US Pharm. 2018;43(11):21-25.
ABSTRACT: Occasional nightmares are fairly common, but nightmare disorder occurs in 2% to 6% of adults. Recurrent nightmares may be idiopathic, but they are often related to posttraumatic stress disorder (PTSD), underlying psychiatric disorders, or medication use. The American Academy of Sleep Medicine’s 2018 position paper provides guidance on nonpharmacologic and pharmacologic treatment. Behavioral intervention with imagery-rehearsal therapy is currently the only treatment strategy recommended for all patients with recurrent nightmares. Prazosin may be used to treat both PTSD-associated and idiopathic nightmare disorder. Antidepressants, anxiolytics, anticonvulsants, antipsychotics, and other agents have been studied, with mixed results.
Nightmares are vivid, disturbing, or frightening nocturnal episodes that often involve intense feelings of dread, humiliation, or failure. The International Classification of Sleep Disorders, Third Edition (ICSD-3), defines nightmare disorder as a parasomnia (i.e., abnormal or unusual nervous-system behavior during sleep) usually associated with rapid-eye-movement sleep. The minimal diagnostic criteria include recurrent episodes of awakenings from disturbed dreams, full alertness on awakening with clear recall of dream, and either delayed return to sleep or occurrence of the episode in the second half of the sleep period.1-3
Epidemiology and Etiology
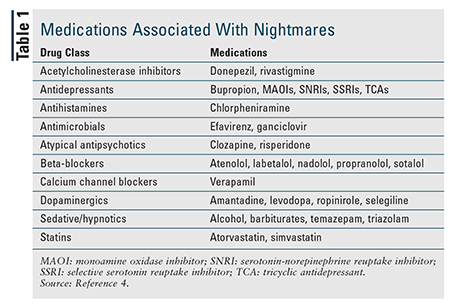
Occasional nightmares are relatively common, but nightmare disorder affects between 2% and 6% of adults.1,3 It is more common in younger adults than in older adults. More women than men have nightmare disorder during young adulthood, but there is no difference in those aged 60 years and older.1,3 Although they can be idiopathic, recurrent nightmares are often related to posttraumatic stress disorder (PTSD) and other psychiatric disorders, and PTSD patients—up to 80% of whom experience nightmares—have been studied the most regarding nightmares.3 Medications that affect neurotransmitter levels in the central nervous system are also associated with nightmares (TABLE 1).4
Therapy Selection
In 2018, the American Academy of Sleep Medicine (AASM) published a position paper on the treatment of nightmare disorder that replaced their best-practice guide from 2010.3 Treatment options are designated recommended or not recommended if they are clearly useful or harmful based on clinical evidence. The may be used position is designated when evidence is less clear. The AASM further differentiates treatments for PTSD-associated nightmares and nightmares without a clear etiology (which is termed nightmare disorder).3 Although selection of therapy depends on the clinician and the patient’s access to resources, pharmacists should be aware of the various treatment options discussed in the position paper.
Nonpharmacologic Treatment
The only treatment strategy with enough evidence to be recommended in the AASM’s position paper is behavioral intervention with imagery-rehearsal therapy (IRT).3 IRT is based on the theory that nightmares are a learned behavior and can be replaced by a less disruptive behavior that will not ultimately affect sleep or daytime functioning. Patients are asked to recall the nightmare, write it down, alter its content to a positive outcome, and rehearse the rescripted dream for 10 to 20 minutes each day. Additional treatments that may be used for both PTSD-associated nightmares and nightmare disorder include cognitive behavioral therapy and exposure, relaxation, and rescripting therapies. Other behavioral strategies include hypnosis, lucid-dreaming therapy, eye-movement desensitization and reprocessing, and progressive deep-muscle relaxation techniques.3
Pharmacologic Treatment
Although no pharmacologic agent is recommended in the position paper, many of the medications discussed are designated may be used.3 TABLE 2 provides a detailed summary of these agents, along with available trial data. Prazosin remains the drug of choice and is the only one indicated for both nightmare types.3 Therefore, prazosin will be discussed first, followed by the remaining agents and drug classes in alphabetical order.
