US Pharm. 2017;(6)(Generic Drugs suppl):6-12.
The FDA announced in late August 2016 that the agency is now requiring significant changes to drug labeling with the addition of a black box warning (BBW) regarding potentially fatal risks associated with the combined use of benzodiazepines and/or other central nervous system (CNS) depressants prescribed concurrently with opiates. These medication class–related risks will also be communicated to patients through the distribution of enhanced patient FDA Medication Guides describing the serious risks of such concomitant use; these risks are primarily related to respiratory depression, but also include extreme sedation. This updated labeling is expected to affect nearly 400 products in total, involving opioid pain medications, cough products, and benzodiazepines used for insomnia, anxiety, and other neuropsychiatric conditions.1 Considering that the majority of these products have been available for decades, most of these agents are almost exclusively available in generic formulations.
One of the Newer BBWs
This FDA action follows the release of the CDC’s Prescribing Opioids for Chronic Pain Guideline in March 2016, which was prompted by the February 2016 Citizen Petition submitted to the FDA. The petition requested the removal of certain opiates from the market owing to risk of addiction and overdose. In a press release issued August 31, 2016, the FDA commissioner, Robert Califf, MD, highlighted the importance of evaluating the risk of combined use before considering co-prescribing, stating; “We implore health care professionals to heed these new warnings and more carefully and thoroughly evaluate, on a patient-by-patient basis, whether the benefits of using opioids and benzodiazepines—or CNS depressants more generally—together outweigh these serious risks.”1
Dr. Califf also reinforced the societal concern that we are experiencing a health crisis that is partially preventable through more responsible prescribing that avoids unnecessary combinations of these drugs. Because each medication class has unique pharmacokinetic and pharmacodynamic profiles, the FDA is requiring the labeling of these two drug classes to be slightly different.
Prior to requiring changes in drug labeling and issuing the BBW, the FDA evaluated extensive data, including two studies that showed a 41% increase in coprescribing of opioids along with benzodiazepines between 2002 and 2014, and an increasing frequency of misuse, abuse, and overdoses for both classes of drugs. For these drug classes, when both prescribed doses and greater-than-prescribed doses were used, overdoses increased by 13% as measured by national overdose death rates and emergency room visits.2,3
Background on BBWs and Product Labeling
The FDA is responsible for establishing efficacy and safety standards for new medications released to market. A drug is defined by the FDA as a product intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease and that is intended to affect the structure or any function of the body.4 The timeline for drugs to become available for use in the U.S. market is lengthy and costly for drug sponsors. The process for a new drug approval takes several years and includes three phases of testing.
Phase I: Phase I testing is intended to assess for toxicity and to also determine the drug’s unique pharmacodynamic and pharmacokinetic profiles, which allows for initial exposure within normal healthy human volunteers. This early exposure is critical to understanding the dosing that is necessary to achieve the adequate and intended therapeutic drug effects.
Phase II: Phase II explores early initial effectiveness of the drug. It also focuses on the primary goal of safety and the short-term risks that may be associated with the agent, using controlled studies with enrollments of a limited sample population of patients.
Phase III: Finally, phase III involves both placebo-controlled (and doubled-blinded) studies as well as uncontrolled studies that allow for the enrollment of even larger patient numbers and the confirmation of results observed in phase II trials.5 The FDA Office of Surveillance and Epidemiology continues to evaluate postmarketing safety and includes reports generated through MedWatch, which are collected in the Adverse Event Reporting System (AERS). Once a safety issue is identified, the FDA decides what action is required; this may include requiring a new BBW, which may be issued years after a drug has been released to the public.6
Prescription Drug Product Labeling Requirements
The final rule, commonly known as the Physician Labeling Rule (PLR), which amended the requirements for both the format and content of labeling for human prescription drug and biological products, was published by the FDA in 2006. This rule is critical because it addresses the essential drug labeling that is used by prescribers and other healthcare practitioners for essential drug information. One aspect of this PLR was the intent to make drug information in the labeling easier for healthcare practitioners to access and understand, in order to facilitate informed prescribing decisions. PLR labeling includes three sections7:
Highlights of Prescribing Information (Highlights): Contain specific information that providers consider most important and most commonly reference.
