US Pharm. 2015;40(5):46-50.
ABSTRACT: Irritable bowel syndrome (IBS) causes abdominal discomfort and pain and can worsen quality of life in children and adolescents. IBS is associated with changes in bowel function, resulting in diarrhea and/or constipation. Diagnosis involves assessing the patient’s history and clinical symptoms and applying the Rome III criteria. The main goals of managing IBS in children and adolescents are to improve abdominal function and reduce discomfort or pain, thereby enhancing quality of life. Pharmacologic management is beneficial as adjunctive short-term therapy, and nonpharmacologic management has been widely shown to produce the same or better outcomes in children and adolescents. Pharmacists can educate patients and their families, identify pediatric providers and community resources for IBS management, and ensure that patients avoid medications that can aggravate IBS.
Irritable bowel syndrome (IBS) is a common abdominal condition affecting children and adolescents. In this population, IBS typically presents as abdominal pain and is associated with bowel changes, including diarrhea, constipation, and alternating diarrhea and constipation. IBS can be debilitating and can negatively impact young patients’ quality of life, resulting in poorer school attendance and increased healthcare expenditures. The incidence and prevalence of IBS-associated abdominal pain in children and adolescents range from 8% to 17% and 13% to 38%, respectively.1 The cause of IBS is not completely understood; suspected factors include infection, inflammation, visceral hypersensitivity, allergy, and gut motility. The bowel changes observed in IBS patients have been linked to a state of dysregulation in the gastrointestinal (GI) enteric system and the central nervous system that results in motility alterations and GI immune-system dysfunction.2 This article will review IBS, its diagnosis, and available management strategies for children and adolescents.
DIAGNOSIS
There is no specific test for diagnosing IBS. Consequently, obtaining a detailed patient history and clinical symptoms remains extremely important for diagnosis. IBS is diagnosed by applying the Rome III criteria (TABLE 1) and ruling out red-flag symptoms that may be caused by other organic GI diseases. The Rome III criteria identify symptom clusters for various functional GI disorders, including IBS.3-5

The Rome III criteria define IBS as recurrent abdominal pain or discomfort that is associated with alterations in defecation and changes in stool consistency. Accordingly, recurrent abdominal pain or discomfort for 3 or more days per month in the preceding 3 months that is associated with two or more of the following is diagnostic of IBS: improvement of symptoms upon defecation; onset associated with change in form and/or appearance of stool; abnormal stool frequency; straining during defecation; urgency or feeling of defecation; mucus in stool; and bloating.6 Red-flag symptoms that indicate a more serious abdominal disorder or GI disease other than IBS include anemia, weight loss, rectal bleeding, nocturnal or progressive abdominal pain, severe diarrhea or vomiting, delayed puberty, fever of unknown origin, and family history of inflammatory bowel disease.
MANAGEMENT
The major goal in managing IBS in children and adolescents is to control the symptoms and eliminate factors that can exacerbate the condition.7 Factors that can exacerbate IBS include diet and stress. Useful approaches for the management of IBS include pharmacologic and nonpharmacologic methods.
Pharmacologic Management
Several classes of pharmacologic agents may benefit patients with IBS, based on these agents’ mechanisms of action. Among these are antispasmodics, antidepressants, acid suppressants, prokinetic agents, and antibiotics (TABLE 2). Currently, data are insufficient to support the use of any pharmacologic agent as first-line therapy for IBS in children and adolescents. Therefore, the selection and use of the agents reviewed below should be tailored to the individual patient and limited to short-term therapy.
