US Pharm. 2024;49(4):HS6-HS12.
ABSTRACT: Infective endocarditis (IE), also known as bacterial endocarditis, involves inflammation of the endocardium (the lining of the heart) and valves that is caused by bacteremia.1 Bacteremia allows for colonization of a vegetation that is formed when damaged endocardium results in platelet aggregation and activation of the coagulation cascade. Approximately 80% to 90% of IE cases are due to gram-positive streptococci, staphylococci, and enterococci, with Staphylococcus aureus responsible for about 30% of cases. Other organisms, including Haemophilus, Aggregatibacter, Cardiobacterium, Eikenella, and Kingella (HACEK), are less frequently the cause of infection. Although fungal endocarditis due to Candida and Aspergillus is rare, it can be fatal.1
Risk factors for children and young adults include heart birth defects such as valve malformations or septal defects, which allow blood to leak from one part of the heart to another. Risk factors for adults include dental procedures, particularly in those with cardiac valve disease, previous valve surgeries, heart transplant, calcium deposits in the mitral or aortic valve, congenital heart defects or a history of endocarditis, prolonged IV drug use, and illicit IV drug use.2,3 These risk factors can influence the type of bacteria causing IE. TABLE 1 identifies common pathogens and sources of infection.
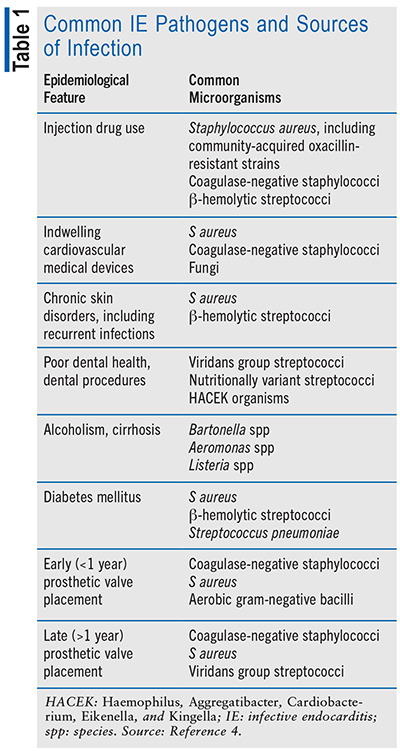
According to the American Heart Association, the annual incidence of IE is rare, with three to seven incidences per 100,000 person-years.4 More males than females are infected with endocarditis, with a ratio of 2:1, and the average age of IE patients is older than 65 years. The incidence in older patients is proposed to be related to prosthetic valves, indwelling cardiac devices, valvular disease, hemodialysis, and diabetes in this age group. Ten percent of IE is now related to recreational IV-drug use.1
CLINICAL PRESENTATION OF IE
IE can present as an acute or subacute infection. Acute infections present and progress rapidly. They are characterized by high fevers, rigors, and sepsis in healthy hearts. Subacute endocarditis is delayed and presents with nonspecific symptoms, including weight loss, fatigue, and dyspnea over weeks to months, often in patients with preexisting heart disease. Subacute cases are usually due to penicillin-sensitive viridans group Streptococcus (VGS), while S aureus is responsible for acute IE. Unfortunately, even after treatment, for patients who present with acute IE, there is an increased chance of death due to cardiac failure within weeks to months, while those who have subacute IE rarely have severe cardiac damage.5
DIAGNOSIS
Many patients with endocarditis present with nonspecific symptoms that prompt a broad medical workup. Patients presenting with fevers and chills will likely undergo a sepsis protocol evaluation. If patients present with symptoms like chest pain or dyspnea, they will likely undergo a workup for acute coronary syndrome, pulmonary embolism, and pneumonia. A 12-lead ECG will evaluate for ischemia, dysrhythmias, or structural disease, but an ECG in IE will generally appear normal. Further initial workup may include a chest x-ray and/or CT scan depending on a patient’s symptoms and subsequent differential diagnosis. Broad laboratory workup will be conducted, and a complete blood count with leukocytosis will indicate an underlying infection. Erythrocyte sedimentation rate and C-reactive protein have been found to be increased in 60% of IE cases.1
The diagnosis of IE has been based upon microbiologic and echocardiographic evidence recommendations from the Modified Duke Criteria, last updated in 2000.6 In 2023, the International Society for Cardiovascular Infectious Diseases suggested significant updates to the Duke Criteria due to changes in microbiology, diagnostics, epidemiology, and treatment. Updates were suggested in three areas: pathologic criteria, major clinical criteria, and minor clinical criteria. Overall, clinical criteria for diagnosis remain unchanged and require 1) two major criteria, 2) one major criterion and three minor criteria, or 3) five minor criteria (see SIDEBAR 1).

