US Pharm. 2018;43(3):16-20.
ABSTRACT: The United States is experiencing a growing opioid epidemic. Deaths from overdoses of prescription pain relievers, especially opioids, have risen dramatically over the last few years. Naloxone has been available for almost 50 years as an antidote to opioid overdose, and it has taken on an increased role in saving lives. A variety of dosage forms, including a nasal spray and a self-injector that verbally instructs the user about proper administration, are available. Every state has enacted new laws to increase naloxone availability. Pharmacists can monitor patient use, counsel patients about the addiction potential of opioids, and make naloxone more accessible to opioid users and caregivers in their practice.
According to the President’s Commission on Combating Drug Addiction and the Opioid Crisis, an epidemic is sweeping the United States.1 In order to address the opioid crisis, President Donald Trump declared a nationwide public-health emergency on October 26, 2017.2 In 2015, the CDC reported 33,091 deaths from opioids and estimated that more than 60,000 deaths would occur in 2017.3 The annual cost of prescription opioid abuse was estimated at $78 billion for 2013.4 The epidemic is fueled, in part, by experimentation with drugs of abuse, as well as the belief that drugs can alleviate pain and suffering. In addition, many substance abusers attempt to treat symptoms such as those seen in anxiety, depression, bipolar disorder, and other co-occurring psychiatric disorders.5
The number of opioid prescriptions, in terms of morphine milligram equivalents (MME), peaked in 2010 but has been declining.6 In 2012 alone, 259 million prescriptions were written for opioids in the U.S., translating to at least one opioid prescription for every adult.7 The number of prescriptions has contributed to the opioid public-health emergency. In the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, opioid-use disorder is defined as “a problematic pattern of opioid use leading to clinically significant impairment or distress.”8 Individuals who have an opioid-use disorder are 40 times more likely than those without it to turn to heroin.9 These statistics are alarming and should serve as a warning. Over the past couple of years, prescribing guidelines and prescription drug monitoring programs (PDMPs) have been established in an effort to combat the opioid epidemic.10,11 Increased access to naloxone has helped reduce the number of deaths.12
Pathogenesis
Opioids produce their effects on the body through three different opioid receptors: mu, kappa, and delta. Mu receptors are divided into the subtypes mu1 and mu2. These receptors, which are located primarily in the brainstem and medial thalamus, produce different effects. The mu1 receptor is mainly responsible for analgesia, euphoria, and serenity, whereas the mu2 receptor is involved in respiratory depression, pruritus, dependence, sedation, and prolactin release.13 Kappa receptors are located in the brainstem, spinal cord, limbic system, and other diencephalic areas and are responsible for spinal analgesia, sedation, dyspnea, dependence, dysphoria, and respiratory depression.13 Delta receptors, which are found mostly in the brain, have analgesic effects.13
When opioids bind to receptors in the limbic system, feelings of pleasure, euphoria, and relaxation occur in response to the release of dopamine. When an opioid is bound to receptors in the brainstem, autonomic responses such as breathing may be depressed. Opioid receptors in the spinal cord inhibit the transduction of signals to the brain, and, when activated, will decrease the feeling of pain even after an injury.14 These responses depend on both the type and the quantity of opioid in the body. Morphine is the gold standard for dosing; all other opioids are based on MME. Calculating the MME allows for improved patient monitoring to prevent possible overdoses and death.
Opioid Overdose
After taking an opioid for an extended period of time, an individual becomes tolerant to most of its effects, such as euphoria. Over time, the euphoria the individual experiences after opioid use becomes blunted, and greater doses are required to achieve the original effect. According to the CDC, a person taking >50 MME of an opioid per day is twice as likely as someone taking <20 MME per day to overdose. To put this into perspective, 50 MME equal 50 mg of hydrocodone, 33 mg of oxycodone, or 12 mg of methadone.15 See TABLE 1 for risk factors for opioid overdose.

