US Pharm. 2012;37(12):45-48.
ABSTRACT: Irritable bowel syndrome (IBS) is a common functional gastrointestinal disorder that typically affects adults between the ages of 30 and 50 years, with female predominance. Its hallmark features include abdominal discomfort, bloating, and abnormal defecation, which may be constipation or diarrhea predominant. IBS is difficult to diagnose and does not have an established cure at this time. Currently, treatment strategies are patient specific, focusing on symptom control and improvement in quality of life. Nonpharmacologic options such as exclusion diets, dietary changes, peppermint oil, exercise, relaxation therapy, or acupuncture have been evaluated. Pharmacologic therapy includes antidiarrheals, laxatives, probiotics, anticholinergics, antidepressants, antibiotics, and the newly approved drug linaclotide. New treatment options are also currently in the pipeline. Pharmacists can play an important role in assisting the patient with IBS, as they are easily accessible health care professionals.
Irritable bowel syndrome (IBS) is a common functional gastrointestinal (GI) disorder that is characterized by abdominal discomfort, bloating, and abnormal defecation.1,2 The two main classifications of IBS are IBS with constipation predominant (IBS-C) and IBS with diarrhea predominant (IBS-D). Patients with IBS-C experience hard stools more than 25% of the time and loose stools less than 25% of the time, while patients with IBS-D experience loose stools more than 25% of the time and hard stools less than 25% of the time.3 Some patients may also have IBS with alternating diarrhea and constipation (IBS-A).4 This change in frequency and form of stool found with IBS-C, IBS-D, or IBS-A can be noted in the symptom-based (Rome III) criteria for IBS diagnosis (TABLE 1).

The prevalence of IBS in the United States is estimated to be between 3% and 20%, depending on the criteria used, and it decreases slightly with advanced age.4 Patients with new onset of IBS symptoms are typically between the ages of 30 and 50 years and are twice as likely to be women.1,3
In the U.S., IBS accounts for a significant financial burden estimated at $1.6 billion in direct and $19.2 billion in indirect costs.1 It also accounts for at least 1 in 10 primary care visits and a number of specialty referrals. Unfortunately, only a limited number of patients actually seek medical care; hence, the disease often goes unrecognized.4 Lack of insurance or access to care may be a contributing factor.
PATHOPHYSIOLOGY
Although the pathophysiology of IBS is not fully understood, there are many theories regarding the etiology of the condition. Some possible causes include genetic mutations, abnormal GI motility, enhanced gut pain sensation (visceral hypersensitivity), bacterial flora and inflammatory cell alteration, or psychological changes.1,3,5 Most likely a combination of these factors leads to IBS.5
One theory concerning the pathophysiology of IBS involves the neurotransmission between the central nervous system (CNS) and the intestines. Numerous structures in the CNS connect to the GI tract through serotonergic nerves that help regulate GI motility, sensation, and secretion. In patients with IBS, an imbalance in the function of serotonin leads to the deregulation of intestinal motility. Based on this serotonin imbalance theory, various treatment approaches have been suggested.5,6
DIAGNOSIS
IBS is difficult to diagnose. There is no reliable biological marker for this condition, and the symptoms can mimic other GI disorders. In an attempt to guide clinicians in establishing a diagnosis, symptom-based criteria have been developed for IBS. The most recent, the Rome III criteria (TABLE 1), is typically used in clinical research to diagnose and classify patients.7
Historically, IBS has been a diagnosis of exclusion.2 However, more recently, clinicians are encouraged to diagnose IBS based on a comprehensive history, using symptom-based criteria and considering the presence or absence of specific alarm features. These alarm features include unintended weight loss of more than 10 lbs, fevers or chills, high-volume diarrhea, nocturnal diarrhea, family history of GI malignancy, and older age (≥50 years). If these features are present, further testing is warranted, but if absent, further testing is typically not recommended.7