Updates to Pathologic Criteria
Pathologic criteria are used to diagnose definitive IE. In the past, pathologic criteria required the identification of microorganisms or histopathologic evidence in operative or postmortem specimens.7 The 2020 pathologic criteria for definitive endocarditis included 1) microorganisms demonstrated by culture or histologic examination of a vegetation, a vegetation that has embolized, or an intracardiac abscess specimen or 2) pathologic lesions, vegetation, or intracardiac abscess confirmed by histologic examination showing active endocarditis.6 The 2023 update provided clarity and broadened these criteria to include 1) microorganisms identified in the context of clinical signs of active endocarditis in a vegetation, from cardiac tissue, from an explanted prosthetic valve or sewing ring, from an ascending aortic graft (with concomitant evidence of valve involvement), from an cardiovascular intracardiac implantable electronic device (CIED), or from an arterial embolus; or 2) active endocarditis (may be acute or subacute/chronic) identified in or on a vegetation, from cardiac tissue, from an explanted prosthetic valve or sewing ring, from an ascending aortic graft (with concomitant evidence of valve involvement), from a CIED, or from an arterial embolus.7
Updates to Major Clinical Criteria
These criteria have been updated to include three subsections: Microbiologic Major Criteria, Imaging Major Criteria, and Surgical Major Criteria, which is a 2023 suggested addition. For the Microbiologic Major Criteria suggestions, blood cultures removed requirements for timing and separate venipunctures for blood cultures, but this is still strongly recommended when possible. Definitions of typical organisms were added to include Staphylococcus lugdunensis and Enterococcus faecalis, among multiple others. New criteria were added for fastidious pathogens like Bartonella species. The Imaging Major Criteria have been updated to include cardiac CT, as its findings are equivalent to ECG, as well as positron emission tomography (PET) with [18F]-fluorodeoxyglucose PET/CT imaging. Finally, Surgical Major Criteria were added, as intraoperative inspection can be considered as a major criterion if cardiac imaging or histopathology is not available.7 TABLE 2 includes a summary of the updates for this section.
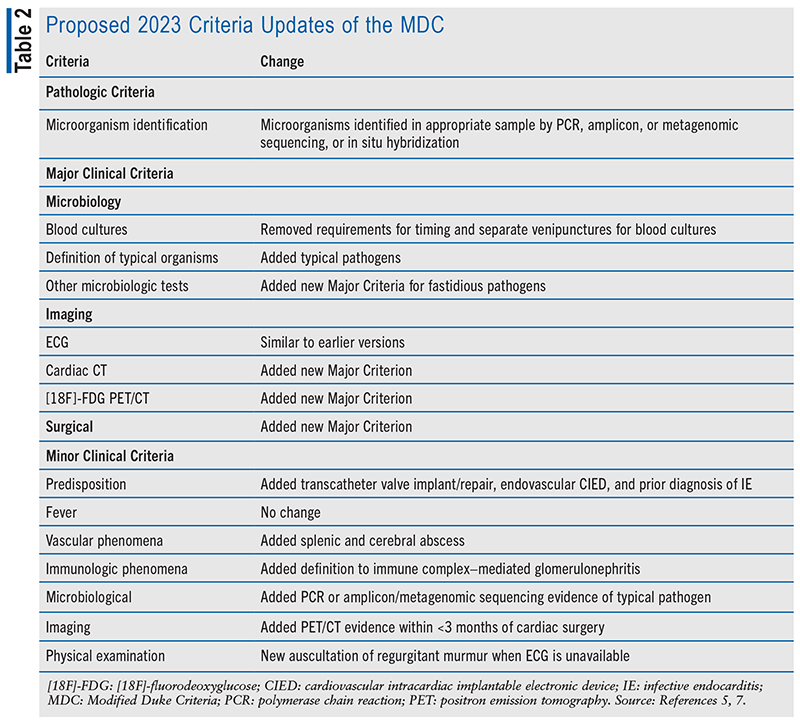
Updates to Minor Clinical Criteria
The minor clinical criteria for diagnosis span seven categories (see TABLE 2). Changes have been made to several of these categories. Two additional factors, previous history of IE and CIED, have been added under Predisposition. For Vascular Phenomena, splenic and cerebral abscess are included as additional examples in the category. The definition for immune complex–mediated glomerulonephritis has been incorporated into Immunologic Phenomena. Microbiologic evidence added the alternative of polymerase chain reaction or amplicon/metagenomic sequencing evidence of typical pathogen. Imaging Criteria was suggested as an addition, as it did not exist previously and includes PET/CT evidence within <3 months of cardiac surgery. Lastly, Physical Examination Criteria now include new auscultation of regurgitant murmur if ECG is not available, based on expert opinion.7 TABLE 2 includes a summary of updates for this section.