Table of Contents (Contents): Providers will find the organization and listing of sections and subsections for at-a-glance reference to desired information.
Full Prescribing Information (FPI): Contains the necessary detailed prescribing information for prescribers and providers to ensure safe and effective drug therapy.
In order to achieve the goal of making labeling easier to use, the PLR established minimum requirements for the format of the labeling and reorganized the order and content of the information available in the FPI. The final rule has a progressive implementation schedule and requires newly approved drugs to comply by a specific date. Older drugs that were approved under old-format labeling requirements were subject to only minor changes to regulation and are not subject to the new requirements.7
The FDA has published guidelines for manufacturers regarding the recommended structure and content of BBWs. These guidelines are strongly encouraged but not legally enforceable. Some of the recommendations include7:
• Only one BBW per drug monograph
• A drug can have multiple warnings, but they all must be contained in the same box
• Each subject (topic/warning) must be in bold print
• A warning should be displayed or presented in bulleted or itemized format
• Length should be limited to 20 lines (excluding title and reference to more information)
BBWs can be established either during clinical trials or postmarketing surveillance. If human data are not available, serious animal toxicities can be used to develop BBWs. Drugs with pharmacokinetic/pharmacodynamic properties similar to those of an established drug may inherit that BBW via theoretical risk extrapolations. Manufacturers are responsible for creating BBWs for their products based on the guidelines established by the FDA.7
Exploring BBW Consistency
Panagiotou and colleagues evaluated different BBW labeling for same-class drugs; of the 20 drug classes evaluated, 176 different agents were identified, seven of which had been withdrawn by the FDA for safety reasons.8 In only two of the seven cases, the reasons for these withdrawals had been BBWs in other same-class agents. In other drug classes, differences identified corresponded to BBWs that were not present for all drugs within the same therapeutic class/category. The format of some BBWs differed from that of other agents within the same therapeutic class, and in others only appeared as a warning in text. In other cases the warning was entirely absent. The median interval from the time the BBW had appeared in another drug of the same class was 66 months.8
Cheng et al found similar limitations, in that a drug with a BBW may be correctly identified from drug-information resources, but the description of the warning itself may be incomplete and suboptimal.9 Among the notable limitations is the absence of an official, comprehensive list that is published and updated regularly. Drug information resources do provide information about BBWs; however, even these are not uniformly updated or adequately comprehensive in all cases. MedWatch notifications containing BBWs from 1996 onward are available on the FDA website and are archived by date of release. (Table 1 summarizes inconsistencies in BBWs.) For generic agents, the difficulty encountered when trying to find products when searching by drug name becomes even more challenging.
Labeling Changes and BBWs: FDA Response
All holders of marketing applications for drug products, both New Drug Applications (NDAs) for brand drugs and Abbreviated New Drug Applications (ANDAs) for generic drugs have an ongoing obligation to ensure that their product labeling is accurate and not false or misleading. When new information becomes available that causes the labeling to become inaccurate, false, or misleading, the application holder must take steps to update its labeling.10
