US Pharm. 2011;36(9):HS25-HS28.
Breast cancer is the most common type of cancer among females in the United States. In 2007, 202,964 women in the U.S. were diagnosed with breast cancer.1 In 20% to 30% of breast cancers, there is an overexpression of human epidermal growth factor receptor 2 (HER2), which is a protein that normal breast epithelial tissue expresses in low levels.2 Similarly, there is an overexpression in gastric cancers. Overexpression of HER2 allows for the transmission of growth signals, which alter normal cell growth and division and have been associated with increased tumor aggressiveness and recurrence risk.
Trastuzumab (Herceptin) is a monoclonal antibody that targets the HER2 receptor protein through antibody-dependent cellular cytotoxicity. Trastuzumab demonstrates decreased mortality rates in HER2-positive breast and gastric cancers when administered with chemotherapy.2 Clinical trials have demonstrated the efficacy of this biological agent with improvements in objective response rate, duration of response, and overall survival.3 However, benefits of this agent appear to be realized only in the presence of HER2-positive breast and gastric cancers. Therefore, a method to identify overexpression of this protein is critical.
HercepTest: Background Information
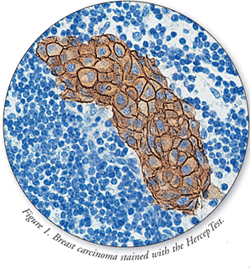
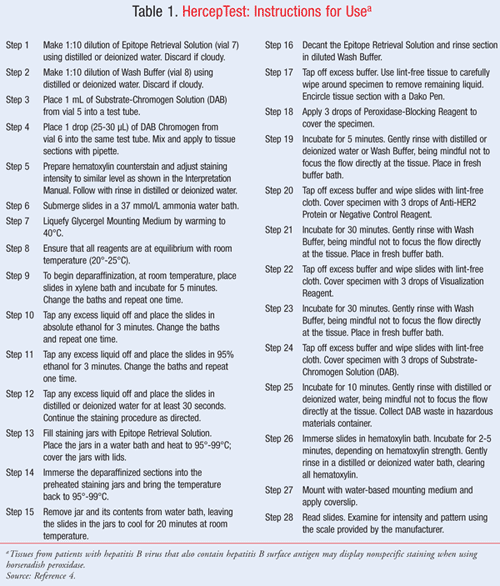
The HercepTest (Dako North America, Inc.) is a semiquantitative immunocytochemical assay used to determine HER2 protein overexpression in breast cancer tissues routinely processed for histologic evaluation (FIGURE 1) and in formalin-fixed, paraffin-embedded cancer tissue from patients with metastatic gastric or gastroesophageal junction adenocarcinoma. HercepTest is indicated as an aid in the determination of whether trastuzumab treatment may be considered. This test is approved for breast and gastric cancers only.4 Instructions for its use are provided in TABLE 1.
Evaluation and Assessment: Breast and Gastric Cancers
Although several methods for HER2 testing have been developed, approximately 20% of current testing may be inaccurate.5 The American Society of Clinical Oncology (ASCO) and the College of American Pathologists (CAP) have developed guidelines on HER2 testing to ensure accuracy.5 Recommendations suggest that breast cancer should initially undergo HER2 testing by a validated immunohistochemistry (IHC) assay such as HercepTest to determine HER2 protein expression. The HER2 expression is based on the cell membrane staining pattern (TABLE 2).5
Breast cancers with equivocal IHC should undergo validation using a HER2 gene amplification method, such as fluorescence in situ hybridization (FISH). Though FISH is thought to be more reliable than IHC, it is also more expensive. Ambiguous IHC can be seen in 15% of invasive breast cancers, whereas uncertain HER2 FISH results (TABLE 3) are seen in less than 3% of invasive breast cancer specimens previously considered HER2 positive.5 Newer methodologies for establishing HER2 expression include reverse transcriptase-polymerase chain reaction and chromogenic in situ hybridization (CISH). These have not yet been validated.