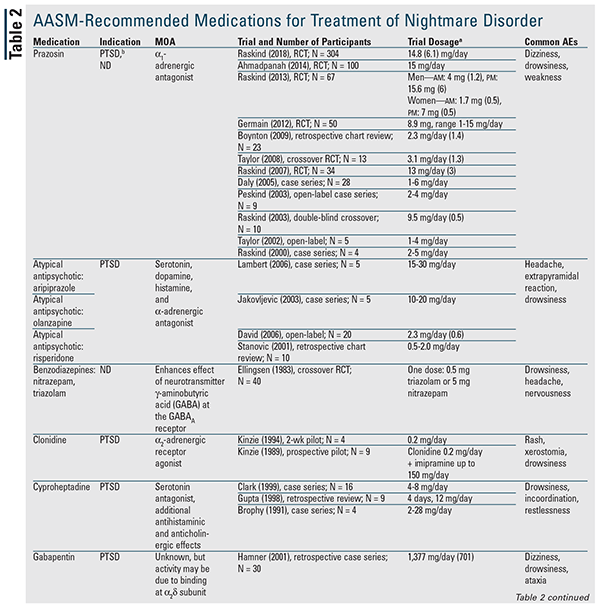
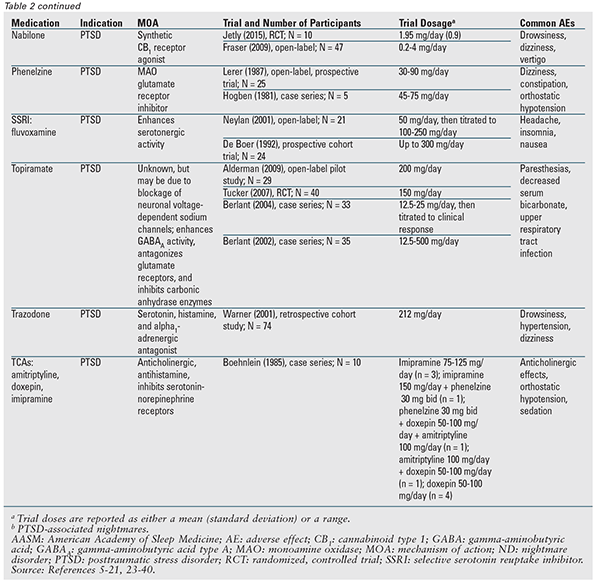
Prazosin: The 2010 AASM best-practice guide recommended prazosin for nightmare disorder; however, the current position paper has downgraded its classification to may be used based on a recent publication that did not find a statistical difference versus placebo.3,5 This trial had the largest patient population to date and was the first to show a lack of benefit with prazosin; however, the majority of patients in both groups were concurrently receiving an antidepressant.5-16 This is important because a prior trial noted a decreased response to prazosin in patients concurrently taking a selective serotonin reuptake inhibitor (SSRI).7 Further clarification of this possible interaction is needed.
Atypical Antipsychotics: In small studies, aripiprazole, olanzapine, and risperidone have been evaluated as adjunctive treatments for PTSD, and all of these agents have demonstrated some benefit for the associated nightmares. These medications, however, are limited by their adverse-effect profile.17-20
Benzodiazepines: Nitrazepam and triazolam were assessed in a single 3-day trial in which patients with disturbed sleep (nightmare type was not identified) reported a decrease in “unpleasant dreams.”21 Patients took just one dose of each medication, followed by a 1-day washout period. Clonazepam is currently not recommended because it was found to be ineffective for PTSD-associated nightmares in a randomized clinical trial.22
Clonidine: The two studies of clonidine conducted in PTSD patients had positive results; however, there were only 13 participants.23,24
Cyproheptadine: Three small trials of cyproheptadine in PTSD patients had conflicting data. Adverse effects may outweigh the benefit.25-27
Gabapentin: A single retrospective study of gabapentin in patients with PTSD showed a marked or moderate improvement in sleep, as well as a decreased frequency or intensity of nightmares.28
Nabilone: In a single open-label study, the majority of PTSD patients receiving nabilone experienced cessation of nightmares or a significant reduction in nightmare intensity. A smaller randomized trial also found a decreased incidence of PTSD-related nightmares.29,30
Phenelzine: Two studies of phenelzine in PTSD patients indicated a benefit. However, all patients in the larger study ultimately withdrew because the improvement in nightmare severity was negligible, was short-lived, or plateaued.31,32
SSRIs and Serotonin-Norepinephrine Reuptake Inhibitors: Of these agents, only fluvoxamine is designated may be used. Two small clinical studies of fluvoxamine showed benefit; however, in one study, many patients withdrew because of side effects.3,33,34 Venlafaxine has shown benefit for general PTSD symptoms, but not nightmares; therefore, it is not recommended.1,3
Topiramate: Despite positive results in several studies of PTSD-associated nightmares, topiramate use may be limited because of adverse effects.3,35-38
Trazodone: The only study to evaluate trazodone found it to be effective, but 19% of patients were unable to maintain an effective dose, and many experienced at least one side effect.39
Tricyclic Antidepressants (TCAs): One small case series in patients with PTSD suggested beneficial effects; however, findings were limited because TCA treatment was varied.40
The Pharmacist’s Role
Pharmacist awareness of nightmare disorder is becoming increasingly important. As the number of patients with PTSD continues to rise owing to war, mass shootings, and other traumatic events, the incidence of nightmare disorder will increase accordingly. Because patients may be hesitant or embarrassed to discuss nightmares with their primary care provider, pharmacists should learn to recognize the symptoms and common causes of nightmare disorder so that they can identify patients who may need referral for evaluation and treatment. Pharmacists should also be familiar with the pharmacologic agents that may be used to treat this condition so that they can appropriately counsel patients dealing with this disorder.
