US Pharm. 2016;41(12):HS2-HS8.
ABSTRACT: Bariatric surgeries have increased significantly in recent years as a viable long-term treatment option for severe obesity or obesity with comorbid conditions such as type 2 diabetes. These procedures, especially the Roux-en-Y gastric bypass, can have significant effects on medication and nutrient absorption. Data currently are limited on effects of bariatric surgery on specific medications. Pharmacists, as the dosage-form experts, serve an important and unique function in helping to ensure appropriate medication and nutrient therapy in the post–bariatric surgery patient in both inpatient and outpatient settings as well as during transition of care.
Obesity is a chronic medical condition associated with significant medical and psychological comorbidity, as well as increased mortality. In 2009-2010, more than one-third of adults in the United States were considered obese, defined as a body mass index (BMI) ≥30 kg/m2.1 Evidence has shown that bariatric surgery not only serves as a means of weight reduction but also reduces morbidity and mortality rates in severely obese patients. Bariatric surgery candidates include those with a BMI ≥40, or a BMI ≥35 with one or more significant obesity-associated comorbidities, including type 2 diabetes, hypertension, obstructive sleep apnea, hyperlipidemia, obesity-hypoventilation syndrome, asthma, venous stasis disease, severe urinary incontinence, severe arthritis, gastroesophageal reflux disease (GERD), nonalcoholic fatty liver disease, or nonalcoholic steatohepatitis.2
Bariatric Procedures
Currently, bariatric procedures are the most successful and durable treatment for obesity.3-6 The number of bariatric procedures performed in the U.S. has increased significantly from an estimated 16,000 procedures performed annually in the early 1990s to approximately 179,000 in 2013.7,8
The three bariatric procedures primarily performed in the U.S. are laparoscopic adjustable gastric banding (LAGB), laparoscopic sleeve gastrectomy (LSG), and laparoscopic Roux-en-Y gastric bypass (RYGB).2 Bariatric surgery achieves weight loss by altering the anatomy of the gastro-intestinal (GI) tract via restrictive and/or malabsorptive mechanisms. Restrictive procedures achieve weight loss by limiting the volume of food intake while diminishing the stomach’s capacity. Malabsorptive procedures achieve weight loss by surgically bypassing regions of the small intestine and diverting biliopancreatic secretions, which limit nutrient absorption.9 The various procedures are summarized in TABLE 1.9-14
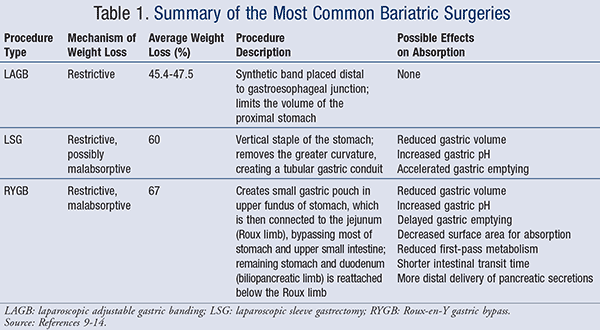
Due to the increased prevalence of obesity and use of bariatric surgery worldwide, it is crucial that pharmacists and healthcare providers have a better understanding of nutrient and medication malabsorption in the post–bariatric surgery population. RYGB is the most frequently performed bariatric procedure in the U.S. and is associated with more pharmacologic/metabolic concerns when compared to other procedures. Thus, the purpose of this article is to review the effects of bariatric surgery, particularly RYGB, on supplement and medication malabsorption.
