US Pharm. 2015;40(10)(Diabetes suppl):3-7.
ABSTRACT: Afrezza is a rapid-acting inhaled insulin that has been FDA-approved as an option for insulin therapy in adults with type 1 or type 2 diabetes. Currently, it is the only inhaled insulin on the market, as an earlier product was discontinued. Spirometry testing is required prior to the initiation of therapy due to the risk of acute bronchospasm. The most common adverse effects with Afrezza include hypoglycemia, cough, throat pain or irritation, and headache. Inhaled insulin may be an option for a certain niche of patients. Pharmacists are able to provide education on dose conversion and proper inhaler usage.
Diabetes affects approximately 29.1 million Americans and is considered the seventh leading cause of death in the United States.1 Research and development have led to the expansion of available treatment options for patients with diabetes mellitus. These options include oral medications, injectables, and insulin in addition to lifestyle modifications. A study in adults diagnosed with diabetes showed that 2.9 million (14%) were treated with insulin alone, while 3.1 million (14.7%) were treated with both insulin and oral medications.2
Insulin Background
Insulin is vital to regulate blood glucose. Since its discovery in 1923, the types of insulin developed have included bovine, porcine, human, and analogue preparations.3 There have been many changes to the mode of delivery of insulin, including needle and syringe, pen injection, continuous SC insulin infusion (insulin pumps), and now inhaled insulin. Additionally, there is ongoing research to develop a smart insulin patch.4 The ideal insulin formulation is one that closely mimics human physiology with minimal hypoglycemia and is convenient and affordable to patients.5-7
Even though insulin can be important in controlling diabetes, nonadherence with insulin regimens is associated with higher rates of diabetes-related hospitalizations and higher A1C levels.8 There are many factors related to insulin nonadherence, including injection phobia, negative impact on work and social life, inconvenience in daily routine, and concerns regarding long-term use. In a recent study, some patients believed that requiring insulin was associated with a lack of ability to care for oneself or that taking insulin resulted in diabetes-related complications such as blindness, renal failure, and amputations.9 It is critical that healthcare providers identify these concerns and establish a treatment regimen that works for the individual. Inhaled insulin may be an option for a certain niche of patients.
Inhaled Insulin
Inhaled insulin has been studied since 1924.10 The first dry-powder insulin product approved by the FDA was Exubera in 2006.11 However, the manufacturer (Pfizer) withdrew its product from the market the following year due to low sales.12 The inhaler was larger than inhalers traditionally used by patients with asthma and required patients to schedule additional office visits with providers for spirometry testing every 6 months. Additionally, the cost of Exubera was higher than that of available SC insulins.12
In 2014, MannKind Corporation’s inhaled insulin product Afrezza received FDA approval.13 Currently, it is the only inhaled insulin on the market and is distributed by Sanofi-Aventis. Afrezza is a rapid-acting insulin indicated to improve glycemic control in adults with type 1 or type 2 diabetes. It must be used in combination with long-acting insulin in patients with type 1 diabetes. Afrezza is not currently approved for pediatric use and is designated as Pregnancy Category C.14
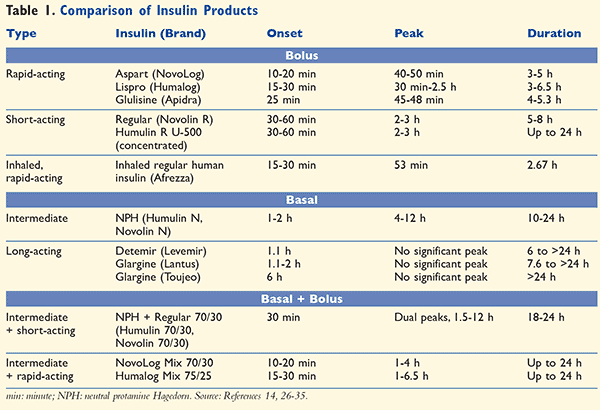
Insulin products vary by onset, peak, and duration, and each serves a role in the management of diabetes. A comparison of available insulin products is shown in TABLE 1. The intraindividual variability of early insulin action varies from 25% to 30% for short-acting SC insulin formulations.15 Afrezza is regular human insulin, so once it is absorbed in the pulmonary tissue, metabolism and excretion are similar to those of other regular human insulins.14
Technosphere Technology
