rheumatoid arthritis and psoriasis. Folic acid deficiency is common in these
patients, since methotrexate inhibits dihydrofolate reductase (an enzyme that
reduces dihydrofolic acid to tetrahydrofolic acid).11 Once
metabolized to tetrahydrofolate, folic acid aids in multiple biochemical
processes to synthesize DNA, RNA, and various proteins. Studies have shown
that folic acid supplementation reduces toxicities of methotrexate without
affecting efficacy in long-term, low-dose methotrexate therapy for rheumatoid
arthritis or psoriasis. Pharmacists should recommend folic acid
supplementation in patients prescribed methotrexate for rheumatoid arthritis
or psoriasis, especially if adverse effects or toxicities, such as
abnormalities in blood cell counts and varying degrees of mucositis and
diarrhea, are present. However, it is important to note that some evidence
suggests that folic acid reduces the efficacy of methotrexate in cancer
therapy.12
Folic acid has also been
reported to decrease the efficacy of phenytoin, but only at doses 5 mg/day or
greater, which would be an unlikely dose for supplementation with
over-the-counter products.13
Minerals and Drug
Interactions
Calcium:
Calcium is a mineral supplement taken primarily to prevent or treat
osteoporosis. It is found in dairy products and is available as a supplement
or as a component of some antacids, such as Tums. One study determined that
67% of women and 25% of men take a calcium supplement regularly; because
calcium is associated with significant drug interactions, pharmacists should
question their patients about calcium intake.14
Calcium interacts with
prescription medications by limiting their absorption through chelation.
Significant interactions have been observed between calcium and certain
antibiotics--namely tetracyclines and fluoroquinolones. Calcium carbonate can
reduce the bioavailability of ciprofloxacin by 40%, which could result in
inadequate infection treatment and increased complications.15
Patients taking tetracyclines or fluoroquinolones should avoid the supplement
altogether during the course of therapy, which is usually a short period of
time.16,17
Typically, pharmacists will
instruct patients to space the doses of medications and/or supplements whose
absorption is affected by binding. Controversy, however, exists on how much
time is long enough to wait between doses. A minimum of two hours is usually
mandated, with some sources citing four to six hours as a minimum
recommendation. For example, it is recommended that doses of calcium and
levothyroxine be separated by at least four hours, because the former
decreases the bioavailability of the latter.18
In addition, it is important
to note that several medications, when used chronically, can affect the body's
utilization of calcium. Corticosteroids decrease the absorption of calcium,
which, over time, can lead to osteoporosis. Loop diuretics increase the
excretion of calcium, while drugs that affect vitamin D (which promotes of
calcium absorption), such as phenytoin, phenobarbital, and orlistat, may
decrease the amount of calcium absorbed from the diet.19 Patients
taking these medications could benefit from a calcium supplement, especially
one containing vitamin D. Pharmacists are in a key position to speak with
patients taking these medications and to recommend adequate calcium intake and
supplementation, particularly for patients with other risk factors for
osteoporosis.
Aluminum and Magnesium:
Aluminum and magnesium are unlikely to be used solely as supplements;
however, they are found in common over-the-counter antacid products. Like
calcium, they can bind to vulnerable medications, decreasing their
bioavailability and lessening their efficacy. Fluoroquinolone and tetracycline
antibiotics, bisphosphonates, and levothyroxine may be affected by aluminum
and magnesium; therefore, doses of these drugs should not be taken within two
hours of aluminum or magnesium consumption. If the patient is not responding
to therapy as expected, the pharmacist should recommend that the aluminum or
magnesium product be discontinued and an alternative identified.
Iron:
Iron supplements are needed if the body cannot produce a sufficient amount of
red blood cells. Lack of iron may lead to tiredness, shortness of breath,
decreases in physical performance, learning problems, and an increased risk of
infection.
Patients taking iron
supplements or multivitamins that contain iron should be instructed to avoid
taking their supplement within two hours of a dose of tetracycline or
fluoroquinolone antibiotics, digoxin, or levothyroxine.20,21 In
addition, patients taking both calcium and iron supplements should be informed
that because these supplements compete for absorption, they should be dosed at
different times of the day.
Iron salts can also interfere
with the absorption of levodopa, decreasing peak levels by 55% and area under
the curve by 51%.20 If the patient is experiencing breakthrough
parkinsonian symptoms, iron should be avoided. If this is not possible, the
dose of the levodopa should be increased. Iron can also cause worsening of
hypertension in patients taking methyldopa, and concomitant administration is
not recommended.22
The absorption of iron can be
affected by gastric acidity, and a fair amount of evidence exists to support
limited iron absorption in patients taking proton pump inhibitors and
medications that decrease gastric acidity.23-26 In iron-deficient
patients who require proton pump inhibitors, intravenous administration of
iron is recommended. Interactions between iron and omeprazole, which was
recently switched to over-the-counter status, may not be easily identifiable;
therefore, pharmacists should question patients about their consumption of
omeprazole and iron supplements.
Potassium:
Although most patients taking potassium supplementation receive this mineral
in the form of a prescription product, some over-the-counter products contain
potassium. Any medication that increases potassium levels in the body has the
potential to interact with supplemental potassium. Patients should use caution
when taking extra potassium if they take any of the following prescription
medications: angiotensin-converting enzyme inhibitors or angiotensin receptor
blockers, digoxin, indomethacin, prescription potassium supplements, and the
potassium-sparing diuretics triamterene or spironolactone. While the
amount of potassium found in over-the-counter vitamin and mineral supplements
is unlikely to cause major interactions, the pharmacist should warn patients
of the potential for interaction, particularly if the patient is at risk for
renal insufficiency. When counseling patients about the importance of avoiding
excess potassium, pharmacists should mention that most common salt substitutes
available in supermarkets contain potassium; therefore, these products should
be avoided in patients at risk for hyperkalemia. Salt substitutes contain
significantly higher amounts of potassium than do combination vitamin/mineral
supplements sold over-the-counter. For example, a 1/4-teaspoon serving of
NoSalt provides 650 mg of potassium. Considering that a 20-mEq prescription
tablet contains 780 mg of potassium, a patient can easily accumulate potassium
if using a salt substitute and thus should be warned against consuming these
products if taking medications that retain potassium.