Bariatric Surgery and Malabsorption
The risk for malabsorption (nutrient or medication) after bariatric surgery is dependent upon the type of surgical procedure performed; the patient’s level of adherence to medications, supplements, and diet; and the types of dosage forms administered following surgery. Pre- and postoperative nutrient deficiencies have been observed for several decades in the bariatric population.15 In addition, postoperative alterations in the pharmacokinetics of medications have been described and can be attributed to a variety of mechanisms.14,16
Nutritional Deficiency and Supplementation
Morbid obesity (BMI ≥40) has been shown to be associated with micronutrient deficiency, although the mechanism is not completely understood. Potential mechanisms may include impaired expression of transporter proteins due to chronic inflammation, small intestinal overgrowth syndrome, and eating behaviors favoring high energy-dense foods with poor micronutrient content.17 With the high prevalence of preexisting deficiencies in the obese population, the importance of monitoring for deficiencies and providing adequate supplementation, as well as assessing adherence to supplementation post surgery, is vital. Data analyzing adherence to supplementation are scarce; however, adherence seems to be low according to the available literature. Brolin and Leung followed patients over a 10-year period postoperatively and reported that only 33% of patients complied with multivitamin supplemen-tation.18 Sawhney et al assessed adherence in adolescents 6 months post surgery and found that 30.7% of patients remained adherent to multivitamin regimens.19
In addition to supplement adherence, the type of procedure performed and the nutrient salt administered also impact whether or not a deficiency is likely to occur. Malabsorptive procedures can lead to nutrient deficiency through changes in biliary and pancreatic functions, alterations in GI transit time/gastric acid secretion, bypass of the duodenum (a primary site of absorption), and promotion of small intestinal bacterial overgrowth (SIBO). Restrictive procedures can also lead to nutrient deficiency via food intolerance caused by nausea, vomiting, and regurgitation. Deficiencies of fat-soluble nutrients or nutrients absorbed in the bypassed section of the GI tract are most common and include folate, iron, calcium, and vitamins B1, B12, A, and D. A summary of these deficiencies and their associated prevalence rates can be found in TABLE 2.9,17
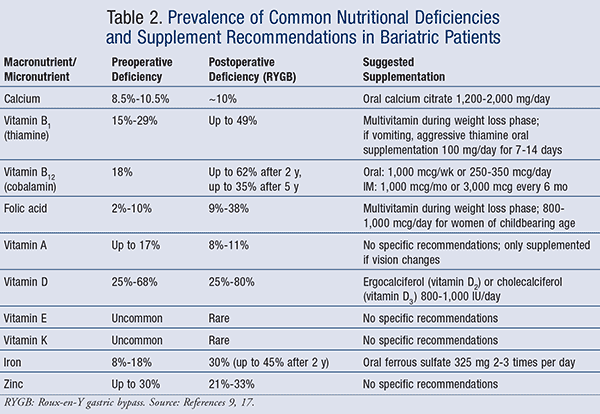
In general, all patients post bariatric surgery should take a multivitamin supplement indefinitely, with the percentage of recommended daily dose dependent on the type of surgery. LAGB/LSG patients should take a multivitamin with 100% of the recommended daily dose of nutrients, and RYGB patients should take a multivitamin with 200% of the recommended daily dose of nutrients.17 Patients should also receive daily calcium supplementation in addition to the multivitamin.
Reduced gastric pH following gastric bypass affects calcium absorption.17 Patients should use calcium citrate rather than calcium carbonate, as the citrate form does not require acid to be absorbed. Although calcium citrate is more expensive and contains less calcium per tablet, it is associated with less GI upset and does not need to be taken with food. It is difficult to assess true calcium deficiency due to homeostatic mechanisms of calcium in the blood via renal calcium reabsorption and bone resorption. Thus, it is important to ensure that patients’ vitamin D, 25-hydroxy levels remain within normal range and to provide adequate amounts of vitamin D to maintain normal levels for proper calcium absorption from the GI tract. Most calcium citrate formulations contain vitamin D, making cosupplementation more convenient.