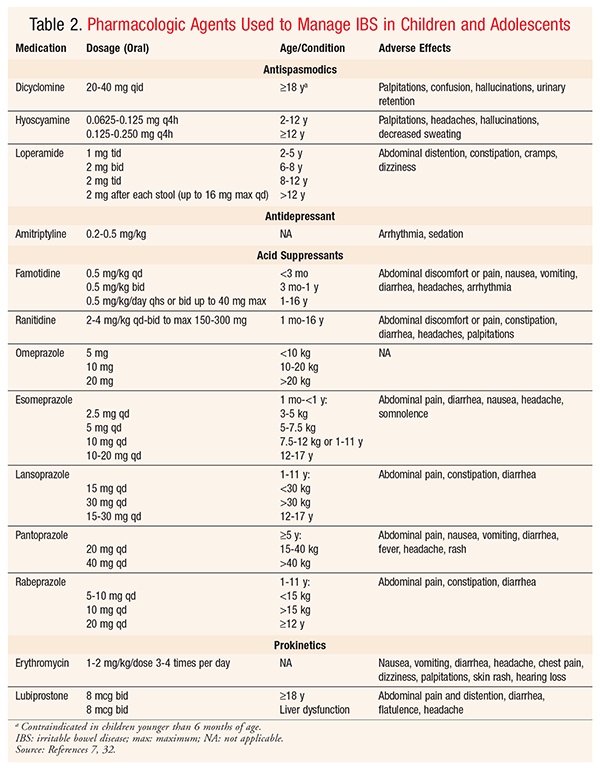
Antispasmodics: In IBS with predominant diarrhea, antispasmodic agents may be helpful. Agents with both antispasmodic and anticholinergic properties, such as dicyclomine and hyoscyamine, have been used effectively for GI pain symptoms in pediatric patients.8 Antispasmodics decrease the exaggerated GI-muscle contractility and motility responsible for diarrhea.
Dicyclomine acts directly on GI smooth muscle to produce antispasmodic effects and on muscarinic receptors to produce anticholinergic effects.8 Dicyclomine use in infants younger than 6 months is contraindicated. Furthermore, safety and efficacy data in patients younger than 18 years are not available. Consequently, dicyclomine use is recommended only in patients older than 18 years. Side effects include tachycardia, confusion, hallucinations, inability to urinate, thirst, sweating, and hot and dry skin.
Hyoscyamine is recommended for managing IBS in children older than 2 years.8 Side effects of hyoscyamine include palpitations, headaches, hallucinations, decreased sweating, and mydriasis. Another agent, loperamide, acts on opiate receptors in the GI wall, inhibiting acetylcholine and prostaglandin release and reducing peristalsis and intestinal transit time.8 Loperamide is indicated for diarrhea associated with IBS. The chief side effects of loperamide are constipation, abdominal distention and cramps, and dizziness.
Antidepressants: Antidepressants have been reported to be effective in managing IBS in children.9,10 These agents may be beneficial for both pain and constipation. The tricyclic antidepressant amitriptyline was shown to be effective at a low dosage. Amitriptyline effectiveness in IBS is linked to its modulatory actions on various GI neurotransmitters, including norepinephrine, acetylcholine, and histamine. Amitriptyline acts on both the norepinephrine and serotonin receptors to decrease pain perception; on the muscarinic receptors to affect their anticholinergic activity, slowing gastric transit time and improving diarrhea, but exacerbating constipation; and on the histamine receptors to reduce acid secretions, improving dyspepsia. A side effect is heart arrhythmia secondary to QT-interval prolongation, necessitating ECG monitoring and sedation; this can be managed by administering the dose at bedtime.11
Selective Serotonin Reuptake Inhibitors (SSRIs): Serotonin is an important GI neurotransmitter that modulates colonic motility. SSRIs can be useful for IBS symptoms, particularly constipation, because they prevent the reuptake of serotonin, thereby increasing its concentration at the receptor site and enhancing colonic tone, which improves bowel regularity.12 However, studies are lacking on SSRI use for IBS in children. Additionally, the black box warning on the potential for suicidal ideation in young adults poses a major obstacle to the routine consideration of SSRI use in adolescents with IBS.
Acid Suppressants: Acid-suppressing medications, such as histamine2 blockers and proton pump inhibitors (PPIs), have been routinely used to manage abdominal pain in children.13 Famotidine has been used for abdominal pain associated with dyspepsia, although there are no specific studies in pediatric patients with IBS. Alternatively, ranitidine may be used. Side effects include headaches, dizziness, constipation, and diarrhea.13
Omeprazole, a PPI, is recommended for children and adolescents.14 Side effects include abdominal pain, diarrhea, flatulence, vomiting, and headache. Alternatives to omeprazole include esomeprazole, lansoprazole, pantoprazole, and rabeprazole.
