Intellectual disability (ID) is a term describing limitations to a person’s ability to learn at an expected level and function in daily life.1 ID was previously referred to as mental retardation in the United States, and as learning disability in the United Kingdom; it is characterized by significantly subaverage intellectual functioning.2,3 The health status of people with ID is influenced by the degree of intellectual limitation and specific syndromic associations.2 Developmental disorders that affect the brain and give rise to ID often affect other body systems as well.2
Although people with ID exhibit considerable morbidity, the numbers surviving into old age are rapidly increasing.2 As the number of people with ID increases, and they live and work in the community and participate in all aspects of community life, there is a growing need for a well-trained workforce to provide the services that enable these individuals to carry out those activities.4
Classification of ID is comprehensive and is expressed as 1) an intelligence quotient (IQ) less than 70, two standard deviations below the mean IQ of the general population (IQ of 100), and 2) limitations of more than two of the following: communication, self-direction, social skills, self-care, use of community resources, and maintenance of personal safety.3 This classification approach helps determine the level of support needed (i.e., ranging from intermittent to ongoing high-level support) for all activities and does not base severity on IQ alone (e.g., mild, 50-69; moderate, 35-49; severe, 20-34; and profound, less than 20).2,3 Further, this approach places the focus on a person’s strengths and weaknesses so as to better relate them to the demands of the individual’s environment, in addition to the expectations and attitudes of the family and community.3 A description of the mental health of a person with ID would include ID severity, ID etiology, and related mental disorders (e.g., developmental disorders, psychiatric illness, personality disorders, problem behaviors, other disorders). 2
Etiology
Intelligence is determined both genetically and environmentally.3 Causes of ID may be attributed to injury, disease, or a neurologic problem (Table 1).1 In many cases, the cause of ID is not known.1 Some of the most common causes of ID occur prenatally, such as Down syndrome (DS), fetal alcohol syndrome, fragile X syndrome, genetic conditions, birth defects, and infections.1 Other causes of ID occur during childbirth (e.g., complications related to prematurity, central nervous system [CNS] bleeding, breech or high forceps delivery), or soon after birth; and still others do not occur until a child is older, such as serious head injury, stroke, or certain infections.1,3

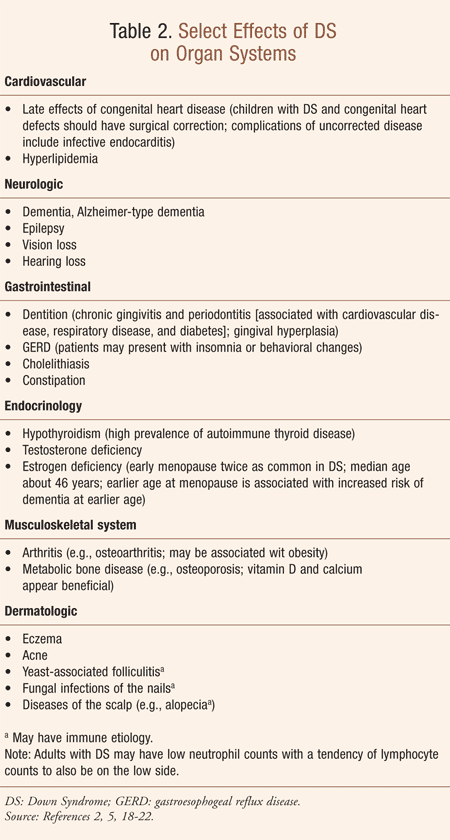
In seniors, the etiology of ID is often unknown.2 Congenital conditions are of particular relevance to the health of seniors with ID, since specific syndromes (e.g., DS, Angelmans syndrome, fragile X syndrome, Klinefelter’s syndrome, Turner syndrome, William’s syndrome) are associated with risk of disease and particular physical disorders.2 Select effects of DS on organ systems may be found in Table 2, including hyperlipidemia, epilepsy, and dementia/Alzheimer-type dementia. Of note, dementia is three to four times more prevalent among people with ID than among the comparably aged general population; in DS, the prevalence is substantially higher.5
Incidence/Prevalence/Prognosis
In the U.S., approximately 3% of the population functions at an IQ of less than 70; when the need for support is entered into the equation, only about 1% of the population functions with severe ID.3 DS is one of the most common genetic birth defects affecting approximately 1 in 800 (approximately 3,400) infants a year; according to the National Down Syndrome Society there are more than 400,000 individuals with DS in this country.6
Many individuals with ID of mild-to-moderate severity live independently, can support themselves, and are successful at jobs that require basic intellectual skills.3 Long-term support, however, is likely to be required for people with severe ID.3 Depending on the etiology of the disability, life expectancy may be shortened; however, the current level of health care is improving long-term health outcomes for individuals with all types of developmental disabilities.3

Overall, management of ID consists of education, family counseling, and social support.3 For a discussion on a comprehensive individualized management program involving multidisciplinary team support (e.g., nutritionists, orthopedists, neurologists, educators, etc.) see Reference 3. Specific therapies usually correspond to comorbidities and syndromic associations (e.g., effects of DS on organ systems–Table 2), as noted above. The mortality risk rises with the severity of the cognitive disability and the degree of the immobility.3 When addressing health promotion for individuals with ID, it is appropriate to aim for equity with the non-ID population with regard to health targets and evidence-based interventions; the medication regimen review is an integral component of this approach.2,10