This patient population is also at increased risk of anemia secondary to potential deficiencies in iron, folate, and vitamin B12.20 The primary site for iron absorption is the duodenum, which is bypassed in RYGB procedures. The reduced gastric acid environment of the stomach can lead to decreased conversion of iron from the ferric form (Fe3+) to the ferrous form (Fe2+), which is required for absorption. In patients identified as having iron deficiency anemia (microcytic anemia) following gastric bypass surgery, coadministration of vitamin C with iron supplementation can increase the conversion of iron to its absorbable form by reducing the pH of the stomach.21 Vitamin B12 and/or folate deficiencies post surgery can lead to development of megaloblastic (macrocytic) anemia. Factors underlying vitamin B12 deficiency are likely due to reduced intake of meat, diminished contact of food with gastric acid, and decreased secretion of intrinsic factor. The limited number of parietal cells in the stomach post surgery leads to subsequent decrease in cobalamin–intrinsic factor complex formation and absorption. On the other hand, folate deficiency is likely due to bypass of the proximal small intestine, which is the primary site of absorption. Folate supplementation is important, especially for women who become pregnant after bariatric surgery, because maternal folate deficiency can lead to neural tube defects in newborns.9,22
Deficiencies in lipid-soluble vitamins (A, D, E, K) may occur post surgery due to fat malabsorption resulting from biliopancreatic diversion. Although deficiency of vitamin D is more common as previously discussed, deficiency in vitamins A, E, and K is uncommon if patients are adherent to multivitamin regimens. However, these vitamins still play an important role in maintaining good health. Vitamin K is required for development of certain clotting factors (II, VII, IX, X) and for chondrogenesis, a process important for fetal development. Vitamin E functions as an antioxidant and protects cells, tissues, and organs from damaging effects caused by free radicals. Vitamin A is important for vision; however, supplementation should be cautioned in women of childbearing age because of teratogenic effects with toxic concentrations. Pre- and postoperative monitoring for deficiency in these vitamins is recommended.22
In addition to malnutrition, deficiencies can lead to development of other disease states and comorbid conditions. Patients can develop low bone density or osteoporosis (from calcium/vitamin D deficiency), muscle contractions, pain/spasms (from calcium/magnesium deficiency), beriberi, neuropsychiatric complications (from vitamin B1 deficiency), and anemia (from iron, folic acid, vitamin B12 deficiency), as well as countless other conditions. Thus, it is important to educate bariatric patients on the importance of adherence to supplementation both pre- and post-operatively.17
Among the benefits of bariatric surgery is that many patients have significant attenuation or resolution of GERD symptoms post surgery. If a patient had been on chronic acid-suppressing therapy prior to surgery, he or she may no longer need to continue this therapy post surgery. The impact of acid-suppressing medications, particularly proton pump inhibitors, on absorption of nutrients has been well documented. Therefore, an attempt to remove acid-suppressing therapies should be performed post surgery with the goal to limit absorption issues of several nutrients including calcium, iron, and vitamin B12, which may limit complications associated with these deficiencies.23,24
Oral Bioavailability of Drugs
Availability of oral medications to the systemic circulation is dependent upon its absorption, also known as oral bioavailability. The rate and extent of absorption depend on the medication’s disintegration, dissolution, and diffusion. Medications may also be metabolized by GI or hepatic enzymes before reaching systemic circulation. Prior to drug approval, oral bioavailability of medications is typically studied in patients with normal GI tracts. Bariatric surgery results in anatomical changes to the GI tract, and thus it can affect absorption and metabolism of oral medications. As mentioned previously, mechanisms include changes in gastric/intestinal pH, gastric emptying time/intestinal transit time, surface area available for absorption, active transport mechanisms for absorption, and first-pass metabolism.25 Possible effects on the GI tract for the various procedures can be found in TABLE 1.
Utilizing dosage forms that can help reduce issues associated with the anatomical changes of the GI tract is paramount for bariatric patients. This applies primarily to patients who have undergone RYGB surgery. Dosage forms that are designed for normal GI tracts such as those requiring disintegration (solid dosage forms), acidic environments (enteric-coated and delayed-release products), prolonged dissolution (extended-release products), and slower diffusion (oily solutions, suspensions) should be avoided, if possible.25 Aqueous solutions are the optimal dosage form because they are more rapidly absorbed and exempt from the disintegration and dissolution processes. If a drug is only available as a solid, the use of an immediate-release or crushing/chewing formulation is preferred. In addition, other routes of administration should be considered (if available) including IV, rectal, vaginal, intranasal, sublingual, transdermal, subcutaneous, and intramuscular.
