US Pharm. 2015;40(1):HS5-HS10.
ABSTRACT: Peripheral neuropathy is a serious condition characterized by symmetrical, distal damage to the peripheral nerves that may be caused by several classes of drugs, including chemotherapeutic agents. Chemotherapy-induced peripheral neuropathy (CIPN) is an adverse effect estimated to occur in up to 40% of patients undergoing chemotherapy, with its incidence increasing in patients being treated with multiple agents. Pharmacists play a pivotal role in the prevention and management of CIPN by recommending evidence-based pharmacologic and nonpharmacologic strategies appropriate for the individual patient.
Peripheral neuropathy (PN) is a systemic disease characterized by symmetrical, distal damage to the peripheral nerves that negatively impacts patient quality of life (QOL).1,2 Prolonged symptoms associated with PN can cause pain, interfere with functional ability (e.g., dressing, driving, house-work), and disrupt emotional health.3
The presentation of PN varies according to the type of nerve fibers affected.4 Sensory nerve damage may manifest as sensations of numbness, tingling, stinging, pain, weakness, or burning that occur in a “stocking and glove” distribution throughout the lower extremities and hands.1,4 Additional sensory manifestations include a feeling of “walking on pins and needles,” altered pain threshold, and changes in temperature sensitivity.4 Motor nerve damage often manifests as muscle spasms and muscle wasting, along with loss of muscle dexterity and strength.4 These patients tend to experience difficulty performing activities of daily living such as buttoning a shirt and opening medication bottles. Patients who have developed autonomic nerve damage may experience orthostatic hypotension, constipation, and dryness, which may worsen after exercise, hot baths, and drinking alcohol.5
It is hypothesized that inflammatory cells are involved in the pathophysiology of PN due to their release of pro-inflammatory mediators capable of contributing to neuropathic pain.1,6
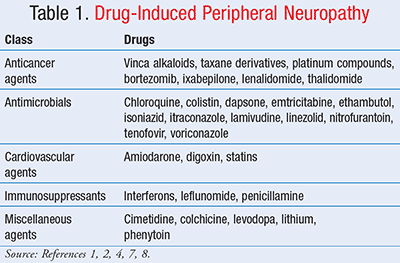
The origin of PN varies with each individual patient. Systemic diseases, metabolic imbalances, nutritional deficiencies, alcoholism, infections, genetic disorders, and drugs all have the potential to cause PN.1 Drug classes identified as having the potential to cause PN include anticancer agents, antimicrobials, cardiovascular agents, immunosuppressants, and other miscellaneous agents (TABLE 1).1,2,4,7
Chemotherapy-Induced Peripheral Neuropathy
Chemotherapy-induced peripheral neuropathy (CIPN) is a severe, toxic effect of many chemotherapy agents including vinca alkaloids, taxane derivatives, and platinum compounds. The anticancer drugs thalidomide, lenalidomide, bortezomib, and ixabepilone have also been implicated in causing PN.2,7 CIPN is classified as a dose-limiting toxicity that negatively impacts patient QOL and chemotherapy treatments, occasionally resulting in dose reductions and/or premature discontinuation of therapy.2,8 This toxicity is estimated to occur in 30% to 40% of patients undergoing chemotherapy, with its incidence increasing in patients treated with multiple chemotherapeutic agents.2,8 The overall incidence of CIPN varies depending on the chemotherapy regimen, dose, treatment duration, patient age, patient race, concomitant therapies, and presence of preexisting conditions with neurotoxic potential.2,8
The mechanism of CIPN is dependent upon the chemotherapeutic agent of interest. Vinca alkaloids (e.g., vincristine, vinblastine, vinorelbine) are antimicrotubule agents derived from the Madagascar periwinkle plant with multiple indications, including malignancies of the hematologic and lymphatic systems as well as solid tumors.7,9 Although the exact mechanism of vinca alkaloid–induced PN is unknown, vincristine has been identified as the most neurotoxic in the class.7 Taxane derivatives (e.g., paclitaxel, docetaxel) are also a class of antimicrotubule agents, which are derived from the bark of the Pacific and European yew conifers and are used to treat breast, ovarian, prostate, lung, gastric, head, and neck malignancies.10 The mechanism of taxane-induced PN is not entirely understood but is believed to be related to disruption of the microtubules and a subsequent effect on axonal transport activity.7