Behavioral Issues
Behavior problems are more prevalent in individuals with ID, as compared to the general population; aggressive challenging behavior is common in ID, and up to one-third of those with ID have a mental health or behavior problem requiring specialized support services.11,12 The development of a behavioral intervention, based on information obtained from a thorough functional behavioral assessment (FBA) is part of best practice in the ID field. For a discussion on training staff and parents to conduct a FBA, see Reference 11.11
While aggressive behavior is common among patients with ID, it frequently is not indicative of a psychiatric illness.12 Nonetheless, approximately 20% of community-dwelling patients with ID receive antipsychotic medications.12 One randomized, controlled trial, conducted by Tyrer et al, was designed to shed light on this issue of treatment of aggressive challenging behavior in patients with ID by comparing the effects of haloperidol (a typical, first-generation antipsychotic drug), risperidone (an atypical, second-generation antipsychotic drug), and placebo in this patient population.12,13 The study suggested that antipsychotic medications are not effective in treating aggressive behavior among patients with ID.13 While assessment and treatment of such patients with behavioral and drug interventions require further study, the debate on the treatment of aggression in patients with ID, a very vulnerable group, continues. 12,13
Seizure Disorders
Precipitating Factors: Individuals with ID are at an increased risk for seizures, as are those with cerebral palsy, head injury, or strokes.14 Epilepsy is not only common in ID, including DS, but it may also mark the onset of dementia in older adults—and this may particularly be the case with late-onset myoclonic epilepsy.2 There is a greater incidence of epilepsy associated with a more profound degree of ID.14 Of note, occurrence of a first seizure has a bimodal distribution: one peak occurring in newborn and young children and the second peak occurring in patients older than 65 years of age.14
A variety of factors have been shown to precipitate seizures in susceptible individuals: absence seizures can be precipitated by hyperventilation; low birth weight and perinatal injuries are risk factors for partial-onset seizures; an increase in the frequency of seizures may occur with sleep, sleep deprivation, sensory stimuli, and emotional stress.14 Onset of or an increased frequency of seizures may be seen with changes in hormone levels occurring around the time of menses, puberty, or pregnancy.14 Furthermore, a careful history should be obtained from patients presenting with seizures, since medications may be the precipitating factor. High-dose phenothiazines, antidepressants (e.g., bupropion, maprotiline), theophylline, alcohol, and street drugs have been associated with seizure provocation.14 According to Rogers and Cavazos, immunizations have not been associated with an increased risk of epilepsy. 14
Antiepileptic Drugs (AEDs) in the Elderly: Clinicians are faced with special challenges regarding the use of AEDs in the elderly. Attention to avoiding AEDs that interact with concomitant medication therapy is paramount, particularly since polypharmacy is a common occurrence in this population. Pharmacists should have a heightened awareness of the potential for a detrimental outcome secondary to changes in therapeutic drug levels of concomitant medications, caused by induction or inhibition of the CYP450 system by many AEDs.14 For example, carbamazepine may increase the levels/effects of alcohol, CNS depressants, lithium, monoamine oxidase (MAO) inhibitors, and phenytoin, among others, while it may decrease the levels/effects of aripiprazole, contraceptives (estrogens), valproic acid, and vitamin K antagonists, among many others.15
Also problematic in the elderly is the common occurrence of hypoalbuminemia, which can pose difficulty in monitoring and adjusting serum drug levels of highly albumin-bound AEDs (e.g., valproic acid, phenytoin, tiagabine).14,15 Since age-related body mass changes (i.e., decrease in body water, increased ratio of fat to lean body mass) can affect the volume of distribution of certain drugs, elimination half-life may be affected in the elderly. Lower AED dosage requirements may be necessary in seniors in light of declining or impaired renal and/or hepatic function.