MANAGEMENT OF IE
The goal of treatment in IE is eradication of infection, including sterilization of vegetations. However, IE has unique characteristics that make treatment challenging, including high bacterial burdens and the potential for biofilm formation. Additionally, host factors and the ability of antimicrobial agents to achieve adequate concentrations within vegetations contribute to the complexity of management. Because of this, certain therapeutic principles are important to consider, and management typically requires prolonged courses of appropriate antimicrobial therapy.4 While previously thought to require prolonged durations of parenteral therapy, new literature is emerging on the effectiveness of transitioning to oral therapy.8
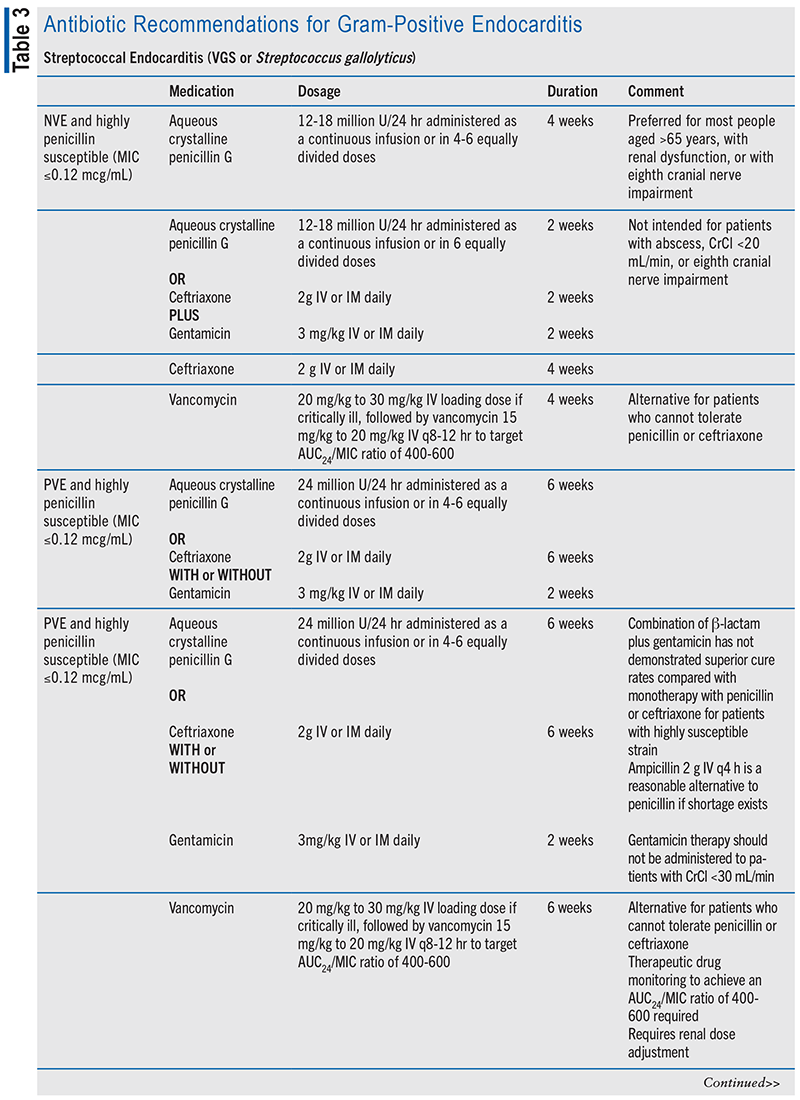
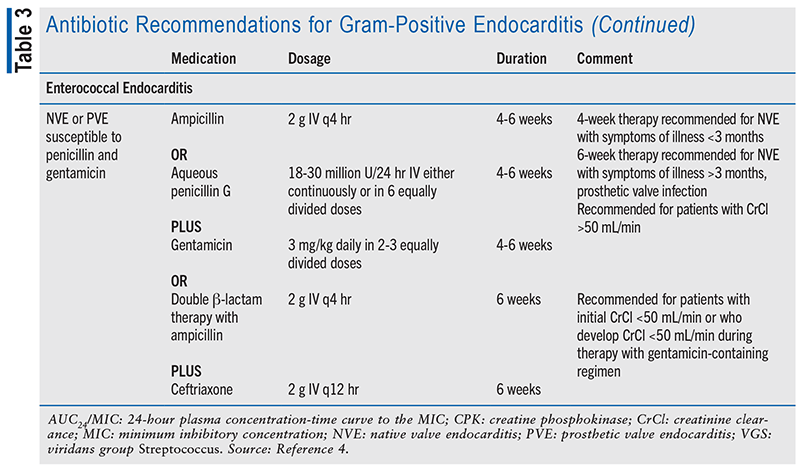
As per the IE guidelines, infectious diseases consultation is recommended for all patients with IE to assist with selecting optimal empiric therapy and timing of therapy. Additionally, in complicated cases, further evaluation may be required to determine if surgical intervention is needed.4 Definitive antimicrobial therapy selection for IE depends on multiple factors such as the organism isolation, and presence of native valve endocarditis (NVE) or prosthetic valve. TABLE 3 provides an overview of treatment recommendations for gram-positive endocarditis.
Streptococcal Endocarditis
Streptococcal endocarditis is typically caused by VGS species or Streptococcus gallolyticus (formerly known as Streptococcus bovis), and antibiotic treatment regimens are determined based on penicillin-susceptibility data. In patients with NVE and highly penicillin-susceptible infection, defined as a penicillin minimum inhibitory concentration (MIC) of ≤0.12 mcg/mL, aqueous crystalline penicillin G or ceftriaxone therapy for 4 weeks is recommended. Alternatively, if aqueous crystalline penicillin G or ceftriaxone is used in combination with gentamicin, a shorter treatment duration of 2 weeks may be utilized. However, this shorter treatment duration is not recommended for patients