US Pharm. 2015;40(1):34-38.
ABSTRACT: Mental health disorders are frequently seen in patients with Parkinson’s disease (PD), possibly as a result of the complex imbalance of neurotransmitters in both disease states. This imbalance poses various treatment challenges, such as the exacerbation of both disease states and drug interactions between the medications used to treat PD and mental health disorders. Owing to these challenges, mental health disturbances in PD patients often go untreated. By assisting with ruling out causative medications and underlying disease states, simplifying antiparkinsonian regimens, and recommending antipsychotics, the pharmacist can help ensure that both of these disease states are adequately treated.
Parkinson’s disease (PD), which impacts millions of people worldwide, is a neurodegenerative disorder involving the deterioration of motor, mental, and functional skills.1 This degenerative decline increases mortality rates and negatively affects patients’ quality of life. Motor movement disorders are heavily emphasized as cardinal signs of PD; however, nonmotor manifestations such as depression, anxiety, and psychosis are major concerns that must be addressed in this patient population.
Mental health (MH) abnormalities are common in PD; more than 60% of patients have one or more psychiatric symptoms.2 Of these abnormalities, depression is the most common neuropsychiatric disturbance in PD, with reported rates of 20% to 90%.3 Besides depression, patients with PD also experience psychosis, impulse-control disorders, mania, and sleep disturbances. The origins of MH disorders in this population have been attributed to both the underlying pathophysiology of PD and the adverse effects of antiparkinsonian drug therapy. These origins more likely lie at the neurotransmitter level and involve the complex interactions between dopamine, acetylcholine, and serotonin.3 This poses a significant treatment challenge, given the dopaminergic, anticholinergic, and central nervous system (CNS)–acting medications used to treat both of these disease states. The object of this article is to outline the major difficulties encountered when providing medication treatment for patients with a dual diagnosis of PD and an MH disorder, as well as to proffer treatment recommendations and considerations.
MAJOR CHALLENGES
When treating patients with PD and a comorbid MH disturbance, it is essential for providers to address both disorders adequately. However, there are many challenges that make it difficult to provide sound treatment for both disease states. The two most common issues are 1) the exacerbation or worsening of both MH and PD symptoms and 2) drug-drug interactions (DDIs) between the agents used to treat the disease states.
Exacerbation of MH Conditions
The medications used to treat PD often cause psychiatric disturbances; specifically, psychosis, impulse-control disorders (ICDs), and mania.
Psychosis: One of the most prominent and best-documented neuropsychiatric symptoms related to antiparkinsonian drug therapy is psychosis. PD-related psychosis is often characterized by visual and auditory hallucinations, illusions, and delusions.4,5 While auditory hallucinations are infrequent, visual hallucinations occur in 20% to 30% of drug-treated PD patients.4-8
The onset of psychosis-related symptoms related to antiparkinsonian drugs may occur soon after initiation or not until after 1 year of therapy.5,9 Early onset of psychosis is more likely in patients with a past medical history of psychiatric illness, and the psychosis resolves after drug discontinuation or dosage reduction.9
The antiparkinsonian medications associated with psychosis include dopaminergics and anticholinergics, amantadine, and monoamine oxidase B inhibitors (MAOI-Bs) (FIGURE 1).5,10-13 Of these, dopaminergics (e.g., levodopa and bromocriptine) have the highest potential for inducing psychosis in PD patients.14 Levodopa exhibits dose-related psychiatric symptoms in a high proportion of PD patients.5,14,15
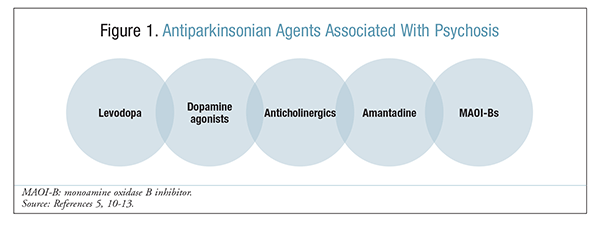
There are various theories about the mechanism by which antiparkinsonian drugs cause psychosis. Both PD and schizophrenia (symptoms of psychosis) result from a dysfunction of dopamine in the CNS. PD is characterized by the loss of midbrain dopamine, whereas the hallucinations and delusions associated with schizophrenia are caused by increased dopamine in the mesolimbic pathways.16 Therefore, when PD is treated with agents designed to increase dopamine for improved movement, there may be an exacerbation of psychosis. Furthermore, chronic stimulation of dopamine receptors by dopamine and/or dopamine agonists is thought to induce dopamine receptor site hypersensitivity, which may also play a role in psychosis.17