Further, seniors may be more “sensitive” to a variety of neurocognitive adverse effects of AEDs due to age-related changes in the pharmacodynamic response.14 Possible mechanisms for altered pharmacodynamics in older adults include 1) altered sensitivity secondary to changes in receptor number or affinity or changes in postreceptor response, and 2) age-related impairment of physiological and homeostatic mechanisms.16,17 In general, relatively lower AED serum concentrations may be efficacious (e.g., for seizure control) in the elderly.14 For a detailed discussion of the pathophysiology and clinical presentation of epilepsy, treatment algorithm for epilepsy, and drugs of choice for specific seizure disorders, see Reference 14.
Conclusion
The number of adults with ID surviving into old age is rapidly increasing. Understanding the nuances related to comorbidities and medication therapy in this population can help pharmacists with careful drug selection and medication regimen review, while at the same time guide patients, caregivers, and other health care professionals in this regard.
REFERENCES
1. Intellectual Disability. Fact Sheet. Centers for Disease Control and Prevention.
http://www.cdc.gov/ncbddd/
2. Starr JM. The older adult with intellectual disability. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:445-452.
3. Intellectual disability. Merckmanuals.com. Pediatrics. Learning and Developmental Disorders. February 2009.
www.merckmanuals.com/
4. Subtitle A general provisions. Administration on Developmental Disabilities (ADD). Administration for Children and Families. U.S. Department of Health and Human Services. Last reviewed June 2, 2011.
http://www.acf.hhs.gov/
5. Strydom A, Livingston G, King M, Hassiotis A. Prevalence of dementia in intellectual disability using different diagnostic criteria. Br J Psychiatr. 2007;191:150-157.
6. Down syndrome. Marchofdimes.com. July 2009.
http://www.marchofdimes.com/
7. Zagaria ME. Family caregiving: seniors continue to receive and deliver care. US Pharm. 2006;31(12):23-29.
http://www.uspharmacist.com/
8. Singer P. Long-term care system leans heavily on aging caregivers at home. Coshocton Tribune. May 31, 2003.
9. Ansberry C. Parents devoted to a disabled child confront old age. Wall Street Journal, January 7, 2004.
10. Beange H, Lennox N, Parmenter T. Health targets for people with an intellectual disability. J Int Disab Res. 1999; 24:283-297.
11. Busko M. Aggressive behavior in intellectual disability does not warrant routine antipsychotics. Medscape Medical News. January 9, 2008.
http://www.medscape.org/
12. Functional behavioural assessment in people with intellectual disabilities [mental retardation and developmental disorders] Updated: 10/04/2006. Curr Opin Psychiatry. 2006;19(5):475-480.
www.medscape.org/viewarticle/
13. Tyrer P, Oliver-Africano PC, Ahmed Z, et al. Risperidone, haloperidol, and placebo in the treatment of aggressive challenging behaviour in patients with intellectual disability: a randomised controlled trial. Lancet. 2008;371:57-63.
14. Rogers SJ, Cavazos JE. Epilepsy. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill Inc; 2008:927-951.
15. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 16th ed. Hudson, OH: Lexi-Comp, Inc; 2011.
16. Guay DR. The pharmacology of aging. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:138-144.
17. Feely J, Coakley D. Altered pharmacodynamics in the elderly. Clin Geriatr Med. 1990;6:269-283.
18. Roussot MA, Lawrenson JB, Hewitson J. Is cardiac surgery warranted in children with Down syndrome? A case-controlled review. SA Med J. 2006;96:924-930.
19. Schupf N, Zigman W, Kapell D. Early menopause in women with Down’s syndrome. J Int Disab Res. 1997;41:264-267.
20. Cosgrave MP, Tyrrell J, Gill M Lawlor BA. Age at onset of dementia and age of menopause in women with Down’s syndrome. J Int Disab Res. 1999;43:461-465.
21. Zubillaga P, Garrido A, Mugica I: Effect of vitamin D and calcium supplementation on bone turnover in institutionalized adults with Down’s Syndrome. Eur J Clin Nutr. 2006;60:605-609.
22. Prasher V. Screening of medical problems in adults with Down Syndrome. Down Syndrome Res Pract. 1994;2:59-66.
To comment on this article, contact
rdavidson@uspharmacist.com