Once the FDA approves a change to the labeling of the brand drug—i.e., the “reference listed drug” for an ANDA—the generic-drug manufacturer is required to revise its product labeling to conform to the approved labeling of the corresponding brand drug. The FDA advises that this update should occur at the very earliest time possible; however, there may be a delay of varying lengths between the date on which revised brand-drug labeling is approved and the date on which the generic- drug manufacturer submits such labeling updates.10
In circumstances in which the brand-drug manufacturer is no longer marketing the drug and the FDA has withdrawn approval of the NDA, the brand-drug manufacturer can no longer update the drug product labeling. Generic drugs that rely on a withdrawn brand drug may continue to be marketed if the FDA withdrew approval of the NDA for the brand drug for reasons other than safety or effectiveness. The FDA has issued draft guidance to clarify existing procedures for updating the labeling of generic drugs that rely on withdrawn brand drugs.10
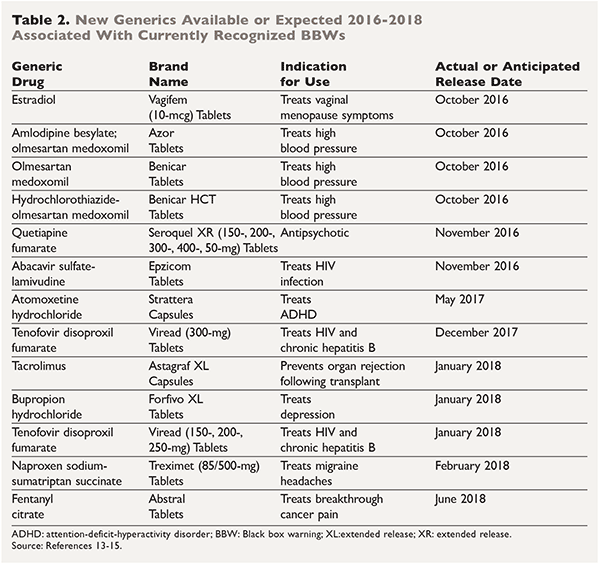
When asked about a forecast of what may be going generic soon, a spokesperson for the FDA noted that federal law prevents the agency from discussing existing or pending applications. However, the agency did provide a link to a generic-drug “dashboard” where pending ANDAs are listed for public view. The dashboard is updated quarterly and is provided by the Office of Generic Drugs for the purpose of enhancing transparency during the transition to full implementation of the Generic Drug User Fee Amendments established in 2012.11,12 The user-fee amendments are part of the ongoing effort to improve patient access to affordable generic drugs as soon as they become available. Table 2 provides information regarding generic products that recently became available and some that are expected to become available by 2018, and are currently associated with BBWs.13-15
Opiate and Benzodiazepine BBWs
The potential benefit of using benzodiazepines concomitantly with opiates in most cases does not outweigh the risk of respiratory depression and possible fatal outcomes. Prescribers are not mandated to follow the recommendations provided in BBWs; however, alternatives to using benzodiazepines in patients who require opiates are numerous. Table 3 provides some questions to consider when faced with a BBW.The majority of concomitant use is based on different indications for use; for example, with the opiate ordered for pain and the benzodiazepine ordered for anxiety. However, there are rare occasions where both agents may be ordered for a common indication, such as back pain with muscular spasm. One example of a clinically preferred alternative is replacement of a benzodiazepine ordered for routine anxiolysis, as these agents are no longer recommended as the drug of first choice for anxiety disorders and are no longer recommended at all for post-traumatic stress disorder.16 Replacing a benzodiazepine with a selective serotonin reuptake inhibitor/selective norepinephrine reuptake inhibitor (SSRI/SNRI) antidepressant for these patients can provide an excellent alternative, although many prescribers may still be willing to accept the BBW risk by continuing to provide a very brief, low-dose benzodiazepine for initial anxiolysis while waiting for the antidepressant actions to fully take effect. A benzodiazepine used for insomnia can also be replaced with a novel sleep agent such as an orexin antagonist or a melatonin agonist, and benzodiazepines used for seizure disorders can be replaced with one of the newer antiepileptic agents that offer mood-stabilizing effects for patients who can use that additional benefit.15
Regardless of whether a patient is taking a benzodiazepine or not, all patients who are candidates for pain management should have nonopiate agents and nonpharmacologic interventions fully explored before starting (or continuing) an opiate, and even patients who are appropriately receiving opiates should continue to receive nonpharmacologic and nonopiate interventions concurrently.17 For non–pain conditions such as cough, alternatives such as benzonatate or dextromethorphan should be considered in any patient receiving a benzodiazepine or other CNS depressant.