Prokinetics: Prokinetic agents stimulate GI motility using different mechanisms to improve IBS involving constipation.15 Erythromycin, a motility receptor agonist, stimulates the smooth-muscle cells of the GI tract to improve gastric transit time. Taking 1 to 2 mg/kg/dose orally three to four times per day may be beneficial for pain symptoms and dyspepsia. However, there are no studies supporting the use of erythromycin specifically in pediatric patients with IBS. Lubiprostone, a type 2 chloride channel activator, produces electrolyte and fluid secretion in the small intestine and also stimulates colonic motility.16 Lubiprostone, which is useful for IBS involving constipation, is approved for adolescent IBS patients older than 18 years. The recommended dosage is 8 mcg administered twice daily with food. The frequency must be decreased to once daily in patients with hepatic dysfunction. Side effects often accompanying lubiprostone use include abdominal distention and pain, diarrhea, flatulence, and headaches.
Antibiotics: GI infections have been shown to lead to IBS, particularly in the young. In children and adolescents, a longer duration of GI infection increases the risk of developing IBS. A meta-analysis has reported a causative link between acute GI infection and the incidence of IBS in children and adolescents. The cause of GI infection–induced IBS is not well defined, but the disorder is hypothesized to be related to increased intestinal permeability and leakage, mucosal inflammation, and changes in the microflora of the intestine, which result in GI mucosal-wall dysfunction.17 The routine use of antibiotics in children with postinfection-related IBS is not recommended because it can potentially lead to antibiotic resistance, and there is also the difficulty of selecting the appropriate antibiotic, particularly if bacterial overgrowth in the small intestine has not been demonstrated.
Nonpharmacologic Management
Dietary Restriction: A trial of a lactose-free diet may be beneficial in older children and adolescents. Lactose deficiency is not rare in younger children, but currently no studies recommend routine use of a lactose-restricted diet to manage IBS in pediatric patients. Fructose intolerance due to malabsorption of carbohydrates—mostly fructose from high-fructose corn syrup in many food products consumed by children and adolescents—has been linked to osmotic diarrhea and the production of gas in the colon through fermentation. Fructose restriction in children with IBS symptoms has been shown to produce sustained improvement.18
Fiber: A high-fiber diet may be useful for IBS involving constipation. The efficacy of fibers such as bran, husk, and methylcellulose has been reported in clinical trials to be around 10%. The negative aspects of fiber therapy are that it does not improve pain symptoms and it may worsen flatulence and bloating.19
Fermentable Carbohydrates: Fermentable oligo-, di-, and monosaccharides and polyols (FODMAP) have been shown to trigger GI symptoms such as pain, bloating, flatulence, and diarrhea in IBS patients.20 The FODMAP diet showed promise in reducing abdominal pain, bloating, flatulence, and diarrhea in patients who followed it for up to 6 months.21
Probiotics: Several studies of probiotics in pediatric patients have shown improvement of many IBS symptoms, such as abdominal pain, stool frequency, bloating, mucus, blood in the stool, straining, and the urge to defecate.22,23 The studies evaluated the effects of different probiotics given to children with IBS for 12 weeks and compared outcomes with those in children given placebo. The observed beneficial actions of probiotics were suggested to be related to several properties of the microorganisms in the probiotic formulation, including enhancing intestinal barrier function; inhibiting pathogens’ ability to bind to the intestinal wall and induce injury; modulating the gut’s inflammatory response to injury; reducing the visceral hypersensitivity response to inflammation and psychological stress; and stabilizing colonic microflora and fermentation activity.23
Acupuncture: Acupuncture, a form of traditional Chinese medicine, has been used for centuries to manage various conditions. It is known to reduce gastric acid secretions, increase gastric motility, and decrease visceral pain.24 Acupuncture may be helpful for managing IBS with constipation in children and adolescents, although there are no controlled studies specifically evaluating its use in these populations.25
Cognitive Behavioral Therapy (CBT): CBT is a form of psychotherapy based largely on interactions between thoughts, feelings, and behavior. The main therapeutic goal of CBT is to improve coping and problem-solving skills, which can help patients better identify IBS triggers and improve their maladaptive responses to these triggers. Learned techniques include monitoring symptoms, feelings, and behaviors; documenting responses to learned relaxation; and using distraction strategies, as well as positive and negative reinforcement mechanisms. CBT is beneficial in the management of IBS in children and adolescents, especially when IBS is associated with pain symptoms. Studies of CBT in children aged 5 to 18 years have shown it to be as beneficial as dietary or pharmacologic interventions in managing somatic symptoms of IBS.26 However, CBT may be inaccessible to many children because of the limited number and availability of trained pediatric providers.