Platinum compounds (e.g., cisplatin, carboplatin, oxaliplatin) possess DNA interstrand cross-linking activity and are indicated for ovarian, testicular, bladder, and colorectal malignancies.8 The mechanism of platinum compound–induced PN is hypothesized to result from its induction of axonal hyperexcitability, alterations in voltage-dependent Na+ channels resulting in continual discharges, and/or oxidative stress.7 Cisplatin-induced PN is more common and severe than PN caused by other drugs in its class. Oxaliplatin-induced PN may manifest in an acute or chronic sensory form.7
Risk factors associated with the development of CIPN include a treatment regimen utilizing the agents previously mentioned; higher initial and cumulative doses; longer treatment durations; advanced age; race (black women have a 2- to 3-fold higher risk of taxane-induced PN compared to white women)8; concomitant therapy (adjunct chemotherapeutic agents or other drugs known to be associated with PN); and preexisting conditions associated with PN development (e.g., diabetes, alcoholism, nonalcoholic liver disease, HIV infection, peripheral vascular disease, amyloidosis, nutritional deficiencies).2,8,11
The onset and duration of CIPN signs and symptoms have a variable time line, beginning weeks to months after treatment initiation and peaking upon, or after, treatment discontinuation.7 In some patients, PN signs and symptoms continue after treatment cessation and can be permanent.7 The highly variable nature of CIPN manifestation requires individualized management strategies.
Diagnosis
A diagnosis of CIPN should be based on the results of both subjective and objective assessment methods. A subjective evaluation should include patient accounts, prior and current treatments (both chemotherapeutic and nonchemotherapeutic agents), and social history.4 Objective evaluation strategies may include a physical examination, laboratory tests, standardized questionnaires, electrodiagnostic studies, and nerve biopsy. The National Comprehensive Cancer Network (NCCN) details the most common physician-based grading scales used to assess the severity of CIPN. These grading scales include the Ajani Sensory, Eastern Cooperative Oncology Group (ECOG), National Cancer Institute–Common Terminology Criteria for Adverse Events (NCI–CTCAE), and World Health Organization (WHO) systems, which grade CIPN severity on a scale of 0 (normal) to 5 (death).12
Electrodiagnostic studies and nerve biopsies are considered when a diagnosis is unclear after initial diagnostic testing and when patient symptoms persist, especially prior to the initiation of additional chemotherapeutic regimens.1,4 When implemented, nerve biopsies are preferably performed on sural and superficial peroneal nerves, but this practice is uncommon.1
Prevention Strategies
There is currently no strong evidence to support the utilization of pharmacologic agents for prevention of CIPN. Agents such as amifostine, erythropoietin, glutathione, vitamin E, glutamine, and acetyl-L-carnitine (ALC) have all been studied for their efficacy in preventing CIPN. Many of these pharmacologic agents under investigation are hypothesized to target the direct and indirect mechanisms of neurotoxicity but require additional efficacy and safety data before being recommended for use in clinical practice. Although IV calcium/magnesium has been hypothesized to reduce the cumulative toxicity associated with oxaliplatin-based chemotherapy, its use remains controversial. This infusion has been hypothesized to decrease the hyperexcitability of neurons while in the presence of oxaliplatin by increasing extracellular calcium. Recent data show that there is no significant difference between IV calcium/magnesium and placebo regarding neuropathy outcomes.13
Cryotherapy, the application of cold temperatures, is also a potential nonpharmacologic prevention tool for CIPN. This intervention is currently being used in a clinical trial investigating prevention strategies for paclitaxel-induced PN.14
Venlafaxine is the only medication to demonstrate clinical efficacy in preventing oxaliplatin-induced PN, but it is still not recommended for routine use in clinical practice due to its unknown effect on oxaliplatin’s antitumor activity.2 Patients are encouraged to consume a healthy, balanced diet and abstain from alcohol as additional preventive strategies for CIPN.8
Clinical Management
Treatment of CIPN should focus on minimizing symptoms and preventing further neurotoxicity in affected patients. A comprehensive treatment approach that includes pharmacologic and nonpharmacologic therapies may be warranted in treating this serious condition.