ICDs and Mania: Antiparkinsonian medications have been found to cause symptoms of ICDs and mania. The development of ICDs in patients with PD is well documented.18 ICDs, which are characterized by the failure to resist a risky behavior such as compulsive sexual behavior, binge or compulsive eating, compulsive buying, or pathologic gambling, are often associated with levodopa and dopamine agonist treatment. ICDs are thought to be caused by dopaminergic overstimulation of the mesocorticolimbic circuit. In a study by Weintraub and colleagues, dopamine agonist treatment in PD patients was associated with a twofold to 3.5-fold increase in the likelihood of having an ICD.18 Other factors that independently increased ICD occurrence were younger age and a family history of gambling problems.18
Documentation of the onset of mania or hypomania in patients receiving antiparkinsonian medications remains low.19 Unlike ICDs, which mostly present as compulsive behaviors, mania often involves a broader range of symptoms, including mood elevation, pressured speech, inflated self-esteem, and decreased sleep without subsequent daytime somnolence. There are several reports of euphoria and mania development in PD patients treated with levodopa, pramipexole, ropinirole, or selegiline.20-22 These symptoms often resolved after discontinuation or reduction of the offending agent.19,20,22
Exacerbation of PD Symptoms
Treating MH comorbidities, especially psychosis, in patients with PD often leads to an exacerbation of PD symptoms. First-generation antipsychotics (FGAs) and second-generation antipsychotics (SGAs) have been linked to an increase in PD symptoms, owing primarily to prolonged use of dopamine receptor blockers. PD patients may experience worsening of extrapyramidal symptoms (EPS), including akathisia, parkinsonism, dystonia, and tardive dyskinesia (TD).23
TD develops in approximately 20% of patients on long-term antipsychotic therapy, and there is some debate over a lower incidence with SGAs.23 It can present after brief exposure to a dopamine-blocking agent, but mostly develops after months or years of therapy.23 The comparatively lesser severity of EPS with SGAs is postulated to be due to dual serotonin-dopamine receptor antagonism.6 The risk of TD development in PD patients poses a problem for therapy.
SGAs are thought to have a more favorable side-effect profile than FGAs. The mechanisms of these agents will be discussed in the treatment recommendations section.
DDIs
There are multiple DDIs between the medications used to treat PD and MH conditions. The most common DDIs occur between MAOI-Bs and antidepressants and between dopaminergics and antipsychotics/anticholinergics (TABLE 1).

MAOI-Bs and Antidepressants: MAOI-Bs such as selegiline (Eldepryl) and rasagiline (Azilect) are used to manage PD. Although MAOI-Bs inhibit dopamine metabolism, they cause serotonin toxicity when given with other serotonergic agents, such as selective serotonin reuptake inhibitors (SSRIs).24 Therefore, MAOI-Bs should be used cautiously and only at recommended dosages with all antidepressants, including tricyclic antidepressants and SSRIs.25 Antidepressants without serotonergic properties, such as bupropion, also may interact with MAOI-Bs. This is due to the potential for increasing dopamine levels, which can lead to further hallucinations or psychotic behaviors.
Dopaminergics and Antipsychotics: Levodopa, the current major therapeutic agent for most PD patients, is converted to dopamine.26,27 This enables the possibility of various drug interactions with agents that inhibit or modulate dopamine pathways, such as those used to manage schizophrenia. Therefore, when dopaminergic medications for PD are given with antipsychotics, they can antagonize the effect of the medications for each disease state.
TREATMENT RECOMMENDATIONS FOR DUAL DIAGNOSIS
Although the treatment of PD patients with an MH comorbidity or exacerbation can be a challenge, there are recommended steps to ensure that each disease state is adequately treated.
The first step in treating PD patients with an MH condition or disturbance is to rule out underlying conditions and causative nonantiparkinsonian medications, followed by simplification of the PD medication regimen.28,29 The reduction or removal of causative antiparkinsonian medications can facilitate the treatment of psychosis, ICDs, and mania.18-22 If a loss of motor function occurs in a patient undergoing dosage or medication reduction, the addition of an antipsychotic may be considered. These steps are outlined in FIGURE 2.
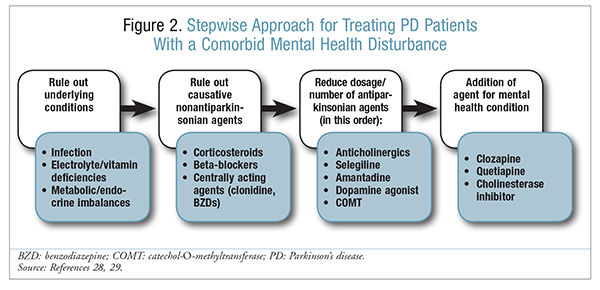
Because of their antagonistic effects on dopamine—which is essential for smooth motor function—antipsychotics have the potential to worsen movement, especially in PD (which is characterized by low dopamine). Movement disorders can occur with both FGAs and SGAs, but the likelihood is greater with FGAs, owing to their high affinity for D2 receptors.16,30 SGAs have a lower risk of EPS owing to their lower affinity for and antagonistic activity on D2 receptors and their antagonistic effects on serotonin via 5-hydroxytryptamine 2a.31 The antagonistic action of serotonin increases dopamine levels in the striatum, countering the reduction of dopamine produced by D2 antagonist activity and thereby reducing EPS. SGAs with higher D2 antagonistic activity, such as risperidone and paliperidone, should be avoided in PD patients.
