ABSTRACT: The past decade has brought several new developments in the treatment of type 2 diabetes, including the glucagon-like peptide-1 receptor agonists (GLP-1 RAs). Their mechanism of action makes them unique due to their potential for weight loss, low risk of hypoglycemia, and A1C-lowering potential with effects on both fasting and postprandial blood glucose values. Administration burden ranges from twice daily to once weekly. These features make GLP-1 RAs an advantageous alternative to rapid-acting insulin for patients inadequately controlled on a long-acting basal insulin. Head-to-head comparator trials have been conducted, supporting the use of GLP-1 RAs in this role.
There have been significant advances in type 2 diabetes mellitus management within the past decade. One of the more recent developments is the introduction of glucagon-like peptide-1 receptor agonists (GLP-1 RAs). The first agent in this class, exenatide, was approved by the FDA in 2005.1 Since then, several additional formulations have been approved. GLP-1 RAs have effects on both fasting and prandial blood glucose levels.2 They are also associated with a significant potential to lower A1C. A head-to-head review of clinical studies determined that among available GLP-1 RAs, there were A1C reductions from 0.78% to 1.9%.3
Pharmacology
GLP-1 is one of several gastric incretin hormones released in response to oral glucose intake.1 The “incretin effect” was termed when researchers noted that individuals had a greater secretion of insulin following an oral glucose challenge as opposed to IV glucose administration.1 This led researchers to further investigate the link between gastric hormones and insulin release.
It has been shown that patients diagnosed with both prediabetes and diabetes mellitus have a deficiency in GLP-1 secretion.1 The reduced incretin response leads to decreased insulin release and increased blood glucose concentrations following a meal.1 This mechanism suggested that GLP-1 would be an excellent target for controlling postprandial blood glucose levels. In addition, the insulin release stimulated by GLP-1 is glucose-dependent.1 This translates into a lower risk of hypoglycemia compared to other diabetes treatment options like sulfonylureas and insulin.
Currently Available GLP-1 RAs
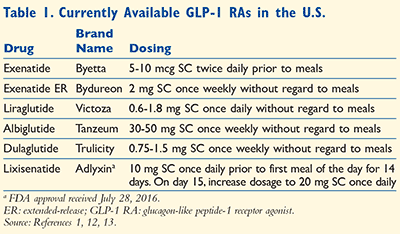
Currently available GLP-1 RAs are summarized in TABLE 1. These agents require careful dose titrations due to their adverse drug event (ADE) profile, which commonly includes nausea and vomiting.1 GLP-1 RAs should be titrated until A1C goal, maximum dose, or patient tolerability has been met.1 If intolerable, providers should return to the last tolerable dosage or discontinue the drug.1 Although GLP-1 RAs are considered to have effects on both fasting and prandial glucose control, evidence suggests that the shorter-acting agents (exenatide and liraglutide) have a greater effect on postprandial values compared to the weekly administered GLP-1 RAs.4
Benefits
In addition to stimulating insulin release in response to a meal, GLP-1 RAs have demonstrated many other beneficial effects. Administration of a GLP-1 RA may decrease glucagon secretion and hepatic gluconeogenesis; increase insulin sensitivity; delay gastric emptying; promote central satiety; and reduce overall caloric intake.1 All of these effects would be beneficial at overcoming different aspects of the pathophysiology of type 2 diabetes.