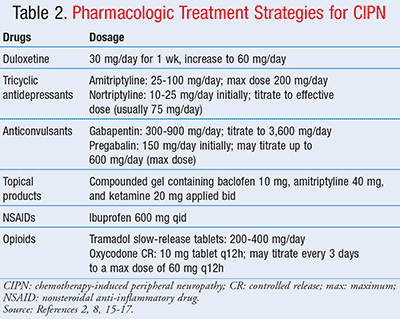
Pharmacologic Therapy: Treatment strategies include duloxetine, tricyclic antidepressants (TCAs), anticonvulsants, compounded topical products, nonsteroidal anti-inflammatory drugs (NSAIDs), and opioid therapy (TABLE 2).2,8,15-17 The serotonin-norepinephrine reuptake inhibitor (SNRI) duloxetine is considered the first-line agent for treatment of CIPN.2 Duloxetine is the only pharmacologic therapy recommended in the American Society of Clinical Oncology (ASCO) clinical practice guidelines for treatment of CIPN in patients with cancer.8 While duloxetine, TCAs, anticonvulsants, and compounded topical products have been investigated for their efficacy in treating CIPN, NSAIDs and opioids have only been investigated for their efficacy in treating diabetic neuropathy and generalized neuropathic pain; further studies are needed to evaluate their specific benefits for patients experiencing CINV.8,15-17
In a 2013 randomized, phase III, double-blind, crossover clinical trial that compared duloxetine versus placebo in the treatment of paclitaxel- and oxaliplatin-induced PN, duloxetine demonstrated a significantly greater reduction in pain scores (determined using the self-administered Brief Pain Inventory–Short Form questionnaire) than placebo (-1.06 vs. -0.34, P = .003).2 Patients in the treatment group received 30 mg of duloxetine daily for 7 days followed by an increased dose of 60 mg daily until study completion. Additional study findings included improved QOL and daily functioning in the duloxetine group without significant differences in adverse-effect incidence when compared to the placebo group. It is important to note, however, that patients in the duloxetine group reported more moderate-intensity adverse effects (fatigue, somnolence, insomnia) compared to those in the placebo group, which resulted in a higher dropout rate (11% vs. 1%).2
Although there is limited high-quality, consistent scientific evidence for the use of TCAs (amitriptyline, nortriptyline), anticonvulsants (gabapentin, pregabalin), and topical products (compounded gel containing baclofen, amitriptyline, and ketamine) in CIPN treatment, it is considered reasonable for clinicians to consider these agents as long as patients are well-informed about their limited evidence, adverse effects, benefits, and costs. The combination of these adjuvant agents with standard doses of opioids can be used to reduce the risk of complications (e.g., constipation, dizziness, nausea, vomiting, respiratory depression, tolerance, physical dependence) seen in patients using high-dose opioids to alleviate CIPN symptoms.2,18
Nutraceuticals, which are isolated/purified food components with possible health benefits, have also been studied for efficacy in the treatment of CIPN.19,20 In a 2013 systematic literature review, ALC, alpha lipoic acid, calcium, glutamine, glutathione, magnesium-acetylcysteine, omega-3 fatty acids, vitamin B6, and vitamin E were evaluated from a pool of 24 studies to determine their individual efficacies in the treatment of CIPN. Efficacy results were mixed for the nutraceutical treatments. This review demonstrates the need for more evidence on nutraceutical efficacy prior to recommendation in clinical practice.19
Nonpharmacologic Therapy: Due to the limited high-quality evidence on pharmacologic treatment options for CIPN, it is critical that clinicians and patients regularly discuss nonpharmacologic treatment strategies for relieving painful and uncomfortable symptoms associated with this toxicity. Nonpharmacologic treatment options with published evidence of efficacy in patients with CIPN include chemotherapy dose reduction, discontinuation of the offending chemotherapeutic agent(s), acupuncture, avoiding cold temperatures/maintaining warmth, physical activity, and massaging affected areas (TABLE 3).6,21,22 In a 10-week pilot study conducted in 2011 to determine the therapeutic benefit of acupuncture on CIPN, five out of six patients treated with acupuncture showed improvement in symptoms.21