Opioids depress the central nervous system and the respiratory centers located in the brainstem. Symptoms of an overdose may include a bluish hue to the fingertips and/or lips, snoring, gasping, gurgling, slow and/or shallow respirations of <12 per minute, or no respirations at all.16 During respiratory depression, oxygen is not delivered to the extremities, and organ systems start to shut down. The brain requires a constant flow of oxygenated blood to function, and it can be permanently damaged after 4 to 5 minutes of oxygen deprivation. The hallmark signs of an opioid overdose are miosis (pinpoint pupils), respiratory depression, and unconsciousness, a combination known as the overdose triad.17 Miosis occurs when stimulation of the parasympathetic nervous system causes the pupils of the eye to constrict. Use of other substances, such as methamphetamine and marijuana, may cause the pupils to dilate instead.18 In a suspected overdose, looking at the individual’s pupils can help determine whether immediate medical intervention is necessary.
Naloxone
The FDA approved naloxone hydrochloride as an antidote for opioid overdose in 1971; the drug is available for IV, SC, and IM injection, as well as intranasal administration. Currently, eight companies are marketing six FDA-approved formulations of naloxone products.19 In 1996, a variety of organizations began training laypeople in naloxone administration and distributing kits to combat opioid overdoses. From 1996 to 2014, more than 26,000 overdoses were reversed through prompt administration of naloxone.12 In April 2014, Evzio (naloxone injection), a prefilled auto-injector, was approved. The device incorporates voice and visual guidance to enable immediate intervention and foster confidence in the person administering the injection during an overdose emergency. Narcan became the first FDA-approved nasal spray in November 2015. Another option is the use of a prefilled syringe with an atomizer, which enables intranasal administration, although this is an off-label use. See TABLE 2 for a comparison of the different naloxone products.
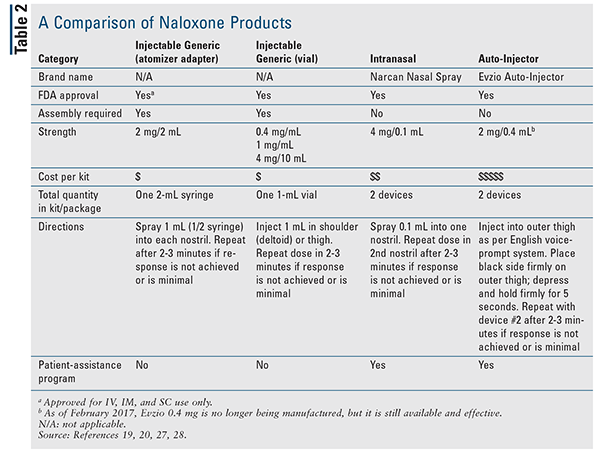
Naloxone is a competitive opioid antagonist that competes with opioids at the mu receptors. Effects such as respiratory depression, sedation, and hypotension caused by an overdose are temporarily reversed upon administration. Depending on the concentration of the opioid in the bloodstream, multiple doses of naloxone may be necessary to fully reverse the effects of an overdose. After the first dose is administered, emergency services should be contacted. The patient must be constantly monitored to assess whether a second dose is needed. A second dose is necessary if, after 2 to 3 minutes, the desired effect is not observed. Naloxone is an effective tool to counter respiratory depression until emergency personnel arrive.20
Laws Concerning Naloxone Access
As of July 2017, laws have been passed in all 50 states and the District of Columbia to increase public access to naloxone.21 Even given this expanded access, naloxone remains underutilized in overdose situations. Many first responders have access to naloxone and have been trained to administer it in an emergency. Unfortunately, many overdoses occur in places where immediate access to naloxone is unavailable and first responders cannot reach the patient in time. As stated previously, brain damage occurs within 4 to 5 minutes; however, it takes an average of 9.4 minutes for first responders to arrive at an emergency.22 Naloxone-access laws vary significantly from state to state.1
Standing orders are used in more than half of all U.S. states. In some states, a statewide standing order allows the state medical director to issue a written order allowing the distribution of naloxone by designated healthcare professionals, including pharmacists.23 Pharmacist prescriptive authority permits pharmacists to initiate prescriptions for naloxone. A collaborative-practice agreement is a formal relationship allowing pharmacists to provide naloxone directly to patients on behalf of a prescriber. As with any medication, a prescriber may issue a prescription directly to a patient or caregiver. In at least one state (Nebraska), naloxone is considered a nonprescription medication.24