with cardiac abscess, creatinine clearance (CrCl) <20 mL/min, or impaired eighth cranial nerve function. In patients who are unable to tolerate penicillin or ceftriaxone therapy, a 4-week course of vancomycin may be used instead.9-11 Penicillin-resistant streptococcal endocarditis is relatively uncommon, however. Antibiotic treatment recommendations for cases of resistance can be found in the IE guidelines in TABLE 3.4
In patients with endocarditis of prosthetic valve caused by highly penicillin-susceptible VGS or S gallolyticus (MIC ≤0.12 mcg/mL), longer treatment durations are recommended compared with native valve infection. Recommended treatment regimens include aqueous crystalline penicillin or ceftriaxone for 6 weeks that may or may not be combined with gentamicin for 2 weeks. Vancomycin should only be used in patients who cannot tolerate penicillin, ceftriaxone, or gentamicin.4
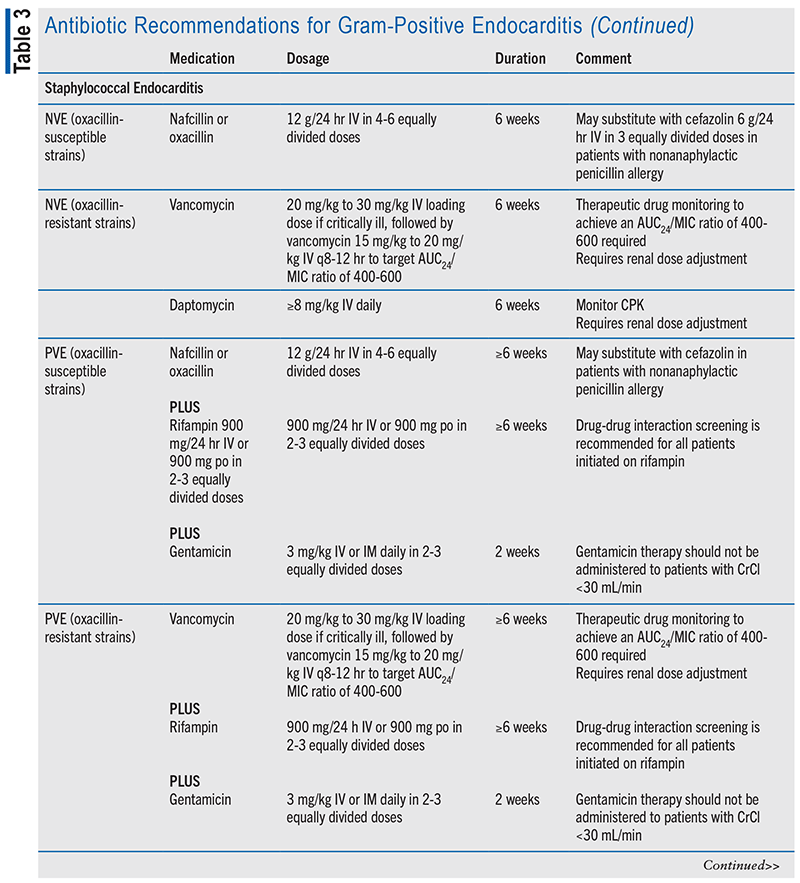
Staphylococcal endocarditis is most commonly caused by coagulase-positive staphylococci, specifically S aureus. In high-income countries, S aureus is the most common cause of IE overall, accounting for 30% of cases.1 Coagulase-negative staphylococci, such as Staphylococcus epidermidis, S lugdunensis, and various other species, are also a prominent cause of IE. Both coagulase-positive and coagulase-negative staphylococci are associated with native and prosthetic valve endocarditis. Therefore, both pathogens should be considered in a patient with suspected IE and a Gram stain suggesting staphylococci.12,13
Antibiotic treatment regimens in patients with staphylococcal infection are determined based on presence of native or prosthetic valve. In patients with a native valve and oxacillin-susceptible strains, nafcillin or oxacillin is recommended for 6 weeks. Cefazolin may be used as an alternative in patients with a nonanaphylactoid penicillin allergy. In patients with a native valve and oxacillin resistant strains, vancomycin or daptomycin is recommended for 6 weeks.4