As reported in the ASCO clinical practice guidelines, a systematic review of seven clinical studies evaluating the effectiveness of acupuncture in the treatment of CIPN was conducted by Franconi et al.8 These seven clinical studies had varying designs and methodologies. The review provided evidence that acupuncture may be useful in improving symptoms and pain for patients experiencing CIPN, but this evidence is considered to be limited. Larger, randomized, controlled clinical trials are needed to evaluate the benefits of acupuncture for CIPN.
Transcutaneous electrical nerve stimulation (TENS), also discussed in the ASCO clinical practice guidelines, is currently being investigated for its efficacy in alleviating pain associated with CIPN.8 Although a small, randomized, placebo-controlled trial did not demonstrate the efficacy of scrambler therapy (a patient-specific TENS device) in treating CIPN, a small pilot study was able to show improved pain scores (59% reduction at 10 days, P <.001) in 16 patients with CIPN who received TENS via the MC5-A Calmare device. No adverse effects were reported by patients enrolled in this pilot study.8
In a 2013 mixed-methods, descriptive, cross-sectional study on self-care strategies for CIPN reported by colorectal cancer survivors treated with oxaliplatin, a host of self-management strategies were documented that warrant further investigation for nonpharmacologic treatment options that are easily accessible, safe, and effective.22 Physical therapy and exercise activities are specific treatment interventions that require further research to determine the type, frequency, and duration of activities beneficial for alleviating CIPN symptoms in affected patients.22
Conclusion
PN is a serious condition that can be caused by several classes of medications, including chemotherapeutic agents. Pharmacists may play a pivotal role in the prevention of CIPN by recommending evidence-based pharmacologic and nonpharmacologic strategies appropriate for the individual patient, including a healthful diet and alcohol abstinence. There is inadequate evidence to recommend IV calcium/magnesium, cryotherapy, or venlafaxine for use in clinical practice.
Once CIPN has been detected, pharmacists may assist in developing treatment strategies by providing evidence-based recommendations, specifically duloxetine. TCAs, gabapentin, and a topical gel treatment containing baclofen, amitriptyline, and ketamine are pharmacologic treatment options that may also be tried if patients are adequately educated about the limited evidence, risks, and benefits of these products. Nonpharmacologic treatment options include chemotherapy dose reduction, discontinuation of the offending chemotherapeutic agent(s), acupuncture, avoiding cold temperatures/maintaining warmth, physical activity, and massaging the affected areas. CIPN is a serious dose-limiting toxicity that requires individualized prevention and treatment strategies in patients with cancer in order to minimize negative impact on patient QOL.
REFERENCES
1. Azhary H, Farooq MU, Bhanushali M, et al. Peripheral neuropathy: differential diagnosis and management. Am Fam Physician. 2010;81(7): 887-892.
2. Piccolo J, Kolesar JM. Prevention and treatment of chemotherapy-induced peripheral neuropathy. Am J Health-Syst Pharm. 2014;71:19-25.