Afrezza utilizes Technosphere technology, a novel delivery system that makes the pulmonary delivery of insulin possible. It is based on Technosphere particles that form by the assembly of fumaryl diketopiperazine (FDKP) crystals.16-18 The FDKP molecules form in a diameter of 2 to 3 micrometers that are lyophilized into a dry powder for inhalation.16,17 Upon inhalation, Technosphere insulin is distributed evenly and rapidly dissolves in the lung tissue. At physiological pH, the insulin is absorbed through the pulmonary epithelium into systemic circulation.16,17 The FDKP molecules that reach circulation are cleared and excreted without metabolites.17 Technosphere insulin achieves maximum concentration within 15 minutes and is eliminated faster than SC insulin.14,18
Efficacy
There have been two products approved and brought to market in a continued effort to develop an effective inhaled insulin product. Initially, for the 2006 FDA approval of Exubera, Rosenstock et al conducted a trial of patients with type 1 or type 2 diabetes randomized to inhaled or SC insulin.19 At the conclusion of a 12-week study, the change in A1C was similar in both treatment arms in type 1 diabetes with inhaled insulin versus SC injection (reduction of -0.69% vs. -0.85%). In the type 2 diabetes sample, inhaled insulin led to a reduction in A1C of -0.61%, and SC insulin led to a reduction of -0.79%.19
The efficacy of inhaled insulin (Exubera) was also investigated in patients with type 2 diabetes not controlled with diet and exercise.20 Study participants were randomized to receive inhaled insulin prior to meals or rosiglitazone twice daily in addition to diet and exercise. Dietary education sessions were conducted during a 4-week lead-in phase. Patients were also instructed to exercise 30 minutes per day for at least 3 days per week. After completing the 12-week treatment phase, 82.7% of patients in the inhaled insulin group reached the study goal of A1C <8% compared to 58.2% of those in the oral therapy group. However, more patients in the inhaled insulin treatment arm experienced episodes of hypoglycemia.20
In 2008, Rosenstock et al compared Technosphere insulin with powder placebo in addition to oral antidiabetic agents in 107 patients with type 2 diabetes for 12 weeks.21 The overall reduction in A1C was statistically significant between the Technosphere insulin and placebo, -0.7% and -0.3%, respectively. The incidence of hypo-glycemia in the Technosphere insulin group was similar to placebo.21
In an open-label, randomized study, patients with type 2 diabetes poorly controlled on insulin therapy with or without oral medication were assigned to 1 year of treatment with basal insulin and prandial inhaled insulin (Afrezza) or twice-daily premixed insulin.22 The premixed insulin was 70% insulin aspart protamine and 30% insulin aspart suspension, and the basal insulin used was insulin glargine. The Technosphere inhaled insulin dose was provided using 15- and 30-unit cartridges. At the end of the study period, the number of patients with an A1C of ≤7% was similar in both treatment groups, with less weight gain in the inhaled insulin treatment group. Hypoglycemic events were reported more in the premixed insulin treatment group (49%), compared to 31% in those treated with inhaled insulin therapy.22
According to a recent study, patients with type 2 diabetes who are poorly controlled with oral medications may benefit from adding Afrezza at mealtime. The A1C reduction was 0.8% with the inhaled Technosphere insulin compared to a 0.4% reduction in the inhaled placebo arm.23
Role in Therapy
The 2015 guidelines of the American Association of Clinical Endocrinologists (AACE) state that when control of postprandial hyperglycemia is needed, consideration should be given to rapid-acting insulins (the analogues lispro, aspart, glulisine, and inhaled insulin) over regular human insulin because the analogues have a rapid onset and offset of action, leading to a lower incidence of hypoglycemia. In the management of type 1 diabetes, multiple daily injections are recommended, which may involve one to two SC daily basal insulin injections to control glucose levels between meals and overnight, and SC injections of prandial insulin or inhaled insulin before each meal to control blood glucose.24
Adverse Effects and Precautions
The most common adverse effect with all forms of insulin, including inhaled insulin, is hypoglycemia. The delivery mechanism of insulin inhalation powder also requires healthcare professionals to be aware of pulmonary adverse effects. In order to assess if a patient is an appropriate candidate for inhaled insulin, a thorough medical history should be completed. Prior to initiation, patients should undergo spirometry testing (forced expiratory volume in one second [FEV1]) to identify potential lung disease. Afrezza has a boxed warning for risk of acute bronchospasm in patients with chronic lung disease, and inhaled insulin is contraindicated in patients with asthma or chronic obstructive pulmonary disease (COPD). In a clinical trial of Afrezza, broncho-constriction occurred in 29% of patients diagnosed with asthma.14