REFERENCES
1. Nadorff MR, Lambdin KK, Germain A. Pharmacological and non-pharmacological treatments for nightmare disorder. Int Rev Psychiatry. 2014;26:225-236.
2. American Academy of Sleep Medicine. International Classification of Sleep Disorders. 3rd ed. Darien, IL: American Academy of Sleep Medicine; 2014.
3. Morgenthaler TI, Auerbach S, Casey KR, et al. Position paper for the treatment of nightmare disorder in adults: an American Academy of Sleep Medicine position paper. J Clin Sleep Med. 2018;14(6):1041-1055.
4. Foral P, Knezevich J, Dewan N, Malesker M. Medication-induced sleep disturbances. Consult Pharm. 2011;26:414-425.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. New Engl J Med. 2018;378(6):507-517.
6. Ahmadpanah M, Sabzeiee P, Hosseini SM, et al. Comparing the effect of prazosin and hydroxyzine on sleep quality in patients suffering from posttraumatic stress disorder. Neuropsychobiology. 2014;69(4):235-242.
7. Raskind MA, Peterson K, Williams T, et al. A trial of prazosin for combat trauma PTSD with nightmares in active-duty soldiers returned from Iraq and Afghanistan. Am J Psychiatry. 2013;170(9):1003-1010.
8. Germain A, Richardson R, Moul DE, et al. Placebo-controlled comparison of prazosin and cognitive-behavioral treatments for sleep disturbances in US military veterans. J Psychosom Res. 2012;72(2):89-96.
9. Boynton L, Bentley J, Strachan E, et al. Preliminary findings concerning the use of prazosin for the treatment of posttraumatic nightmares in a refugee population. J Psychiatr Pract. 2009;15(6):454-459.
10. Taylor FB, Martin P, Thompson C, et al. Prazosin effects on objective sleep measures and clinical symptoms in civilian trauma posttraumatic stress disorder: a placebo-controlled study. Biol Psychiatry. 2008;63:629-632.
11. Raskind MA, Peskind ER, Hoff DJ, et al. A parallel group placebo controlled study of prazosin for trauma nightmares and sleep disturbance in combat veterans with post-traumatic stress disorder. Biol Psychiatry. 2007;61:928-934.
12. Daly CM, Doyle ME, Radkind M, et al. Clinical case series: the use of Prazosin for combat-related recurrent nightmares among Operation Iraqi Freedom combat veterans. Mil Med. 2005;170(6):513-515.
13. Peskind ER, Bonner LT, Hoff DJ, Raskind MA. Prazosin reduces trauma-related nightmares in older men with chronic posttraumatic stress disorder. J Geriatr Psychiatry Neurol. 2003;16(3):165-171.
14. Raskind MA, Peskind ER, Kanter ED, et al. Reduction of nightmares and other PTSD symptoms in combat veterans by prazosin: a placebo-controlled study. Am J Psychiatry. 2003;160(2):371-373.
15. Taylor F, Raskind MA. The alpha1-adrenergic antagonist prazosin improves sleep and nightmares in civilian trauma posttraumatic stress disorder. J Clin Psychopharmacol. 2002;22:82-85.
16. Raskind MA, Dobie DJ, Kanter ED, et al. The alpha1-adrenergic antagonist prazosin ameliorates combat trauma nightmares in veterans with posttraumatic stress disorder: a report of 4 cases. J Clin Psychiatry. 2000;61(2):129-133.
17. Lambert MT. Aripiprazole in the management of post-traumatic stress disorder symptoms in returning Global War on Terrorism veterans. Int Clin Psychopharmacol. 2006;21(3):185-187.
18. Jakovljevic M, Sagud M, Mihaljevic-Peles A. Olanzapine in the treatment-resistant, combat-related PTSD—a series of case reports. Acta Psychiatr Scand. 2003;107(5):394-396.