Conclusion
There are many
different types of drug interactions with vitamins and minerals, ranging in
severity and significance. Patients may not think to share information with
their pharmacist about the vitamins and minerals they take, or they may feel
the substances are harmless and irrelevant to their medication regimen.
Because of the likelihood of an interaction, pharmacists should question
patients not only about the drug allergies they have but also about the
vitamins and minerals they ingest daily. Although this article did not discuss
herbal products and other nutraceuticals, use of these products is important
to document as well. Without this information, pharmacists cannot provide the
necessary screening for interactions. Information about the use of vitamins,
minerals, herbal products, and other nutraceuticals should be documented in
patients' records for future reference. In addition, pharmacists should
encourage software vendors and employers to provide fields in their profile
systems for over-the-counter medications and supplements, since these products
can impact care and cause easily avoidable drug interactions that could put
the patient at risk for poor outcomes or adverse effects.
REFERENCES
1. Radimer K, Bindewald B, Hughes J, et al. Dietary supplement use by US adults: data from the National Health and Nutrition Examination Survey, 1999–2000. Am J Epidemiol. 2004;160:339-349.
2. Dietary Supplement Fact Sheet: Vitamin A and Carotenoids. Bethesda, MD: National Institutes of Health: Office of Dietary Supplements; 2005.
3. Leon AS, Spiegel HE, Thomas G, Abrams WB. Pyridoxine antagonism of levodopa in parkinsonism. JAMA. 1971;218:1924-1927.
4. Hansson O, Sillanpaa M. Letter: Pyridoxine and serum concentrations of phenytoin and phenobarbitone. Lancet . 1976;1:256.
5. Corrigan JJ Jr, Marcus FI. Coagulopathy associated with vitamin E ingestion. JAMA. 1974;230:1300-1301.
6. Schrogie JJ. Coagulopathy and fat-soluble vitamins. JAMA. 1975;232:19.
7. Conklin KA. Dietary antioxidants during cancer chemotherapy: impact on chemotherapeutic effectiveness and development of side effects. Nutr Cancer. 2000;37(1):1-18.
8. Greenblatt DJ, von Moltke LL. Interaction of warfarin with drugs, natural substances, and foods. J Clin Pharmacol. 2005;45:127-132.
9. Cooke HM. Lovastatin- and niacin-induced rhabdomyolysis. Hosp Pharm. 1994;29:33-34.
10. Malloy MJ, Kane JP, Kunitake ST, Tun P. Complementarity of colestipol, niacin, and lovastatin in treatment of severe familial hyper cholesterolemia. Ann Intern Med. 1987;107:616-623.
11. Endresen GK, Husby G. Folate supplementation during methotrexate treatment of patients with rheumatoid arthritis. An update and proposals for guidelines. Scand J Rheumatol. 2001;30:129-134.
12. Drugs & Supplements. Folate (folic acid). Bethesda, MD: National Library of Medicine (US); 2005.
13. MacCosbe PE, Toomey K. Interaction of phenytoin and folic acid. Clin Pharm. 1983;2:362-369.
14. Dawson-Hughes B, Harris SS, Dallal GE, et al. Calcium supplement and bone medication use in a US Medicare health maintenance organization. Osteoporos Int. 2002;13:657-662.
15. Frost RW, Lettieri JT, Noe A, et al. Effect of aluminum hydroxide and calcium carbonate antacids on ciprofloxacin bioavailability. Clin Pharmacol Ther. 1989a;45:165.
16. Product Information: Vibramycin, doxycycline. Pfizer Laboratories, New York, NY, 1990.
17. Product Information: Cipro, ciprofloxacin. Bayer Corporation, West Haven, CT, 2002.
18. Product Information: Synthroid, levothyroxine. Oral tablet, USP, levothyroxine sodium oral tablet, USP. Abbott Laboratories, North Chicago, IL, 2002.
19. Greenberger NJ. Absorption of tetracyclines: interference by iron. Ann Intern Med. 1971;74:792-793.
20. Jellin JM, Batz F, Hitchens K. Pharmacist's Letter/Prescriber's Letter Natural Medicines Comprehensive Database. 2nd ed. Stockton, CA: Therapeutic Research Faculty; 1999:1241-1249.
21. Campbell NR, Hasinoff BB, Stalts H, et al. Ferrous sulfate reduces thyroxine efficacy in patients with hypothyroidism. Ann Intern Med. 1992;117:1010-1013.
22. Campbell NR, Hasinoff B. Ferrous sulfate reduces levodopa bioavailability: chelation as a possible mechanism. Clin Pharmacol Ther. 1989;45:220-225.
23. Campbell N, Paddock V, Sundaram R. Alteration of methyldopa absorption, metabolism, and blood pressure control caused by ferrous sulfate and ferrous gluconate. Clin Pharmacol Ther. 1988;43:381-386.
24. Product Information: Prilosec, omeprazole. Astra Merck Inc., Wayne, PA, 1995.
25. Sharma VR, Brannon MA, Carloss EA. Effect of omeprazole on oral iron replacement in patients with iron deficiency anemia. South Med J. 2004;97:887-889.
26. Product Information: Prevacid,
lansoprazole. TAP Pharmaceuticals, Lake Forest, IL, 2002.
To comment on this article, contact
editor@uspharmacist.com.