SIGNS AND SYMPTOMS
IBS has a wide spectrum of symptoms with significant interpatient variability. It is generally characterized by cramps or lower abdominal pain. Patients may present with altered bowel habits in volume, frequency, and consistency, as well as abdominal bloating and flatulence. Other symptoms may include gastroesophageal reflux, dysphagia, dyspepsia, nausea, impaired sexual function, dysmenorrhea, and an increase in frequency and urgency to urinate.6
TREATMENT OPTIONS
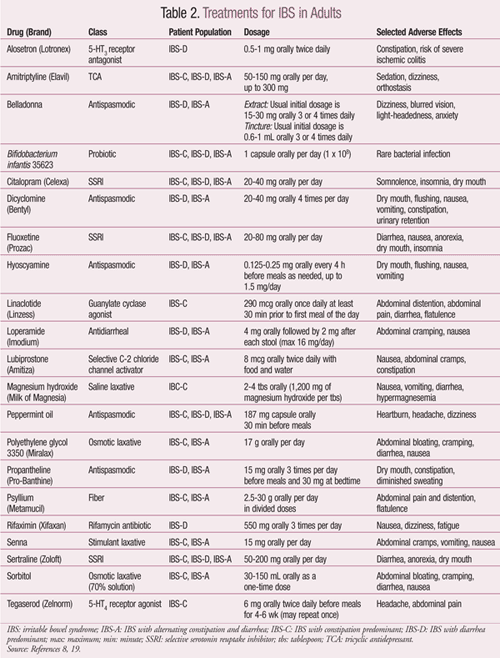
Since the causes of IBS have yet to be elucidated, there are no set treatments. Currently, management depends primarily on patient-specific symptoms and clinical response.1 A summary of pharmacologic treatments is located in TABLE 2.8 There is no cure for IBS at this time.
Nonpharmacologic Treatment Options
Dietary modification is a nonpharmacologic option for patients with IBS. Various foods such as beans, alcohol, caffeine, and fatty meals, and gas-producing foods such as broccoli, legumes, cabbage, and avocado may aggravate symptoms in some patients.1,9 This has led many patients to exclude these suspected aggravating foods from their diet (exclusion diets), although the effectiveness of such practices remains controversial.9 Increased intake of dietary fiber has also been proposed as a treatment, with best evidence supporting ispaghula/psyllium (e.g., Metamucil) over other formulations.9
Patients with IBS should monitor their symptoms using a daily diary for 2 to 3 weeks to help identify times and severity of symptoms, presence of aggravating factors, and possible exclusion diets.1
Homeopathic treatments have been used in the treatment of IBS, although their effectiveness has not been fully established. One homeopathic treatment option is acupuncture, an ancient traditional Chinese medical practice. It is becoming more widely accepted in Western society and may be of benefit. Peppermint oil may be another treatment option, as it has shown to relieve symptoms and be well tolerated in patients with IBS. Exercise, such as bicycle pedaling, and relaxation techniques, such as muscle relaxation and conscious breathing, may help patients by reducing stress and relieving IBS symptoms.5,10
Pharmacologic Treatment Options
IBS-Constipation Predominant: Patients with IBS-C may benefit from the use of laxatives, such as magnesium hydroxide, sorbitol, or polyethylene glycol (PEG).1 However, the only laxative that has been studied in patients with IBS is PEG.11
Lubiprostone, a chloride channel activator for the management of IBS-C in women aged 18 years and older, increases the movement of water and sodium into the lumen from the body, thereby providing the desired clinical benefit. In clinical studies, lubiprostone increased the number of weekly spontaneous bowel movements and improved stool consistency, straining, and bloating as well. Lubiprostone is contraindicated in patients with mechanical bowel obstruction and should be avoided in patients with preexisting diarrhea. Since lubiprostone carries a Category C pregnancy rating, a negative pregnancy test and use of contraception is recommended for women taking it.11
Tegaserod is indicated for the short-term treatment of women with IBS-C who are <55 years of age. This 5-HT4 receptor agonist, previously withdrawn from the market due to safety concerns including increased risk of heart attack and stroke, is currently available only through the FDA upon request in emergency situations.11,12 The FDA may deny the request if there is no reasonable basis for concluding that tegaserod may be effective for the intended use, or if exposure to the medication would pose a significant risk to the patient.12
The most widely used antidepressants in IBS are tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs), as they reduce pain and improve global IBS symptoms. Their analgesic and anxiolytic effect is thought to improve GI motility and function. TCAs should be used with caution in elderly patients due to the increased side effects such as urinary retention, sedation, and tachycardia.13
Probiotics may be considered to help prevent overgrowth of pathogenic bacteria in the GI tract as well as to reduce the inflammatory state of the GI tract. Although efficacy studies for probiotics are lacking due to study design, a systematic review of the studies showed that Bifidobacterium species may be effective in improving abdominal pain, bloating, and flatulence.8