Afrezza has been associated with a decline in pulmonary function. Patients with asthma had a mean decline in FEV1 of 400 mL within 15 minutes after a single dose, while a mean decline in FEV1 of 200 mL occurred within 18 minutes of administration in COPD patients.14 Patients without chronic lung disease experienced a decline of FEV1 of 40 mL that occurred within 3 months of initiation and persisted during the entire treatment duration (up to 2 years). The long-term effects of Afrezza on pulmonary function have not been studied beyond 2 years. The manufacturer recommends spirometry assessment at baseline, within the first 6 months of therapy, and annually thereafter. Providers may consider discontinuation of therapy if patients have a decline of ≥20% in FEV1 from baseline.14
In clinical trials, there were two cases of lung cancer. One case occurred in controlled trials, and the other in uncontrolled trials. The benefits and risks of Afrezza should be considered in patients with lung cancer or at risk for lung cancer.14
The most common adverse effects with Afrezza included cough, throat pain or irritation, and headache. The FDA has deemed necessary a Risk Evaluation and Mitigation Strategy (REMS) to ensure that the benefits of therapy outweigh the risk of bronchospasm in the target population.14
Counseling Points
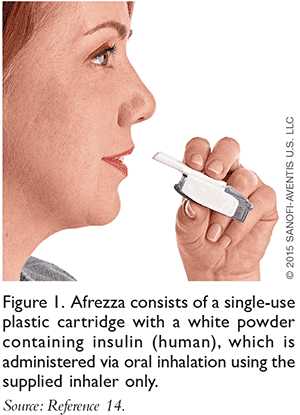
When a prescription for Afrezza is dispensed from the pharmacy, it will include inhalers for administration. This new formulation is a smaller design (FIGURE 1) that will fit in the palm of the hand compared to its predecessor, Exubera, which was similar in size to an inhaler spacer device. Afrezza single-use cartridges are available in three colors (blue, green, and yellow), each corresponding to the dose of insulin it contains (4, 8, and 12 units). The prescription inhaler may be used for up to 15 days after first use but must then be discarded, and the patient will need to start with a new inhaler. Patients should be counseled not to wash the inhaler for reuse.14
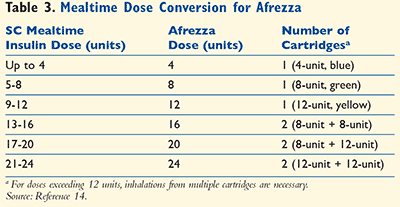
Afrezza must be refrigerated prior to first use. After the foil packaging is opened, its contents must be used within 10 days. The inhaler may also be stored at refrigeration temperature, but prior to use the patient should allow the inhaler to reach room temperature. Insulin cartridges should also be left at room temperature at least 10 minutes prior to medication administration. TABLE 2 discusses storage and expiration information. TABLE 3 contains the dose conversion of rapid mealtime insulin to the Afrezza dose.14

Patients should make sure they have enough cartridges on hand for the prescribed dose prior to administration. If the prescription is for 12 units, the patient will need both a blue (4-unit) and a green (8-unit) cartridge or a single yellow (12-unit) cartridge. The cartridge is then loaded into the provided inhaler. The inhaler will snap closed, and the mouthpiece will have to be removed prior to administering the dose. The inhaler will also need to be held level after loading to avoid losing the medication powder. The patient must exhale completely prior to using the inhaler. Patients should be instructed to keep the head level and place the mouthpiece in the mouth and tilt the inhaler down toward the chin. The patient must then close his or her lips to form a seal as with any other inhaler and inhale deeply. The patient can then remove the inhaler, hold his or her breath, and then exhale and begin breathing normally. As a last step, the patient must replace the mouthpiece cover, open the inhaler, and remove the used cartridge for disposal.18
As with any new prescription for insulin therapy, patients should be counseled on management of hypoglycemic and hyperglycemic episodes. The pharmacist should review with the patient that dose adjustments may be needed in the event of dietary changes and changes in level of physical activity. Inhaled insulin is not recommended in patients who currently smoke or have recently quit. This is an opportunity for pharmacists and other healthcare professionals to offer smoking cessation techniques to patients.