Role of the Pharmacist
Prescribers and pharmacists who are managing drug therapy must review BBWs prior to prescribing and dispensing. The absence of a BBW for a drug, whether from an online drug-information resource or paper labeling, may not ensure that a drug does not carry such serious warnings. Prescribers and pharmacists should be aware that if a drug does not have a BBW, it may still have severe warnings/contraindications associated with its use. Pharmacists can play a significant role in educating and assisting prescribers in cross-referencing resources to establish confidence that BBWs either do or do not exist, especially with older generic drugs that are not subject to the same labeling requirements as the new brand products.
Conclusion
It is important that healthcare providers not rely solely on a BBW to flag serious warnings/precautions or contraindications, as these may still exist in the text narrative within the package insert. And although BBWs are considered the highest level of alert for medication safety, not every serious warning/contraindication of a drug will be included in the BBWs. Healthcare providers should always use clinical judgment when prescribing, dispensing, and administering these agents and consider the use of more than one drug-information resource to evaluate potential risks as a best practice to ensure optimal patient safety.
REFERENCES
1. FDA requires strong warnings for opioid analgesics, prescription opioid cough products, and benzodiazepine labeling related to serious risks and death from combined use. August 31, 2016. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm518697.htm. Accessed March 7, 2017.
2. Hwang CS, Kang EM, Kornegay CJ, et al. Trends in the concomitant prescribing of opioids and benzodiazepines, 2002-2014. Am J Prev Med. 2016;51:151-160.
3. Jones CM, McAninch JK. Emergency department visits and overdose deaths from combined use of opioids and benzodiazepines. Am J Prev Med. 2015;49:493-501.
4. FDA. Drugs@FDA Glossary of Terms. April 10, 2017. www.FDA.gov/Drugs/InformationOnDrugs/ucm079436.htm#D. Accessed May16, 2017.
5. Federal Register clinical trials registration and results information submission. September 21, 2016. www.federalregister.gov/documents/2016/09/21/2016-22129/clinical-trials-registration-and-results-information-submission. Accessed March 14, 2017.
6. O’Connor NR. FDA boxed warnings: how to prescribe drugs safely. Am Fam Physician. 2010;81(3)298-303.
7. Guidance for Industry. Labeling for human prescription drug and biological products – implementing the PLR content and format requirements. February 2013. www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm075082.pdf. Accessed March 7, 2017.
8. Panagiotou OA, Contopoulos-Ioannidis DG, Papanikolaou PN, et al. Different black box warning labeling for same-class drugs. J Gen Int Med, 2011;26(6):603-610.
9. Cheng CM, Guglielmo J, Maselli J, Auerbach AD. Coverage of FDA medication boxed warnings in commonly used drug information resources. Arch Intern Med. 2010;170(9):831:833.
10. FDA. Interview with Kristofer Baumgartner, FDA spokesperson, Center for Drug Evaluation and Research. February 23, 2017.
11. FDA. Draft Guidance. Updating ANDA labeling after the marketing application for the reference listed drug (RLD) has been withdrawn. July 2016. www.fda.gov/downloads/Drugs/Guidances/UCM510240.pdf. Accessed March 7, 2017.
12. FDA. Generic drug review dashboard report. Updated February 25, 2017.
www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/AbbreviatedNewDrugApplicationANDAGenerics/ucm484609.htm. Accessed March 7, 2017.
13. FDA. Orange Book: approved drug products with therapeutic equivalence evaluations. March 2017. www.accessdata.fda.gov/scripts/cder/ob/default.cfm. Accessed March 13, 2017.
14. Boothby LA. New generics in the pipeline. US Pharm. 2016;41(6);12-19.
15. Clinical Pharmacology [online database]. Tampa, FL: Gold Standard, Inc; 2013. www.clinicalpharmacology.com. Accessed March 13, 2017.
16. National Center for PTSD. Use of benzodiazepines for PTSD in Veterans Affairs. Updated March 28, 2017. www.ptsd.va.gov/professional/treatment/overview/benzo-ptsd-va.asp. Accessed March 10, 2017.
17. World Health Organization. WHO’s Pain Ladder. www.who.int/cancer/palliative/painladder/en. Accessed March 13, 2017.
To comment on this article, contact rdavidson@uspharmacist.com.