The effects on delayed gastric emptying, satiety, and decreased caloric intake can promote weight loss. Weight loss is an important factor in type 2 diabetes management and is often complicated by diabetic agents that promote weight gain (i.e., sulfonylureas, thiazolidinediones (TZDs), insulin). In a 2015 meta-analysis, the mean reduction of weight for GLP-1 RAs was -1.01 kg to -1.62 kg compared to placebo.5 As comparison, 70/30 mixed insulin aspart was shown to have a mean increased weight of 4.04 kg.5
Additionally, clinical trials have shown GLP-1 RAs to have beneficial effects on lipid profiles and blood pressure.1 The LEADER trial (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results—A Long-Term Evaluation), a large double-blind, placebo-controlled trial, recently demonstrated that liraglutide added to standard therapy for a relatively short period of time (3.5-5 years) had a significant reduction in cardiovas-cular events and mortality compared to placebo in high-risk patients (hazard ratio [HR], 0.87; 95% CI, 0.78-0.97).6 Additional studies are currently under way to investigate if this effect is seen with other GLP-1 RAs.1
Safety
The most common ADEs seen with GLP-1 RAs are nausea, vomiting, and diarrhea, to which patients can develop tolerance over time.1 Injection-site reactions and the formation of nodules or palpable bumps under the skin can also occur.1 The nodules are not typically harmful, but patients should be warned prior to starting treatment. There have also been documented local skin reactions when using GLP-1 RAs, particularly with the long-acting formulations. One study found that dose-related local skin reactions were more common in patients treated with albiglutide compared to twice-daily exenatide or placebo.7
Serious adverse events, including significant hypoglycemia, are rare.1 Patients on concomitant insulin therapies or insulin secretagogues are at a higher risk for this adverse event.1 Close monitoring and careful dose adjustments, especially when initiating therapy, are recommended for patients with gastroparesis or severe gastroesophageal reflux disorder (GERD) due to the ability of GLP-1 RAs to slow gastric emptying.2 This effect tends to be more severe with shorter-acting GLP-1 RAs.4
Studies in animals have led to concerns about the risk of acute pancreatitis and medullary thyroid carcinoma (MTC).1 In human trials, acute pancreatitis has occurred, although rarely.1 Some have cited concern over confounding variables including the fact that patients with diabetes are at an increased risk of acute pancreatitis regardless of treatment choice.1 Guidelines, manufacturers, and FDA warnings have erred on the side of caution and recommend careful use of GLP-1 RAs in patients with a history or high risk of pancreatitis. It is also recommended to discontinue the drug immediately if acute pancreatitis occurs.2 Based on available evidence, there have been no cases of MTC in humans linked to GLP-1 RAs.1 However, it is recommended to avoid use in patients with a personal or family history of MTC and in patients with multiple neoplasia syndrome type 2.2
Exenatide is eliminated renally and has been associated with rare occurrences of acute kidney injury.1 This agent is not recommended in patients with a creatinine clearance (CrCl) <30 mL/min.2 A few case reports of acute kidney injury have also been seen with liraglutide in patients who had preexisting renal insufficiency.1 This concern has not been seen with the other GLP-1 RAs.1
Finally, there is a concern regarding the development of GLP-1 RA antibodies that put patients at risk of hypersensitivity reactions and reduced efficacy.1 The different GLP-1 RAs vary in the number of antibodies seen in clinical trials, with exenatide (once weekly) having the greatest and dulaglutide and albiglutide having the least.1 In clinical trials, patients with higher antibody titers have been seen to have smaller reductions in A1C compared to those with low or negative titers.1 The incidence of antibody formation has been shown to decrease with time (over 3 years).1
Role in Therapy
Many studies and reviews have explored the role of GLP-1 RAs within diabetes management. Several guidelines currently recommend GLP-1 RAs as add-on therapy to almost all diabetic treatments (with the exception of dipeptidyl peptidase-4 [DPP-4] inhibitors) and even as monotherapy to those intolerant to metformin.2,8 With increasing evidence, additional roles are being proposed. The weight loss, frequency of administration, low risk of hypoglycemia, and mechanism of controlling prandial blood glucose seen with GLP-1 RAs offer many advantages as an alternative to prandial insulin. Both the 2016 consensus statement of the American Association of Clinical Endocrinologists and American College of Endocrinology (AACE/ACE) on management of type 2 diabetes and the 2016 American Diabetes Association (ADA) standards recommend adding on GLP-1 RAs to patients not meeting treatment goals with basal insulin.2,8 This review aims to explore the role of GLP-1 RAs as add-on therapy to basal insulin, as an alternative to prandial or rapid-acting insulin.