Future Implications
With clinical practice guidelines recommending mealtime insulin when glucose levels are above goal, this formulation of inhaled insulin will likely be useful for specific patient populations (TABLE 4).14,24 In addition, an inhaled insulin option may offer more independence to a patient who has limited vision or who lives alone and may need to self-administer insulin. There would be less manipulation of devices (i.e., vial with syringe or attaching a pen needle to the pen) and less potential for overdose due to decreased visual acuity, as premeasured insulin cartridges are provided with the inhaler. Removing the injection route of insulin administration may also lead to greater patient acceptance of insulin therapy. In some cases, patients relate an injection to a past experience in which they received a medication with a different type of needle.25

Patient preference and insurance formulary coverage will also play a major role in selection of inhaled insulin. Spirometry testing is required before the initiation of Afrezza therapy, in addition to a review of medical history and a physical examination.14 Patients and prescribers will need to be educated on this updated formulation and its requirements for continued monitoring. Patients may be opposed to further testing and the cost associated with it or to an additional office visit for a physical examination. Insurance companies will likely require a prior authorization for this medication if it has been added to their formulary, since there are other rapid-acting insulin formulations available that have been on the market for a longer period of time. It may take some insurers until next year to list Afrezza on their formularies. Another variable will be the type and level of insurance coverage provided to or selected by the patient.
Conclusion
Inhaled insulin may offer a therapy option to patients who have never considered taking mealtime insulin for various reasons. The inhaled option may change the perception of and fear associated with insulin therapy, as it does remove the need for injection and is a smaller device. It is imperative that patients receive education at the prescriber level and dispensing site to ensure that the medication is administered correctly and adverse events are avoided.
REFERENCES
1. American Diabetes Association. Statistics about diabetes. June 10, 2014. www.diabetes.org/diabetes-basics/statistics. Accessed July 1, 2015.
2. CDC. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Atlanta, GA: U.S. Department of Health and Human Services; 2014. www.cdc.gov/diabetes/pubs/statsreport14/national-diabetes-report-web.pdf. Accessed July 1, 2015.
3. Gualandi-Signorini A, Giorgi G. Insulin formulations—a review. Eur Rev Med Pharmacol Sci. 2001;5:73-83.
4. Gu Z. Smart insulin patch could replace painful injections for diabetes. NC State News. June 23, 2015. https://news.ncsu.edu/2015/06/gu-smart-patch-2015. Accessed July 1, 2015.
5. Aronoff S, Berkowitz K, Shreiner B, et al. Glucose metabolism and regulation: beyond insulin and glucagon. Diabetes Spectr. 2004;17:183-190.
6. Del Prato S, Marchetti P, Bonadonna R. Phasic insulin release and metabolic regulation in type 2 diabetes. Diabetes. 2002;51(suppl):S109-S116.
7. Herbst K, Hirsch I. Insulin strategies for primary care providers. Clin Diabetes. 2002;20(1):11-17.
8. Peyrot M, Rubin RR, Kruger DF, Travis LB. Correlates of insulin injection omission. Diabetes Care. 2010;33:240-245. http://care.diabetesjournals.org/content/33/2/240.full.pdf+html. Accessed July 1, 2015.
9. Karter AJ, Subramanian U, Saha C, et al. Barriers to insulin initiation. Diabetes Care. 2010;33(4):733-735. www.ncbi.nlm.nih.gov/pmc/articles/PMC2845015. Accessed July 1, 2015.
10. Siekmeier R, Scheuch G. Inhaled insulin—does it become reality? J Physiol Pharmacol. 2008;59:81-113.
11. FDA approves first ever inhaled insulin combination product for treatment of diabetes. FDA news release. January 27, 2006. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/2006/ucm108585.htm. Accessed July 1, 2015.
12. Berenson A. Weak sales prompt Pfizer to cancel diabetes drug. NY Times. October 19, 2007. www.nytimes.com/2007/10/19/business/19pfizer.html?_r=0. Accessed July 1, 2015.