The most frequently used and studied antipsychotics for PD patients are clozapine and quetiapine.29,32-34 Both of these agents, which are structurally similar, have higher serotonergic antagonistic activity and lower D2 agonistic activity. Clozapine has been shown in various clinical trials to effectively treat psychosis without worsening motor symptoms. However, the use of this medication is usually avoided because of the risk of severe agranulocytosis and the frequent monitoring required. In general, clozapine improves tremor, does not worsen other motor functions to a significant extent, and is safe at low dosages.35 Quetiapine has been shown to be effective in the treatment of PD psychosis without significant worsening of motor function, although evidence of efficacy in clinical trials has been conflicting. Compared with clozapine, quetiapine tends to be favored by prescribers owing to the lack of laboratory monitoring and absent risk of agranulocytosis. Additional precautions are given in TABLE 2.
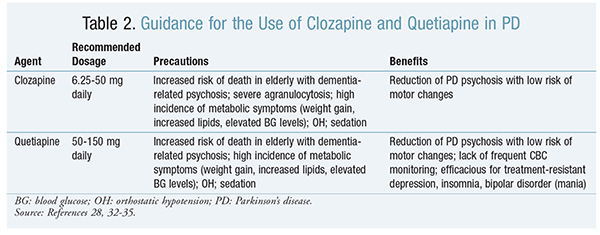
Cholinesterase inhibitors, such as rivastigmine, may be considered. These agents serve as an alternative to antipsychotics in patients with mild-to-moderate psychosis.28 Rivastigmine has been found to be consistently effective for the treatment of PD psychosis.28,36,37 Trials also demonstrate that rivastigmine is more effective than donepezil.6,31
CONCLUSION
Patients with a dual diagnosis of PD and an MH disturbance are a complex population to treat. The common challenges encountered when treating these patients include possible exacerbation of the MH condition and/or the PD symptoms, and DDIs between therapies. Because of these complexities, MH conditions in PD patients are often left untreated.38 As a member of the multidisciplinary team, the pharmacist can play a key role in optimizing treatment of patients with this dual diagnosis by facilitating communication between neurology and MH professionals, providing education on DDIs, and assisting with treatment-regimen designs. Using this approach can ensure that each disease state is adequately treated, thereby improving patient outcomes and ultimately benefiting quality of life.
REFERENCES
1. Dorsey ER, Constantinescu R, Thompson JP, et al. Projected number of people with Parkinson disease in the most populous nations, 2005 through 2030. Neurology. 2007;68:384-386.
2. Schrag A. Psychiatric aspects of Parkinson’s disease—an update. J Neurol. 2004;251:795-804.
3. Fernandez HH. Updates in the medical management of Parkinson disease. Cleve Clin J Med. 2012;79:28-35.
4. Mendis T, Barclay CL, Mohr E. Drug-induced psychosis in Parkinson’s disease: a review of management. CNS Drugs. 1996;5:166-174.
5. Moskovitz C, Moses H III, Klawans HL. Levodopa-induced psychosis: a kindling phenomenon. Am J Psychiatry. 1978;135:669-675.
6. Zahodne LB, Fernandez HH. Pathophysiology and treatment of psychosis in Parkinson’s disease: a review. Drugs Aging. 2008;25:665-682.
7. Inzelberg R, Kipervasser S, Korczyn AD. Auditory hallucinations in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1998;64:533-535.
8. Goetz CG, Vogel C, Tanner CM, Stebbins GT. Early dopaminergic drug-induced hallucinations in parkinsonian patients. Neurology. 1998;51:811-814.
9. Aarons S, Peisah C, Wijeratne C. Neuropsychiatric effects of Parkinson’s disease treatment. Australas J Ageing. 2012;31:198-202.
10. Serby M, Angrist B, Lieberman A. Mental disturbances during bromocriptine and lergotrile treatment of Parkinson’s disease. Am J Psychiatry. 1978;135:1227-1229.
11. de Smet Y, Ruberg M, Serdaru M, et al. Confusion, dementia and anticholinergics in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 1982;45:1161-1164.
12. Harper RW, Knothe BU. Coloured Lilliputian hallucinations with amantadine. Med J Aust. 1973;1:444-445.
13. Klein C, Kömpf D, Pulkowski U, et al. A study of visual hallucinations in patients with Parkinson’s disease. J Neurol. 1997;244:371-377.