Evidence
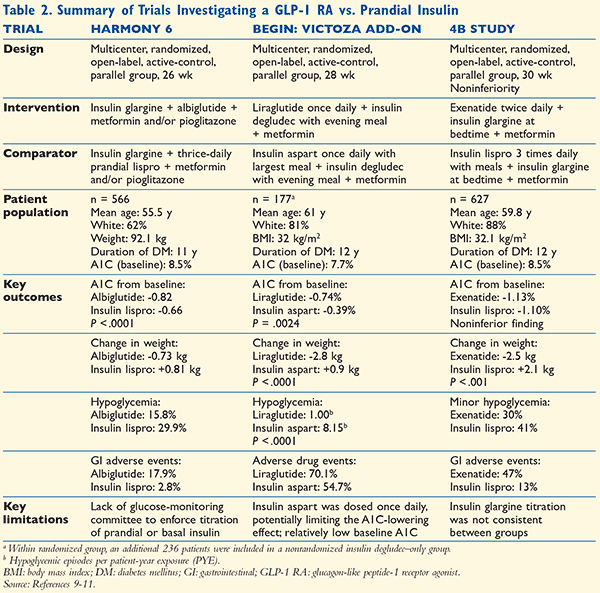
Trials investigating the use of GLP-1 RAs currently approved in the United States in addition to basal insulin versus an active comparator of prandial insulin are summarized in TABLE 2.9-11 It should be noted that there are several well-designed landmark trials investigating a GLP-1 RA in addition to basal insulin with placebo or other comparators. Although these trials are valuable, they were excluded within the scope of this review because they lacked head-to-head comparison with prandial insulin.
Discussion
The three trials summarized in TABLE 2 provide consistent evidence that GLP-1 RAs may be an advantageous alternative to prandial insulin as add-on therapy to basal insulin.9-11 These clinical trials have demonstrated that GLP-1 RAs will provide similar, and sometimes superior, reductions in A1C compared to rapid-acting insulin.9-11 This review should be concluded with the acknowledgement of an important limitation—these trials had a relatively short duration. It is hypothesized that with continued use and continued titration of insulin, superior reductions in A1C would be seen with an intensive insulin therapy compared to GLP-1 RAs. Based on the studies reviewed, the role of GLP-1 RAs may be for patients who have optimized a basal insulin regimen and are 0.5% to 1.5% away from achieving their A1C goal.
GLP-1 RAs demonstrated a beneficial effect on weight loss as opposed to the weight gain seen with insulin.9-11 This effect on weight may provide motivation to patients to continue additional weight loss strat-egies, as they are no longer competing against their insulin to lose weight. Additionally, there is a lower incidence of hypoglycemia with GLP-1 RAs compared to prandial insulin.9-11 Hypoglycemia is something that patients fear, and this threat can lead to distrust or nonadherence to therapy. Avoiding hypoglycemia would not only improve health outcomes, it would potentially improve patient satisfaction.
The 4B trial was able to demonstrate significantly higher scores for quality of life in the exenatide group.11 These measures included treatment satisfaction, treatment convenience, treatment flexibility, physical function, self-esteem, and work as compared to the insulin lispro group.11 These patient-centered outcomes should not be overlooked.
Each patient’s medication regimen should be individualized to his or her needs, and the cost of these therapies should be considered. With additional clinical trials under way, and the development of a fixed-dose combination product of GLP-1 RA and basal insulin, the use of GLP-1 RA as an alternative to prandial insulin seems likely to increase with time.
Recommendations to Pharmacists
Pharmacists should use a patient-centered approach and evaluate whether each individual patient would benefit from the addition of a GLP-1 RA as an alternative to thrice-daily prandial insulin injections. To determine if this is the most appropriate treatment, pharmacists should consider the following:
- Administration burden: Include frequency of use and timing with meals. GLP-1 RAs are considered to have a lower administration burden.4,11
- Efficacy: GLP-1 RAs have been shown to be at least equivalent to prandial insulin in short duration trials with baseline A1C <9%.9-11 Patients requiring stronger therapy would likely benefit from intensive insulin titration.