13. FDA approves Afrezza to treat diabetes. FDA news release. June 27, 2014. www.fda.gov/NewsEvents/Newsroom/Press-Announcements/ucm403122.htm. Accessed July 1, 2015.
14. Afrezza (insulin human inhalation powder) package insert. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; July 2015. http://products.sanofi.us/afrezza/afrezza.pdf. Accessed August 28, 2015.
15. Heinemann L. Variability of insulin absorption and insulin action. Diabetes Technol Ther. 2002;4:673-682.
16. Pfutzner A, Forst T. Pulmonary insulin delivery by means of the Technosphere drug carrier mechanism. Expert Opin Drug Deliv. 2005;2:1097-1106.
17. Richardson P, Boss A. Technosphere insulin technology. Diabetes Technol Ther. 2007;9(suppl 1):S65-S72.
18. Neumiller JJ, Campbell RK, Wood LD. A review of inhaled Technosphere insulin. Ann Pharmacother. 2010;44: 1231-1239.
19. Rosenstock J, Cappelleri JC, Bolinder B, et al. Patient satisfaction and glycemic control after 1 year with inhaled insulin (Exubera) in patients with type 1 or type 2 diabetes. Diabetes Care. 2004;27:1318-1323.
20. Defronzo R, Bergenstal R, Cefalu W, et al. Efficacy of inhaled insulin in patients with type 2 diabetes not controlled with diet and exercise. Diabetes Care. 2005;28:1922-1928.
21. Rosenstock J, Bergenstal R, DeFronzo RA, et al. Efficacy and safety of Technosphere inhaled insulin compared with Technosphere powder placebo in insulin-naïve type 2 diabetes suboptimally controlled with oral agents. Diabetes Care. 2008;31:2177-2182.
22. Rosenstock J, Lorber D, Gnudi L, et al. Prandial inhaled insulin plus basal insulin glargine versus twice daily biaspart insulin for type 2 diabetes: a multicenter randomized trial. Lancet. 2010;375:2244-2253.
23. Rosenstock J, Franco D, Korpachev V, et al. Inhaled Technosphere insulin versus inhaled Technosphere placebo in insulin-naïve subjects with type 2 diabetes inadequately controlled on oral antidiabetes agents. Diabetes Care. 2015 Aug 7 [Epub ahead of print]. http://bit.ly/1Tr1uMs. Accessed September 7, 2015.
24. Handelsman Y, Bloomgarden ZT, Grunberger G, et al. American Association of Clinical Endocrinologists and American College of Endocrinology—clinical practice guidelines for developing a diabetes mellitus comprehensive care plan—2015. Endocr Pract. 2015;21(suppl 1):1-87. www.aace.com/files/dm-guidelines-ccp.pdf. Accessed July 1, 2015.
25. Funnell MM. Overcoming barriers to the initiation of insulin therapy. Clin Diabetes. 2007;25(1):36-38.
26. NovoLog (insulin aspart) package insert. Princeton, NJ: Novo Nordisk Inc; April 2014.
27. Humalog (insulin lispro) package insert. Indianapolis, IN: Eli Lilly and Co; November 2013.
28. Apidra (insulin glulisine) package insert. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; May 2014.
29. Novolin R (human insulin [rDNA origin]) package insert. Princeton, NJ: Novo Nordisk Inc; March 2013.
30. Humulin R U-500 (concentrated human insulin [rDNA origin]) package insert. Indianapolis, IN: Eli Lilly and Co; September 2014.
31. Levemir (insulin detemir) package insert. Princeton, NJ: Novo Nordisk Inc; October 2013.
32. Lantus (insulin glargine) package insert. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; October 2013.
33. NovoLog Mix 70/30 (insulin aspart protamine and suspension and 30% insulin aspart injection) package insert. Princeton, NJ: Novo Nordisk Inc; June 2014.
34. Humalog Mix 75/25 (75% insulin lispro protamine suspension and 25% insulin lispro injection) package insert. Indianapolis, IN: Eli Lilly and Co; November 2013.
35. PL Detail-Document. Comparison of insulins and injectable diabetes meds. Pharmacist’s Letter/Prescriber’s Letter. March 2015. http://pharmacistsletter.therapeuticresearch.com. Accessed September 7, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