In August 2012, the FDA approved linaclotide as a once-daily treatment for IBS-C. Linaclotide is the first-in-class guanylate cyclase (GC-C) agonist and acts locally in the intestine with minimal systemic exposure. It has been shown to reduce abdominal pain and increase bowel movement frequency; however, it is contraindicated in pediatric patients up to 6 years old and should be avoided in patients aged 6 through 17 years because in nonclinical studies, administration of a single, clinically relevant adult dose caused deaths in young juvenile mice.14
IBS-Diarrhea Predominant: IBS-D is associated with increased GI motility; hence, antidiarrheals are commonly used in symptom control. Loperamide, a widely available OTC product, has shown to reduce GI transit time and improve diarrhea and urgency. However, it does not improve abdominal pain and global IBS symptoms.11
A classic complaint in IBS-D is abdominal pain or discomfort that is attributed to intestinal smooth-muscle spasm and an exaggerated motility response of the small bowel and colon. As a result, antispasmodics such as hyoscyamine, dicyclomine, belladonna, and propantheline have been utilized to help relieve this symptom. Although these products have traditionally been used, there is limited clinical evidence supporting their use, especially concerning long-term effectiveness. They are considered to provide short-term relief of abdominal pain or discomfort. Some common side effects that limit use, especially in the elderly, are dry mouth, dizziness, blurry vision, and urinary retention.11
Alosetron, a 5-HT3 receptor antagonist approved by the FDA for the treatment of IBS-D, is indicated for women with severe IBS-D.11 If patients have experienced symptoms for over 6 months, have anatomical or biochemical abnormalities of the GI tract excluded, and have not responded adequately to conventional therapy, alosetron may be effective.15 It has shown to significantly decrease abdominal pain, urgency, and stool frequency while increasing stool consistency. Due to infrequent but serious adverse events including ischemic colitis, the product was withdrawn from the market from November 2000 to June 2002 but is now marketed with a risk management plan.11
The role of antibiotics in IBS is based on the assumption that altered GI flora is partly responsible for the symptoms. Rifaximin, a nonsystemic antibiotic, is indicated for traveler’s diarrhea and hepatic encephalopathy. It has been tried in IBS patients with diarrhea or bloating and has demonstrated improvement in symptoms.8 Antibiotics should be used with extreme caution, as bacterial resistance and side effects are a major concern.
Future Treatment Options
Due to the unmet need for treatment options, there are various products in the pipeline for the treatment of IBS. For example, asimadoline, an orally administered kappa opioid–receptor agonist is being studied in IBS-D patients. Currently in phase III trials, it has shown to produce significant improvements in pain, urgency, frequency, and bloating.16 Pumosetrag (DDP-733), an orally available gastroprokinetic agent and locally acting 5-HT3 partial agonist, is being developed for the potential treatment of IBS-C. It is currently in phase II trials and has shown positive effects.17 Other emerging treatments for IBS include cholecystokinin antagonists such as loxiglumide and dexloxiglumide.18
ROLE OF PHARMACIST
Pharmacists can play an essential role for patients with IBS by helping them identify symptoms, discussing possible treatment options, and educating them about the disease. The community pharmacist is in an ideal position to recognize patients who likely have IBS and either attempt to self-medicate with various OTC products such as herbal supplements or have prescriptions for antidiarrheals or laxatives. Proactive questioning of the patient will allow the pharmacist to assess the appropriateness of the medications or identify a need for referral to other health care professionals for further evaluation. Finally, it is also important for the pharmacist to reassure the patient that IBS remains a manageable illness for the most part.