- Hypoglycemia: The risk is less with GLP-1 RAs.9-11
- Weight: Weight loss is seen with GLP-1 RAs as opposed to weight gain with insulin therapy.9-11
- Gastrointestinal effects: These are a significant concern with GLP-1 RAs compared to insulin, but tolerance can develop with time.9-11
If a GLP-1 RA is determined to be the best option for the patient, pharmacists and providers should consider the following recommendations:
- Follow dosing recommendations as directed by the manufacturer.
- Consider decreasing or closely monitoring the dose of basal insulin to reduce the risk of hypoglycemia.2 A 10% to 20% reduction in basal insulin dose has been used in clinical trials.10,11
- Patients should be instructed to measure blood glucose frequently and be educated on proper hypoglycemia protocols.1
- Monitor renal function and CrCl for all GLP-1 RAs.1 Discontinue exenatide if CrCl <30 mL/min.2
- Patients should be advised about the signs and symptoms of pancreatitis, even though this adverse event is considered to be rare.1 Discontinue immediately if pancreatitis occurs.2
- Patients should also be educated about the common adverse events of nausea, vomiting, and diarrhea. Tolerance to these adverse events can develop, but patients should contact their provider if intolerable.1
- Consider continuing metformin without dose reductions.10,11
REFERENCES
1. Prasad-Reddy L, Isaacs D. A clinical review of GLP-1 receptor agonists: efficacy and safety in diabetes and beyond. Drugs Context. 2015;4:212283.
2. Garber AJ, Abrahamson MJ, Barzilay JI, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinology on the comprehensive type 2 diabetes management algorithm—2016 executive summary. Endocr Pract. 2016;22(1):84-113.
3. Trujillo JM, Nuffer W, Ellis SL. GLP-1 receptor agonists: a review of head-to-head clinical studies. Ther Adv Endocrinol Metab. 2015;6(1):19-28.
4. Hirsch IB, Buse JB, Leahy J, et al. Options for prandial glucose management in type 2 diabetes patients using basal insulin: addition of a short-acting GLP-1 analogue versus progression to basal-bolus therapy. Diabetes Obes Metab. 2014;16(3):206-214.
5. Potts JE, Gray LJ, Brady EM, et al. The effect of glucagon-like peptide 1 receptor agonists on weight loss in type 2 diabetes: a systematic review and mixed treatment comparison meta-analysis. PLoS One. 2015;10(6):e0126769.
6. Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375(4):311-322.
7. Rosenstock J, Reusch J, Bush M, Yang F, Stewart M. Potential of albiglutide, a long-acting GLP-1 receptor agonist, in type 2 diabetes: a randomized controlled trial exploring weekly, biweekly, and monthly dosing. Diabetes Care. 2009;32(10):1880-1886.
8. American Diabetes Association. Approaches to glycemic treatment. Sec. 7. In: Standards of Medical Care in Diabetes—2016. Diabetes Care. 2016;39(suppl 1):S52-S59.
9. Rosenstock J, Fonseca VA, Gross JL, et al. Advancing basal insulin replacement in type 2 diabetes inadequately controlled with insulin glargine plus oral agents: a comparison of adding albiglutide, a weekly GLP-1 receptor agonist, versus thrice-daily prandial insulin lispro. Diabetes Care. 2014;37(8):2317-2325.
10. Mathieu C, Rodbard HW, Cariou B, et al. A comparison of adding liraglutide versus a single daily dose of insulin aspart to insulin degludec in subjects with type 2 diabetes (BEGIN: VICTOZA ADD-ON). Diabetes Obes Metab. 2014;16(7):636-644.
11. Diamant M, Nauck MA, Shaginian R, et al. Glucagon-like peptide 1 receptor agonist or bolus insulin with optimized basal insulin in type 2 diabetes. Diabetes Care. 2014;37(10):2763-2773.
12. FDA approves Adlyxin to treat type 2 diabetes. FDA news release. July 28, 2016. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm513602.htm. Accessed August 24, 2016.
13. Adlyxin (lixisenatide). Bridgewater, NJ: Sanofi-Aventis; July 2016. http://products.sanofi.us/adlyxin/adlyxin.pdf. Accessed August 24, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.