14. Seeman P, Niznik HB. Dopamine receptors and transporters in Parkinson’s disease and schizophrenia. FASEB J. 1990;4:2737-2744.
15. Willner P. The dopamine hypothesis of schizophrenia: current status, future prospects. Int Clin Psychopharmacol. 1997;12:297-308.
16. Lieberman JA. Dopamine partial agonists: a new class of antipsychotic. CNS Drugs. 2004;18:251-267.
17. Klawans HL, Crossett P, Dana N. Effect of chronic amphetamine exposure on stereotyped behavior: implications for pathogenesis of l-dopa-induced dyskinesias. Adv Neurol. 1975;9:105-112.
18. Weintraub D, Koester J, Potenza MN, et al. Impulse control disorders in Parkinson disease: a cross-sectional study of 3090 patients. Arch Neurol. 2010;67:589-595.
19. Maier F, Merkl J, Ellereit AL, et al. Hypomania and mania related to dopamine replacement therapy in Parkinson’s disease. Parkinsonism Relat Disord. 2014;20:421-427.
20. Kim E, Zwil AS, McAllister TW, et al. Treatment of organic bipolar mood disorders in Parkinson’s disease. J Neuropsychiatry Clin Neurosci. 1994;6:181-184.
21. Singh A, Althoff R, Martineau RJ, Jacobson J. Pramipexole, ropinirole, and mania in Parkinson’s disease. Am J Psychiatry. 2005;162:814-815.
22. Thomas N, Jebaraj P, Kishor Kumar S. Bipolar affective disorder in Parkinson’s disease: clinical dilemmas. Indian J Med Sci. 2008;62:374-376.
23. Meyer TA, Belson TE, McAllister R. Tardive dyskinesia: a distressing drug-induced movement disorder. US Pharm. 2014;39(1):HS13-HS16.
24. Sagna A, Gallo JJ, Pontone GM. Systematic review of factors associated with depression and anxiety disorders among older adults with Parkinson’s disease. Parkinsonism Relat Disord. 2014;20:708-715.
25. Richard IH, Kurlan R, Tanner C, et al. Serotonin syndrome and the combined use of deprenyl and an antidepressant in Parkinson’s disease. Parkinson Study Group. Neurology. 1997;48:1070-1077.
26. Dalvi A, Ford B. Antiparkinsonian agents—clinically significant drug interactions and adverse effects, and their management. CNS Drugs. 1998;9:291-310.
27. Bianchine J, Sunyapridakul L. Interactions between levodopa and other drugs: significance in the treatment of Parkinson’s disease. Drugs. 1973;6:364-388.
28. Goldman JG, Holden S. Treatment of psychosis and dementia in Parkinson’s disease. Curr Treat Options Neurol. 2014;16:281.
29. Olanow CW, Watts RL, Koller WC. An algorithm (decision tree) for the management of Parkinson’s disease (2001): treatment guidelines. Neurology. 2001;56(11 suppl 5):S1-S88.
30. Fernandez HH, Friedman JF. The role of atypical antipsychotics in the treatment of movement disorders. CNS Drugs. 1999;11:467-483.
31. Lieberman JA, Mailman RB, Duncan G, et al. Serotonergic basis of antipsychotic drug effects in schizophrenia. Biol Psychiatry. 1998;44:1099-1117.
32. Seppi K, Weintraub D, Coelho M, et al. The Movement Disorder Society Evidence-Based Medicine Review Update: treatments for the nonmotor symptoms of Parkinson’s disease. Mov Disord. 2011;26(suppl 3):S42-S80.
33. Miyasaki JM, Shannon K, Voon V, et al. Practice parameter: evaluation and treatment of depression, psychosis, and dementia in Parkinson disease (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2006;66:996-1002.
34. Friedman JH, Factor SA. Atypical antipsychotics in the treatment of drug-induced psychosis in Parkinson’s disease. Mov Disord. 2000;15:201-211.
35. Factor SA, Friedman JH, Lannon MC, et al. Clozapine for the treatment of drug-induced psychosis in Parkinson’s disease: results of the 12 week open label extension in the PSYCLOPS trial. Mov Disord. 2001;16:135-139.
36. Reading PJ, Luce AK, McKeith IG. Rivastigmine in the treatment of parkinsonian psychosis and cognitive impairment: preliminary findings from an open trial. Mov Disord. 2001;16:1171-1174.
37. Ravina B, Putt M, Siderowf A, et al. Donepezil for dementia in Parkinson’s disease: a randomised, double blind, placebo controlled, crossover study. J Neurol Neurosurg Psychiatry. 2005;76:934-939.
38. Dobkin RD, Rubino JT, Friedman J, et al. Barriers to mental health care utilization in Parkinson’s disease. J Geriatr Psychiatry Neurol. 2013;26:105-116.
To comment on this article, contact rdavidson@uspharmacist.com.