Hypnotherapy: Hypnotherapy helps reduce colonic contractions and normalize abnormal visceral impulses, thereby improving IBS patients’ symptoms and outlook about the condition. Several studies have reported on the effectiveness of hypnotherapy in children with IBS.27-30 As is the case with CBT, the limited number of pediatric hypnotherapists may limit accessibility, but this can be mitigated by using recorded guided therapy at home. In a randomized study evaluating the use of self-guided prerecordings at home, children treated with this method had a reduction in symptoms. The improvements achieved using the prerecordings were sustained for 6 months following completion of the study.31
THE PHARMACIST’S ROLE
Being highly accessible to the public, pharmacists can play a role in the management of IBS in children and adolescents. Pharmacists must understand the signs, symptoms, and presentation of IBS, as well as, management options. Upon encountering a patient with symptoms and a diagnosis of IBS, pharmacists can reassure patients, families, and caregivers by counseling them about the disorder and appropriate care. Pharmacists can identify pediatric CBT therapists or hypnotherapists, as well as other community resources for managing IBS. Pharmacists should routinely identify drugs that may exacerbate the pain, constipation, and diarrhea associated with IBS and advise patients against taking such products. Finally, pharmacists can monitor the patient’s therapy for appropriate outcomes and manage or prevent side effects of prescribed pharmacologic agents.
CONCLUSION
IBS remains a common disorder with a high prevalence in children and adolescents. IBS is distressing and negatively impacts patients’ school and social life. Pharmacists are uniquely positioned to effect better treatment outcomes by educating the parents or caregivers of children and adolescents with IBS about therapy options, as well as by ensuring that patients can gain access to helpful resources and trained pediatric providers in their community.
REFERENCES
1. Saps M, Seshadri R, Sztainberg M, et al. A prospective school-based study of abdominal pain and other common somatic complaints in children. J Pediatr. 2009;154:322-326.
2. El-Matary W, Spray C, Sandlin B. Irritable bowel syndrome: the commonest cause of recurrent abdominal pain in children. Eur J Pediatr. 2004;163:584-588.
3. Mearin F, Lacy BE. Diagnostic criteria in IBS: useful or not? Neurogastroenterol Motil. 2012;24:791-801.
4. Drossman DA. The functional gastrointestinal disorders and the Rome III process. Gastroenterology. 2006;130:1377-1390.
5. Spiegel BM, Farid M, Esrailian E, et al. Is irritable bowel syndrome a diagnosis of exclusion?: a survey of primary care providers, gastroenterologists, and IBS experts. Am J Gastroenterol. 2010;105:848-858.
6. Halpert AD. Importance of early diagnosis in patients with irritable bowel syndrome. Postgrad Med. 2010;122:102-111.
7. Paul SP, Barnard P, Bigwood C, Candy DC. Challenges in management of irritable bowel syndrome in children. Indian Pediatr. 2013;50:1137-1143.
8. Ford AC, Talley NJ, Spiegel BM, et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ. 2008;337:a2313.
9. Bahar RJ, Collins BS, Steinmetz B, Ament ME. Double-blind placebo-controlled trial of amitriptyline for the treatment of irritable bowel syndrome in adolescents. J Pediatr. 2008;152:685-689.
10. Saps M, Youssef N, Miranda A, et al. Multicenter, randomized, placebo-controlled trial of amitriptyline in children with functional gastrointestinal disorders. Gastroenterology. 2009;137:1261-1269.