3. Sasane M, Tencer T, French A, et al. Patient-reported outcomes in chemotherapy-induced peripheral neuropathy: a review. J Support Oncol. 2010;8(6):e15-e21.
4. Kraychete DC, Sakata RK. Painful peripheral neuropathies. Rev Bras Anestesiol. 2011;61(5):641-658.
5. Misra UK, Kalita J, Nair PP. Diagnostic approach to peripheral neuropathy. Ann Indian Acad Neurol. 2008;11(12):89-97.
6. Thacker MA, Clark AK, Marchand F, McMahon SB. Pathophysiology of peripheral neuropathic pain: immune cells and molecules. Anesth Analg. 2007;105:838-847.
7. Vilholm OJ, Christensen AA, Zedan AH, Itani M. Drug-induced peripheral neuropathy. Basic Clin Pharmacol Toxicol. 2014;115:185-192.
8. Hershman DL, Lacchetti C, Dworkin RH, et al. Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guidelines. J Clin Oncol. 2014;32(18):1941-1967.
9. Boulikas T, Pantos A, Bellis E, Christofis P. Designing platinum compounds in cancer: structures and mechanisms. Cancer Therapy. 2007;5:537-583.
10. Park HJ. Chemotherapy-induced peripheral neuropathic pain. Korean J Anesthesiol. 2014;67(1):4-7.
11. Miltenburg NC, Boogerd W. Chemotherapy-induced neuropathy: a comprehensive survey. Cancer Treat Rev. 2014;40(7):872-882.
12. Stubblefield MD, Burstein HJ, Burton AW, et al. NCCN Task Force report: management of neuropathy in cancer. J Natl Compr Canc Netw. 2009;7(suppl 5):S1-S26.
13. Loprinzi Cl, Qin R, Dakhil SR, et al. Phase III randomized, placebo-controlled, double-blind study of intravenous calcium and magnesium to prevent oxaliplatin-induced sensory neurotoxicity (N08CB/Alliance). J Clin Oncol. 2014;32(10):997-1005.
14. Park SB, Goldstein D, Krishnan AV, et al. Chemotherapy-induced peripheral neurotoxicity: a critical analysis. CA Cancer J Clin. 2013;63(6):419-437.
15. Cohen KL, Harris S. Efficacy and safety of nonsteroidal anti-inflammatory drugs in the therapy of diabetic neuropathy. Arch Intern Med. 1987;147(8): 1442-1444.
16. Sindrup SH, Andersen G, Madsen C, et al. Tramadol relieves pain and allodynia in polyneuropathy: a randomised, double-blind, controlled trial. Pain. 1999;83(1):85-90.
17. Gimbel JS, Richards P, Portenoy RK. Controlled-release oxycodone for pain in diabetic neuropathy. Neurology. 2003;60(6):927-934.
18. Benyamin R, Trescot AM, Datta S, et al. Opioid complications and side effects. Pain Physician. 2008;11(2 suppl):S105-S120.
19. Schloss JM, Colosimo M, Airey C, et al. Nutraceuticals and chemotherapy-induced peripheral neuropathy (CIPN): a systematic review. Clin Nutr. 2013;32(6):888-893.
20. Palthur MP, Sajala Palthur SS, Chitta SK. Nutraceuticals: a conceptual definition. Int J Pharm Pharm Sci. 2010;2(3):19-27.
21. Schroeder S, Meyer-Hamme G, Epplee S. Acupuncture for chemotherapy-induced peripheral neuropathy (CIPN): a pilot study using neurography. Acupunct Med. 2012;30(1):4-7.
22. Tofthagen C, Gonzalez L, Visovsky C, Akers A. Self-management of oxaliplatin-related peripheral neuropathy in colorectal cancer survivors. Chemother Res Pract. 2013;2013:547932.
To comment on this article, contact rdavidson@uspharmacist.com.