Good Samaritan laws have been established in 40 states and the District of Columbia that provide protection for persons reporting overdoses in good faith. The scope of offenses and violations that are protected by immunity provisions differs from state to state.21,25
Pharmacist Involvement
Pharmacists can educate patients, family members, or caregivers about the proper use of opioids, including the signs and symptoms of an opioid overdose. This education may be a lifesaving intervention. Because of their accessibility, pharmacists are in a position to assess whether a patient is at risk for opioid overdose and tailor counseling to meet the patient’s needs. The pharmacist should suggest that any patient receiving long-term opioid treatment have naloxone available. This recommendation also applies to patients who have a history of addiction and are taking an opioid. The pharmacist should check the state’s PDMP to ensure that the patient has no red flags indicating potential opioid abuse. Pharmacists in each state can make recommendations to improve existing laws. Finally, pharmacists can help save lives by working with state and local organizations as advocates for patient safety.
Conclusion
The opioid epidemic has swept across the U.S., affecting people from all walks of life. Laws are continually changing to expand access to naloxone in at-risk populations. Prescribing guidelines for chronic pain have reduced the total number of prescriptions written for opioid painkillers. The establishment of PDMPs helps pharmacists and prescribers identify persons who may be at risk for opioid overdose. The pharmacist plays a vital role in combating this epidemic through direct patient education and counseling to ensure the wellbeing of citizens in the community.
REFERENCES
1. The President’s Commission on Combating Drug Addiction and the Opioid Crisis. Final report draft. November 15, 2017. www.whitehouse.gov/sites/whitehouse.gov/files/images/Final_Report_Draft_11-15-2017.pdf. Accessed November 16, 2017.
2. WhiteHouse.gov. President Donald J. Trump is taking action on drug addiction and the opioid crisis. October 26, 2017. www.whitehouse.gov/the-press-office/2017/10/26/president-donald-j-trump-taking-action-drug-addiction-and-opioid-crisis. Accessed November 9, 2017.
3. National Center for Health Statistics. Provisional counts of drug overdose deaths, as of 8/6/2017. www.cdc.gov/nchs/data/health_policy/monthly-drug-overdose-death-estimates.pdf. Accessed November 16, 2017.
4. Florence CS, Zhou C, Luo F, Xu F. The economic burden of prescription opioid overdose, abuse, and dependence in the United States, 2013. Med Care. 2016:54:901-906.
5. National Institute on Drug Abuse. Comorbidity: addiction and other mental illnesses. Why do drug use disorders often co-occur with other mental illnesses? www.drugabuse.gov/publications/research-reports/comorbidity-addiction-other-mental-illnesses/why-do-drug-use-disorders-often-co-occur-other-men. Accessed October 1, 2017.
6. CDC. Vital Signs. Opioid painkiller prescribing. www.cdc.gov/vitalsigns/opioid-prescribing/index.html. Accessed August 30, 2017.
7. Guy GP Jr, Zhang K, Bohm MK, et al. Vital signs: changes in opioid prescribing in the United States, 2006–2015. MMWR Morb Mortal Wkly Rep. 2017;66:697-704.
8. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association; 2013.
9. Murthy VH. Surgeon General’s perspectives. A promise fulfilled—addressing the nation’s opioid crisis collectively. Public Health Rep. 2016;131:387-388.
10. CDC. CDC guideline for prescribing opioids for chronic pain. www.cdc.gov/drugoverdose/prescribing/guideline.html. Accessed August 30, 2017.
