Malaria affects large populations around the globe and is associated with serious health outcomes and death among infected individuals. According to the World Health Organization (WHO), there were an estimated 247 million cases among 3.3 billion at-risk people in 2006.1 Approximately 881,000 deaths were associated with the disease, with more than 91% occurring in Africa and 85% occurring in children under 5 years of age. Among cases outside of Africa, more than 80% occurred in regions of Southeast Asia.1 While malaria is no longer endemic to the United States, up to 2,000 U.S. residents are infected each year owing to globalization and travel.2
Malaria is caused by the protozoan parasite Plasmodium. There are four Plasmodium species: Plasmodium falciparum, Plasmodium vivax, Plasmodium ovale, and Plasmodium malariae. Carried by mosquitoes, P falciparum is the most frequent cause of infection and leads to the most serious outcomes associated with malaria. Within minutes after a person is bitten, sporozoites (a type of infective cell) are carried through the bloodstream to the liver, where they invade hepatocytes and then develop into schizonts (a more mature form of the cell). After a period of 5 to 15 days, the schizonts rupture to release daughter cells into the blood; these cells then invade erythrocytes, leading to hemolysis, anemia, and splenomegaly.3 Symptoms (including headache, abdominal pain, fatigue, chills, and fever) usually manifest 1 to 3 weeks after infection occurs. If the patient is diagnosed with malaria, treatment must be aggressive in order to prevent complications such as severe ischemia, hypoglycemia, pulmonary edema, and renal failure.3
Historically, clinicians have used standard microscopic procedures to identify and diagnose Plasmodium species. Laboratory clinicians use blood smears (thick and thin smears) for proper identification of species. This requires precise and meticulous microscopy, and results can be difficult to interpret.4 Other methods of diagnosing malaria have been developed, including DNA and RNA probes by polymerase chain reaction, rapid dipstick test, and--more recently--antigen-specific protein detection, a technology utilized in the BinaxNOW Malaria Test Kit.3,5
BinaxNOW Diagnostic Malaria Test
Developed by Inverness Medical Professional Diagnostics, the BinaxNOW Malaria Test Kit (FIGURE 1) is a rapid diagnostic test that uses whole blood collected by finger stick or venous draw. Intended for use in the clinical setting, this BinaxNOW test is an in vitro immunochromatographic assay for qualitative detection of Plasmodium antigens. Using monoclonal antibodies, it targets histidine-rich protein II--an antigen specific for P falciparum--and a panmalarial antigen that is present in all four Plasmodium species.4 The duality of the test allows for rapid detection while differentiating P falciparum from the other malaria-causing species.
The kit includes a hinged, book-shaped cardboard device containing the test strip, a reagent product (Tris buffer containing detergent and sodium azide), and capillary tubes for the transfer of whole blood after finger sticks. Other materials that are not provided but are necessary for proper use include negative quality-control samples (3-5 EDTA whole blood samples), positive quality-control samples (EDTA samples containing P falciparum), lancets, sterile wipes, and a stopwatch.4 TABLE 1 gives directions for use.

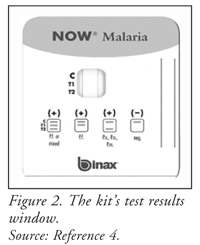
The cardboard device (FIGURE 2) provides a window for viewing results. The control line (C) must appear for a test to be valid. Results are interpreted as follows: T1+, test positive for P falciparum; T2+, positive for single or mixed infection of P vivax, P ovale, or P malariae; T1- and T2+, positive for P falciparum and possible mixed infection with other species; and C, negative result. Negative and possible mixed infection results must be confirmed by microscopic examination.4
Clinical studies of the BinaxNOW Malaria Test Kit show an overall sensitivity:specificity ratio of 99.7%:94.2% for P falciparum. Studies also indicate a sensitivity:specificity ratio of 93.5%:97.8% for P vivax, the second most infective species.5 A study by Wiese et al compared the effectiveness of the BinaxNOW malaria test performed at bedside and in the clinical microbiology laboratory. The study included 542 patients suspected of having malaria, 80 of whom were diagnosed with the disease through microscopy. When used at bedside, the test had a sensitivity of 88% for the detection of P falciparum versus 95% when the test was performed in the microbiology laboratory.6 The risk of technical problems and invalid results was highest when the test was administered at bedside.6
Conclusion
Although the WHO and other prominent organizations have made great strides to eradicate malaria, the disease remains a serious problem worldwide. The BinaxNOW Malaria Test Kit may serve as a much-needed diagnostic tool that allows treatment to be provided in a timely manner. The sensitivity and specificity of the test are slightly lower than those for microscopy, but the test's rapid diagnostic capabilities and its ease of use make it valuable for the detection of malaria.
For more information about this product, contact Inverness Medical Professional Diagnostics at 207-730-5750.
REFERENCES
1. World Malaria Report 2008. Geneva, Switzerland: World Health Organization; 2008.
2. Global Malaria Prevention and Control Program: Moving Ahead in the 21st Century. Atlanta, GA: Centers for Disease Control and Prevention.
3. Chisholm-Burns M, Wells B, Schwinghammer T, et al, eds. Pharmacotherapy Principles & Practice. New York, NY: McGraw-Hill; 2008.
4. BinaxNOW Malaria Test Kit product instructions. Scarborough, ME: Inverness Medical Professional Diagnostics; September 2007.
5. Inverness Medical BinaxNOW Web site. BinaxNOW® Malaria.
www.binaxnow.com/malaria.aspx. Accessed June 9, 2009.
6. Wiese L, Bruun B, Bæk L, et al. Bedside diagnosis of imported malaria using the Binax Now malaria antigen detection test. Scand J Infect Dis. 2006;38:1063-1068.