Treatment of prosthetic valve IE caused by staphylococci requires combination antibiotic therapy. In cases of methicillin-susceptible infection, oxacillin or nafcillin should be combined with rifampin for at least 6 weeks, plus gentamicin for 2 weeks. In cases of methicillin-resistant infection, vancomycin should be used instead of oxacillin or nafcillin.4
Enterococcal Endocarditis
Enterococcal endocarditis is most commonly caused by E faecalis or Enterococcus faecium and accounts for 13% to 18% of all endocarditis cases.14 Both NVE and prosthetic valve infection caused by enterococcus strains susceptible to penicillin and gentamicin can be treated with either ampicillin or aqueous penicillin G plus gentamicin or with double β-lactam therapy of ampicillin plus ceftriaxone. Double β-lactam therapy is preferred for individuals with CrCl <50 mL/min and in those whose CrCl falls below 50 mL/min while on a gentamicin-containing regimen. Treatment recommendations for resistant strains can be found in the guidelines.4
Gram-Negative Endocarditis
Gram-negative endocarditis accounts for 1% to 10% of all cases and is typically caused by organisms in the HACEK group.15 Currently, the treatment of choice for HACEK endocarditis is third-generation cephalosporins such as ceftriaxone 2 g IV daily for 4 weeks in NVE or 6 weeks in prosthetic valve infection. Fluoroquinolones may also be considered in patients whom β-lactam therapy is not appropriate.4,15
Additional information on management of IE including treatment of culture-negative endocarditis, fungal endocarditis, indications for surgery, and role of antimicrobial prophylaxis can be found in the guidelines.4
ROLE OF THE PHARMACIST
Pharmacists play a key role in the management of IE. The recent Wiki Guideline consensus statement on diagnosis and management of IE notes that management by multidisciplinary teams, including pharmacy, may be beneficial to the management of IE.8 Pharmacists can assist with optimizing antibiotic regimens and dosing, recognizing, and managing adverse effects and drug interactions, assisting with transition to oral regimens, and identifying patients who may be candidates for IE prophylaxis.