REFERENCES
1. Drossman DA, Camilleri M, Mayer EA, et al. AGA technical review on irritable bowel syndrome. Gastroenterology. 2002;123:2108-2131.
2. Cash BD, Chey WD. Irritable bowel syndrome—an evidence-based approach to diagnosis. Aliment Pharmacol Ther. 2004;19:1235-1245.
3. Hawbolt J. IBS treatment guidelines. US Pharm. 2009;34(12):Epub. www.uspharmacist.com/content/d/feature/i/902/c/16925/. Accessed September 23, 2012.
4. Choung RS, Locke GR. Epidemiology of IBS. Gastroenterol Clin North Am. 2011;40:1-10.
5. Yoon SJ, Grundmann O, Koepp L, et al. Management of
irritable bowel syndrome (IBS) in adults: conventional and
complementary/alternative approaches. Alt Med Rev. 2011;16:134-150.
6. Vahedi H, Ansari R, Mir-Nasseri MM, et al. Irritable bowel syndrome: a review article. Mid East J Digest Dis. 2010;2:66-77.
7. Furman DL, Cash BD. The role of diagnostic testing in irritable bowel syndrome. Gastroenterol Clin North Am. 2011;40:105-119.
8. Smith L. Irritable bowel syndrome. In: Richardson M, Chant C, Chessman KH, et al, eds. Gastroenterology and Nutrition: Pharmacotherapy Self-Assessment Program. 7th ed. Lenexa, KS: American College of Clinical Pharmacy; 2012:49-65.
9. Eswaran S, Tack J, Chey WD. Food: the forgotten factor in the irritable bowel syndrome. Gastroenterol Clin North Am. 2011;40:141-62.
10. Magge S, Lembo A. Complementary and alternative medicine for the irritable bowel syndrome. Gastroenterol Clin North Am. 2011;40:245-253.
11. Saad RJ. Peripherally acting therapies for the treatment of irritable bowel syndrome. Gastroenterol Clin North Am. 2011;40:163-182.
12. Zelnorm (tegaserod maleate) information. FDA.
www.fda.gov/Drugs/DrugSafety/PostmarketDrugSafetyInformationforPatientsandProviders/ucm103223.htm.
Accessed September 23, 2012.
13. Grover M, Drossman DA. Centrally acting therapies for irritable bowel syndrome. Gastroenterol Clin North Am. 2011;40:183-206.
14. Ironwood and Forest announce FDA approval of Linzess
(linaclotide) for the treatment of irritable bowel syndrome with
constipation and chronic idiopathic constipation. Forest Laboratories,
Inc.
http://news.frx.com/press-release/corporate-news/ironwood-and-forest-announce-fda-approval-linzesstm-linaclotide-treatme.
Accessed September 23, 2012.
15. Lotronex (alosetron) package insert. San Diego, CA: Prometheus Laboratories Inc; September 2010.
16. Product development. Tioga Pharmaceuticals, Inc. www.tiogapharma.com/product_development.html. Accessed September 23, 2012.
17. Evangelista S. Drug evaluation: pumosetrag for the
treatment of irritable bowel syndrome and gastroesophageal reflux
disease. Curr Opin Investig Drugs. 2007;8:416-422.
18. Galligan JJ, Vanner S. Basic and clinical pharmacology of new motility promoting agents. Neurogastroenterol Motil. 2005;17:643-653.
19. Micromedex Healthcare Series [Internet database].
Greenwood Village, CO: Thomson Reuters (Healthcare) Inc; updated
periodically.
To comment on this article, contact rdavidson@uspharmacist.com.