An abundance of literature describing the proposed mechanisms of reduced medication absorption exists; however, research on the malabsorption of specific medications and patient outcomes associated with altered drug efficacy following bariatric surgery is sparse. The consensus of the available literature is that the effect of bariatric surgery on medication absorption appears to be drug-specific.14,26
Clinical Considerations
Pharmacists have the opportunity to change clinical outcomes and improve quality of life for bariatric patients through knowledge of available dosage forms and treatment alternatives, as well as principles of compounding. Pharmacists also can assess tolerability of dosage forms to evaluate a need for change to increase medication adherence. A retrospective pilot study described vitamin/nutrient supplement practices and assessed medication dosage formulations utilized in patients hospitalized with a history of bariatric surgery.27 Of the 133 admissions, at least one discrepancy was found in 117 of them. A multivitamin was ordered in only 33% of admissions and a calcium supplement in 7.7%. Inappropriate medication formulations were ordered in 61.5% of patients (34.7% non–immediate-release formulations, 25% enteric-coated formulations, and 40.3% both formulations). Fifty percent had inappropriate formulations continued upon discharge.27 Pharmacists can, therefore, have a significant role in medication reconciliation and discharge counseling to ensure appropriate drug prescribing.
From the findings of the above study, it is evident that pharmacists can play a role in improving patient care in community, ambulatory, and hospital settings by serving as a resource to healthcare providers, especially regarding recommended vitamin intake and dosage-form selection. Evidence of the positive impact of a multidisciplinary care team including a pharmacist has been demonstrated. A retrospective study evaluating hypertension and diabetes medication adjustments in post-LSG patients demonstrated the value of a multi-disciplinary approach that included an endocrinologist and clinical pharmacist.28 Another study demonstrated the use of pharmacy consultation services for dosage-form adjustments in the perioperative management of bariatric patients.29
Conclusion
In conclusion, bariatric patients are at increased risk of nutrient deficiencies as evident from available literature. These patients should receive adequate supplementation and follow-up to ensure adherence and identification of underlying deficiency. Malabsorption of medications due to surgical changes in the GI tract can be a potential problem in this population. With the limited data available for management of specific pharmacotherapies, it is imperative to assess the efficacy of pharmacotherapy on an individual basis. Because the number of patients receiving bariatric surgery is growing significantly, further procedure-specific and drug-specific studies are required to ensure that bariatric patients are receiving the intended benefits of pharmacotherapies prescribed. Research is especially needed in pharmacotherapy classes where there are significant consequences to decreased absorption, for example, of antimicrobials, anticoagulants, and chemotherapy. The utilization of a multidisciplinary team including a pharmacist is most beneficial in this patient population in order to maximize the long-term benefits of bariatric surgery for the individual patient.
REFERENCES
1. Ogden CL, Carroll MD, Kit BK, et al. Prevalence of obesity in the United States, 2009-2010. NCHS Data Brief. 2012;82:1-8.
2. Mechanick JI, Youdim A, Jones DB, et al. Clinical practice guidelines for the perioperative nutritional, metabolic, and nonsurgical support of the bariatric surgery patient–2013 update: cosponsored by American Association of Clinical Endocrinologists, the Obesity Society, and American Society for Metabolic & Bariatric Surgery. Surg Obes Relat Dis. 2013;9(2):159-191.
3. Sjostrom L, Sjostrom CD, Sullivan M, Wedel H. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351:2683-2693.
4. Sjostrom L, Narbro K, Sjostrom CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741-752.
5. Sjostrom L, Peltonen M, Jacobson P, et al. Bariatric surgery and long-term cardiovascular events. JAMA. 2012;307:56-65.
6. Bradley D, Magkos F, Klein S. Effects of bariatric surgery on glucose homeostasis and type 2 diabetes. Gastroenterology. 2012;143:897-912.
7. Steinbrook R. Surgery for severe obesity. N Engl J Med. 2004;350:1075-1079.
8. American Society for Metabolic and Bariatric Surgery (ASMBS). New procedure estimates for bariatric surgery: what the numbers reveal. Connect. May 2014. http://connect.asmbs.org/may-2014-bariatric-surgery-growth.html. Accessed April 16, 2016.
9. Sawaya RA, Jaffe J, Friedenberg L, Friedenberg FK. Vitamin, mineral, and drug absorption following bariatric surgery. Curr Drug Metab. 2012;13(9):1345-1355.