CONCLUSION
IE diagnosis and management are multifactorial and often complex. The 2023 updates to the modified Duke Criteria consider changes in microbiology, diagnostics, epidemiology, and treatment of IE, allowing for improved diagnosis of IE. Antimicrobial treatment of IE typically depends on the isolated organism, its susceptibility, and presence of a native or prosthetic valve. Treatment durations are usually prolonged, and combination antibiotic therapy may be needed. Pharmacists play an important role in assisting with management of these complex regimens.
REFERENCES1. Yallowitz AW, Decker LC. Infectious endocarditis. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2024 Jan-.
2. American Heart Association. Heart valves and infective endocarditis. May 8, 2020. www.heart.org/en/health-topics/heart-valve-problems-and-disease/heart-valve-problems-and-causes/heart-valves-and-infective-endocarditis. Accessed March 6, 2024.
3. American Heart Association. Infective endocarditis. March 4, 2021. www.heart.org/en/health-topics/infective-endocarditis. Accessed March 6, 2024.
4. Baddour LM, Wilson WR, Bayer AS, et al. Infective endocarditis in adults: diagnosis, antimicrobial therapy, and management of complications. Circulation. 2015;132:1435-1486.
5. Ibrahim AM, Siddique MS. Subacute bacterial endocarditis prophylaxis. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2024 Jan-.
6. Li JS, Sexton DJ, Mick N, et al. Proposed modifications to the Duke criteria for the diagnosis of infective endocarditis. Clin Infect Dis. 2000;30(4): 633-638.
7. Fowler VG, Durack DT, Selton-Suty C, et al. The 2023 Duke–International Society for Cardiovascular Infectious Diseases Criteria for Infective Endocarditis: updating the Modified Duke Criteria. Clin Infect Dis. 2023;77(4): 518-526.
8. McDonald E, Aggrey G, Aslan AT, et al. Guidelines for diagnosis and management of infective endocarditis. JAMA Netw Open. 2023;6(7):e2326366.
9. Francioli P, Etienne J, Hoigné R, et al. Treatment of streptococcal endocarditis with a single daily dose of ceftriaxone sodium for 4 weeks: efficacy and outpatient treatment feasibility. JAMA. 1992;267:264-267.
10. Wilson WR. Ceftriaxone sodium therapy of penicillin G–susceptible streptococcal endocarditis. JAMA. 1992;267:279-280.
11. Sexton DJ, Tenenbaum MJ, Wilson WR, et al. Ceftriaxone once daily for four weeks compared with ceftriaxone plus gentamicin once daily for two weeks for treatment of endocarditis due to penicillin-susceptible streptococci: Endocarditis Treatment Consortium Group. Clin Infect Dis. 1998;27:1470-1474.
12. Wang A, Athan E, Pappas PA, et al. International Collaboration on Endocarditis-Prospective Cohort Study Investigators. Contemporary clinical profile and outcome of prosthetic valve endocarditis. JAMA. 2007;297:1354-1361.
13. Chu VH, Woods CW, Miro JM, et al. International Collaboration on Endocarditis-Prospective Cohort Study Group. Emergence of coagulase-negative staphylococci as a cause of native valve endocarditis. Clin Infect Dis. 2008;46:232-242.
14. Danneels P, Hamel JF, Picard L, et al. EFEMER study group. Impact of Enterococcus faecalis endocarditis treatment on risk of relapse. Clin Infect Dis. 2023;76(2):281-290.
15. Bouza E, Muñoz P, Burillo A. Gram-negative endocarditis: disease presentation, diagnosis and treatment. Curr Opin Infect Dis. 2021;34(6):672-680.
16. Rybak MJ, Le J, Lodise TP, et al. Therapeutic monitoring of vancomycin for serious methicillin-resistant Staphylococcus aureus infections: a revised consensus guideline and review by the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. Am J Health Syst Pharm. 2020;77(11):835-864.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