Gastric cancer is the second leading cause of cancer mortality in the world.6 In the U.S., gastric cancer is currently the 14th most common type of cancer.6 Its incidence has decreased over the last half century due to refrigeration, increased consumption of fresh fruits and vegetables, decreased salt intake, and decreased contamination of food.6 HER2 overexpression is increasingly recognized as a frequent molecular abnormality. There is rising awareness of the significance of HER2 overexpression in patients with gastric cancer, and it has been correlated to poor outcomes and more aggressive disease.7
Trastuzumab is also indicated for treatment of HER2 overexpression in metastatic gastric or gastroesophageal junction adenocarcinoma. It is given in combination with cisplatin and capecitabine or 5-fluorouracil (5-FU) in those who have not been previously treated for metastatic disease.7
Results from five studies (HERceptin Adjuvant [HERA], Finland Herceptin [FinHer], NSABP B-31, BCIRG006, and N9831) demonstrated that the inclusion of trastuzumab produces a 50% improvement in disease-free survival and 33% improvement in overall survival, regardless of the chemotherapy regimen or sequence of trastuzumab delivery.5 These trials randomized 11,650 women with early-stage HER2-positive breast cancer to trastuzumab versus nontrastuzumab chemotherapy. Based on these results, trastuzumab was approved by the FDA for the treatment of HER2-positive cancer.5
The phase III Adjuvant Lapatinib and/or Trastuzumab Treatment Options (ALTTO) and the Neoadjuvant Lapatinib and/or Trastuzumab Treatment Optimization (Neo-ALTTO) are two trials that are currently investigating whether the combination of two anti-HER2 targeted therapies with chemotherapy will yield further benefit in the treatment of early-stage breast cancer.5

Efficacy of HercepTest
Efficacy was evaluated using two studies. One study compared HercepTest to Clinical Trial Assay (CTA), and the other compared HercepTest to five different assays.4 Though CTA is no longer available, it was used to identify patients eligible for participation in trastuzumab clinical trials.
HercepTest was compared to CTA using a group of 274 HER2 protein–positive (2+ or 3+) samples and 274 HER2 protein–negative (0 or 1+) samples.4 The binary concordance of HercepTest to CTA was 79% (95% confidence interval [CI], 76%-82%).4
A 3 × 3 concordance revealed that 42% of HercepTest 2+ results were negative according to CTA. Therefore, participants with these specimens would not have been considered for trastuzumab trials with CTA. Results demonstrated that HercepTest is in agreement with CTA 79% of the time.4
One of the studies used tumors identified as positive for HER2 protein overexpression by five different assays: Southern blot, FISH for amplification of DNA, Northern blot RNA analysis, Western blot, and immunocytochemistry. The results of this study demonstrated an 85% (95% CI, 78%-89%) concordance.4 No specimen considered negative by the five other assays was found to be positive by HercepTest.

New Development: Inform Dual In Situ Hybridization
On June 14, 2011, the FDA approved the Inform Dual ISH test (Ventana Medical Systems).8 Similar to the HercepTest, this is a genetic test that determines if breast cancer tissue is HER2 positive and if the patient is a candidate for treatment with trastuzumab. The test measures the number of copies of the HER2 gene, which is located on chromosome 17 of human cells in tumor tissue. It is designed to detect amplification through light microscopy of the HER2 gene using two-color CISH in formalin-fixed, paraffin-embedded human breast and gastric cancer.8
Conclusion
The HercepTest kit is a safe and reliable method to determine HER2 protein overexpression in breast and gastric cancers. The ASCO and CAP guidelines recommend that such a test be used as an initial assessment for the overexpression of HER2 proteins.5 Based on clinical studies, the HercepTest has been shown to correlate well with other testing methods.4
REFERENCES
1. Breast cancer. Centers for Disease Control and Prevention. www.cdc.gov/cancer/breast/
2. Michaud L, Espirito J, Esteva F. Breast cancer. In: Dipiro J, Talbert R, Yee G, et al. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill; 2008:2121-2156.
3. Harbeck N, Pegram M, Rüschoff J, Möbus V. Targeted therapy in metastatic breast cancer: the HER2/neu oncogene. Breast Care (Basel). 2010;5(suppl 1):3-7.
4. HercepTest package insert. 2nd edition. Carpinteria, CA: Dako North America, Inc; 2010.
5. Markman M, Windle M, Buehler B. Breast cancer and HER2. Medscape. Updated March 29, 2011. www.medscape.com/article/
6. Cabebe E, Mehta V, Fisher G, et al. Gastric cancer. Medscape. www.medscape.com/article/
7. Gravalos C, Jimeno A. HER2 in gastric cancer: a new prognostic factor and a novel therapeutic target. Ann Oncol. 2008;19:1523-1529.
8. Nelson R. FDA approves test to determine HER2 status in breast cancer. Medscape Medical News. June 14, 2011. www.medscape.com/viewarticle/
To comment on this article, contact rdavidson@uspharmacist.com.