19. David D, De Faria L, Mellman TA. Adjunctive risperidone treatment and sleep symptoms in combat veterans with chronic PTSD. Depress Anxiety. 2006;23(8):489-491.
20. Stanovic JK, James KA, Vandevere CA. The effectiveness of risperidone on acute stress symptoms in adult burn patients: a preliminary retrospective pilot study. J Burn Care Rehabil. 2001;22(3):210-213.
21. Ellingsen PA. Double-blind trial of triazolam 0.5 mg vs. nitrazepam 5 mg in outpatients. Acta Psychiatr Scand. 1983;67(3):154-158.
22. Cates ME, Bishop MH, Davis LL, et al. Clonazepam for treatment of sleep disturbances associated with combat-related posttraumatic stress disorder. Ann Pharmacother. 2004;38(9):1895-1899.
23. Kinzie JD, Sack RL, Riley CM. The polysomnographic effects of clonidine on sleep disorders in posttraumatic stress disorder: a pilot study with Cambodian patients. J Nerv Ment Dis. 1994;182(10):585-587.
24. Kinzie JD, Leung P. Clonidine in Cambodian patients with posttraumatic stress disorder. J Nerv Ment Dis. 1989;177(9):546-550.
25. Clark RD, Canive JM, Calais LA, et al. Cyproheptadine treatment of nightmares associated with posttraumatic stress disorder. J Clin Psychopharmacol. 1999;19(5):486-487.
26. Gupta S, Popli A, Bathurst E, et al. Efficacy of cyproheptadine for nightmares associated with posttraumatic stress disorder. Compr Psychiatry. 1998;39(3):160-164.
27. Brophy M. Cyproheptadine for combat nightmares in post-traumatic stress disorder and dream anxiety disorder. Mil Med. 1991;156(2):100-101.
28. Hamner MB, Brodrick PS, Labbate LA. Gabapentin in PTSD: a retrospective, clinical series of adjunctive therapy. Ann Clin Psychiatry. 2001;13(3):141-146.
29. Jetly R, Heber A, Fraser G, Boisvert D. The efficacy of nabilone, a synthetic cannabinoid, in the treatment of PTSD-associated nightmares: a preliminary randomized, double-blind, placebo-controlled cross-over design study. Psychoneuroendocrinology. 2015;51:585-588.
30. Fraser GA. The use of a synthetic cannabinoid in the management of treatment-resistant nightmares in posttraumatic stress disorder (PTSD). CNS Neurosci Ther. 2009;15:84-88.
31. Lerer B, Bleich A, Kotler M, et al. Posttraumatic stress disorder in Israeli combat veterans. Effect of phenelzine treatment. Arch Gen Psychiatry. 1987;44(11):976-981.
32. Hogben GL, Cornfield RB. Treatment of traumatic war neurosis with phenelzine. Arch Gen Psychiatry. 1981;38(4):440-445.
33. Neylan TC, Metzler TJ, Schoenfeld FB, et al. Fluvoxamine and sleep disturbances in posttraumatic stress disorder. J Trauma Stress. 2001;14(3):461-467.
34. De Boer M, Op den Velde W, Falger PJ, et al. Fluvoxamine treatment for chronic PTSD: a pilot study. Psychother Psychosom. 1992;57(4):158-163.
35. Alderman CP, McCarthy LC, Condon JT, et al. Topiramate in combat-related posttraumatic stress disorder. Ann Pharmacother. 2009;43(4):635-641.
36. Tucker P, Trautman RP, Wyatt DB, et al. Efficacy and safety of topiramate monotherapy in civilian posttraumatic stress disorder: a randomized, double-blind, placebo-controlled study. J Clin Psychiatry. 2007;68(2):201-206.
37. Berlant JL. Prospective open-label study of add-on and monotherapy topiramate in civilians with chronic nonhallucinatory posttraumatic stress disorder. BMC Psychiatry. 2004;4:24.
38. Berlant J, van Kammen DP. Open-label topiramate as primary or adjunctive therapy in chronic civilian posttraumatic stress disorder: a preliminary report. J Clin Psychiatry. 2002;63(1):15-20.
39. Warner M, Dorn M, Peabody C. Survey on the usefulness of trazodone in patients with PTSD with insomnia or nightmares. Pharmacopsychiatry. 2001;34(4):128-131.
40. Boehnlein JK, Kinzie JD, Ben R, Fleck J. One year follow-up study of posttraumatic stress disorder among survivors of Cambodian concentration camps. Am J Psychiatry. 1985;42:956-959.
To comment on this article, contact rdavidson@uspharmacist.com.