11. Prescription Drug Monitoring Program Training and Technical Assistance Center. Prescription drug monitoring frequently asked questions (FAQ). www.pdmpassist.org/content/prescription-drug-monitoring-frequently-asked-questions-faq. Accessed September 2, 2017.
12. Wheeler E, Jones TS, Gilbert MK, Davidson PJ. Opioid overdose prevention programs providing naloxone to laypersons—United States, 2014. MMWR Morb Mortal Wkly Rep. 2015;64:631-635.
13. Trescot AM, Datta S, Lee M, Hansen H. Opioid pharmacology. Pain Physician: Opioid Special Issue. 2008;11:S133-S153.
14. National Institute on Drug Abuse for Teachers. How do opioids work? https://teens.drugabuse.gov/teachers/mind-over-matter/opioids/how-do-opioids-work. Accessed October 1, 2017.
15. CDC. Calculating total daily dose of opioids for safer dosage. www.cdc.gov/drugoverdose/pdf/calculating_total_daily_dose-a.pdf. Accessed September 4, 2017.
16. Substance Abuse and Mental Health Services Administration. Opioid overdose. www.samhsa.gov/medication-assisted-treatment/treatment/opioid-overdose. Accessed November 17, 2017.
17. World Health Organization. Information Sheet on opioid overdose. www.who.int/substance_abuse/information-sheet/en/. Accessed September 15, 2017.
18. American Addiction Centers. Signs of drug use in the eyes: pupil dilation, pinning, and redness. http://americanaddictioncenters.org/drug-and-alcohol-rehab/signs-of-drug-use/. Accessed September 15, 2017.
19. Naloxone: how supplied. Clinical Pharmacology [subscription database]. www.clinicalpharmacology-ip.com/Forms/Monograph/monograph.aspx?cpnum=425&sec=monsup&om=h. Accessed January 30, 2018.
20. Narcan nasal spray (naloxone hydrochloride) product information. Radnor, PA: Adapt Pharma, Inc; February 2017.
21. The Network for Public Health Law. Legal interventions to reduce overdose mortality: naloxone access and overdose Good Samaritan laws. www.networkforphl.org/_asset/qz5pvn/legal-interventions-to-reduce-overdose.pdf. Accessed August 24, 2017.
22. Fine PG, Webster LR. Preparing for life-threatening opioid emergencies in nonmedical settings. Pain Med News. September 2015. www.painmedicinenews.com/download/SR1534_Kaleo_WM.pdf. Accessed September 5, 2017.
23. Naloxoneinfo.org. Case studies. Widening the net of naloxone prescribers—the “standing order” model. http://naloxoneinfo.org/case-studies/standing-orders. Accessed September 15, 2017.
24. Nebraska Legislature. Nebraska Revised Statute 28-470. Naloxone; authorized activities; immunity from administrative action, criminal prosecution, or civil liability. https://nebraskalegislature.gov/laws/statutes.php?statute=28-470. Accessed January 30, 2018.
25. National Conference of State Legislatures. Drug overdose immunity and Good Samaritan laws. www.ncsl.org/research/civil-and-criminal-justice/drug-overdose-immunity-good-samaritan-laws.aspx. Accessed September 15, 2017.
26. Dilokthornsakul P, Moore G, Campbell JD, et al. Risk factors of prescription opioid overdose among Colorado Medicaid beneficiaries. J Pain. 2016;17:436-443.
27. National Institute on Drug Abuse. Opioid overdose reversal with naloxone (Narcan, Evzio). www.drugabuse.gov/related-topics/opioid-overdose-reversal-naloxone-narcan-evzio. Accessed August 24, 2017.
28. Prescribe to Prevent. Formulations. http://prescribetoprevent.org/pharmacists/formulations/. Accessed October 12, 2017.
To comment on this article, contact rdavidson@uspharmacist.com.