11. Gutgesell H, Atkins D, Barst R, et al. Cardiovascular monitoring of children and adolescents receiving psychotropic drugs: a statement for healthcare professionals from the Committee on Congenital Cardiac Defects, Council on Cardiovascular Disease in the Young, American Heart Association. Circulation. 1999;99s:979-982.
12. Lebel AA. Pharmacology. J Pediatr Gastroenterol Nutr. 2008;47:703-705.
13. See MC, Birnbaum AH, Schechter CB, et al. Double-blind, placebo-controlled trial of famotidine in children with abdominal pain and dyspepsia: global and quantitative assessment. Dig Dis Sci. 2001;46:985-992.
14. Gibbons TE, Gold BD. The use of proton pump inhibitors in children: a comprehensive review. Paediatr Drugs. 2003;5:25-40.
15. Tack J. Prokinetics and fundic relaxants in upper functional GI disorders. Curr Opin Pharmacol. 2008;8:690-696.
16. Drossman DA, Chey WD, Johnson JF, et al. Lubiprostone in patients with constipation-associated irritable bowel syndrome—results of two randomized, placebo-controlled studies. Aliment Pharmacol Ther. 2009;29:329-341.
17. Thabane M, Kottachchi DT, Marshall JK. Systematic review and meta analysis: the incidence and prognosis of post-infectious irritable bowel syndrome. Aliment Pharmacol Ther. 2007;26:535-544.
18. Gomara R, Halata M, Newman L, et al. Fructose intolerance in children presenting with abdominal pain. J Pediatr Gastroenterol Nutr. 2008;47:303-308.
19. Snook J, Shephard HA. Bran supplementation in the treatment of irritable bowel syndrome. Aliment Pharmacol Ther. 1994:8:511-514.
20. Lozinsky AC, Boé C, Palmero R, Fagundes-Neto U. Fructose malabsorption in children with functional digestive disorders. Arg Gastroenterol. 2013;50:226-230.
21. de Roest RH, Dobbs BR, Chapman BA, et al. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: a prospective study. Int J Clin Pract. 2013;67:895-903.
22. Huertas-Ceballos A, Logan S, Bennett C, Macarthur C. Psychosocial interventions for recurrent abdominal pain (RAP) and irritable bowel syndrome (IBS) in childhood. Cochrane Database Syst Rev. 2008;(1):CD003014.
23. Bu LN, Chang MH, Ni YH, et al. Lactobacillus casei rhamnosus Lcr35 in children with chronic constipation. Pediatr Int. 2007;49:485-490.
24. Schneider A, Enck P, Streitberger K, et al. Acupuncture treatment in irritable bowel syndrome. Gut. 2006;55:649-654.
25. Lembo AJ, Conboy L, Kelley JM, et al. A treatment trial of acupuncture in IBS patients. Am J Gastroenterol. 2009;104:1489-1497.
26. Broide E, Pintov S, Portnoy S, et al. Effectiveness of acupuncture for treatment of childhood constipation. Dig Dis Sci. 2001;46:1270-1275.
27. Vliegar AM, Rutten JM, Govers AM, et al. Long-term follow-up of gut-directed hypnotherapy vs. standard care in children with functional abdominal pain or irritable bowel syndrome. Am J Gastroenterol. 2012;107:627-631.
28. Adinolfi B, Gava N. Controlled outcome studies of child clinical hypnosis. Acta Biomed. 2013;84:94-97.
29. Whorwell PJ. Hynotherapy: first line treatment for children with irritable bowel syndrome? Arch Dis Child. 2013;98:243-244.
30. Gulewitsch MD, Müller J, Hautzinger M, Schlarb AA. Brief hypnotherapeutic-behavioral intervention for functional abdominal pain and irritable bowel syndrome in childhood: a randomized controlled trial. Eur J Pediatr. 2013;172:1043-1051.
31. van Tillburg MA, Chitkara DK, Palsson OS, et al. Audio-recorded guided imagery treatment reduces functional abdominal pain in children: a pilot study. Pediatrics. 2009;124:e890-e897.
32. RxList (Internet Drug Index). www.rxlist.com. Accessed December 27, 2014.
To comment on this article, contact rdavidson@uspharmacist.com.