10. Franco JV, Ruiz PA, Palermo M, Ganger M. A review of studies comparing three laparoscopic procedures in bariatric surgery: sleeve gastrectomy, Roux-en-Y gastric bypass and adjustable gastric banding. Obes Surg. 2011;21(9):1458-1468.
11. Nocca D, Krawczykowsky D, Bomans B, et al. A prospective multi-center study of 163 sleeve gastrectomies: results at 1 and 2 years. Obes Surg. 2008;18:560-565.
12. Roerig JL, Steffen K. Psychopharmacology and bariatric surgery. Eur Eat Disorders Rev. 2015;23:463-469.
13. Wojcicki J, Wojciechowski G, Wojcicki M, et al. Pharmacokinetics of propranolol and atenolol in patients after partial gastric resection: a comparative study. Eur J Clin Pharmacol. 2000;56(1):75-79.
14. Padwal R, Brocks D, Sharma AM. A systematic review of drug absorption following bariatric surgery and its theoretical implications. Obes Rev. 2010;11(1):41-50.
15. Xanthakos SA. Nutritional deficiencies in obesity and after bariatric surgery. Pediatr Clin North Am. 2009;56:1105-1121.
16. Seaman JS, Bowers SP, Dixon P, Schindler L. Dissolution of common psychiatric medications in a Roux-en-Y gastric bypass model. Psychosomatics. 2005;46:250-253.
17. Stein J, Steir C, Raab H, Weiner R. Review article: the nutritional and pharmacological consequences of obesity surgery. Aliment Pharmacol Ther. 2014;40:582-609.
18. Brolin RE, Leung M. Survey of vitamin and mineral supplementation after gastric bypass and biliopancreatic diversion for morbid obesity. Obes Surg. 1999;9:150-154.
19. Sawhney P, Modi A, Jenkins TM, et al. Predictors and outcomes of adolescent bariatric support group attendance. Surg Obes Relat Dis. 2013;9(5):773-779.
20. Ponsky TA, Brody F, Pucci E. Alterations in gastrointestinal physiology after Roux-en-Y gastric bypass. J Am Coll Surg. 2005;201:125-131.
21. Rhode BM, Shustik C, Christou NV, et al. Iron absorption and therapy after gastric bypass. Obes Surg. 1999;9:17-21.
22. Tack J, Deloose E. Complications of bariatric surgery: dumping syndrome, reflux and vitamin deficiencies. Best Pract Res Clin Gastroenterol. 2014;28(4):741-749.
23. Madalosso CA, Gurski RR, Callegarie-Jacques SM, et al. The impact of gastric bypass on gastroesophageal reflux disease with morbid obesity: a prospective study based on the Montreal Consensus. Ann Surg. 2010;251:244-248.
24. Herbella FA, Vicentine FP, Del Grande JC, Patti MG. Postprandial proximal gastric acid pocket and gastric pressure in patients after gastric surgery. Neurogastroenterol Motil. 2011;23(1):52-55.
25. Buxton IL. Pharmacokinetics and pharmacodynamics: the dynamics of drug absorption, distribution, action, and elimination. In: Brunton LL, Lazo JS, Parker KL, eds. Goodman & Gilman’s The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill Companies; 2006:1-39.
26. Darwich AS, Henderson K, Burgin A, et al. Trends in oral drug bioavailability following bariatric surgery: examining the variable extent of impact on exposure of different drug classes. Br J Clin Pharmacol. 2012;74(5):774-787.
27. Lizer MH, Papageorgeon H, Glembot TM. Nutritional and pharmacologic challenges in the bariatric surgery patient. Obes Surg. 2010;20(12):1654-1659.
28. Tritsch AM, Bland CM, Hatzigeorgiou C, et al. A retrospective review of the medical management of hypertension and diabetes mellitus following sleeve gastrectomy. Obes Surg. 2015;25(4):642-647.
29. Silverman JB, Catella JG, Tavakkolizadeh A, et al. Bariatric surgery pharmacy consultation service. Obes Surg. 2011;21(9):1477-1481.
To comment on this article, contact rdavidson@uspharmacist.com.





