US Pharm. 2019;44(3):22-26.
ABSTRACT: Although nonsteroidal anti-inflammatory drugs (NSAIDs) are one of the most commonly prescribed and consumed drug classes, they are associated with a wide range of adverse effects, including NSAID-induced hypersensitivity reactions (NHRs). There are five types of NHRs, categorized as either nonimmunologically mediated or immunologically mediated reactions. Symptoms associated with these reactions can range from respiratory and/or cutaneous reactions to angioedema. Pharmacists should be aware of and understand the differences associated with the various NHR reaction types.
Nonsteroidal anti-inflammatory drugs (NSAIDs) are among the most commonly used analgesic, anti-inflammatory, and antipyretic medications on the market. There are currently more than 15 different forms of NSAIDs commercially available in the United States, and they are available as both prescription and OTC drugs. Despite the fact that they are often consumed daily, their use comes with a broad variety of adverse effects ranging from gastrointestinal irritation to severe, life-threatening anaphylaxis. NSAID-induced hypersensitivity reactions (NHRs) have been reported in about 0.3% to 6% of the general population and are thought to account for 20% to 30% of all drug-related reactions, placing them second behind antibiotic-associated hypersensitivity reactions.1-4
Studies show that patients at highest risk for developing NHRs are those with underlying chronic respiratory and skin conditions. In fact, up to 30% of all patients with chronic respiratory and/or cutaneous diseases will be affected by NHRs.5-8 Although NHRs are more prevalent in patients with these conditions, people who do not have these conditions may also be affected.
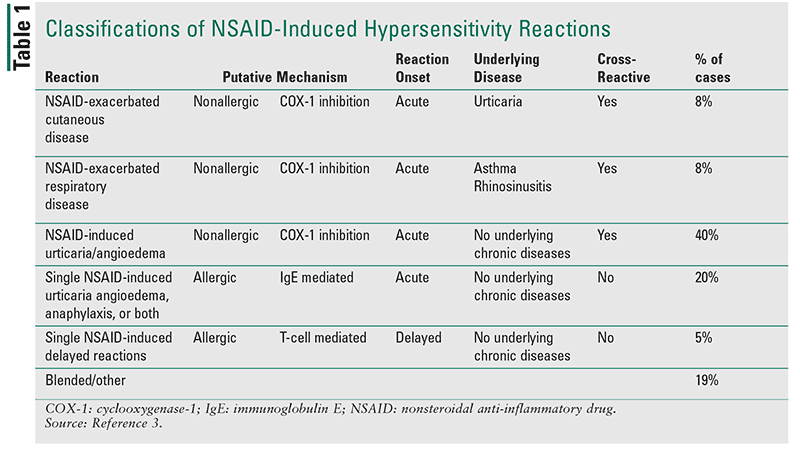
Because of the frequency and complexity of NHRs, international collaborative efforts have been made to help classify and manage these reactions. The 2013 classification and management strategies set forth by the European Academy of Allergy and Clinical Immunology are the most widely accepted.1 These classifications are first divided into two groups: those that are nonimmunologically mediated (nonallergic or cross-reactive reactions) versus those that are immunologically mediated (allergic or selective reactions). They are then further subdivided by the presence of underlying disease and timing of reaction (Table 1).1
Nonallergic NSAID-Induced Hypersensitivity Reactions
Nonallergic NSAID reactions are allergy-like reactions that are not immunologically mediated. These reactions are thought to occur mainly due to the inhibition of cyclooxygenase-1 enzymes (COX-1). COX-1 inhibition shifts the arachidonic acid pathway from the production of the protective anti-inflammatory prostaglandins toward the production of the proinflammatory mediators, eosinophils, and mast cells. These proinflammatory agents can then induce the overproduction of cysteinyl leukotrienes (CysLTs), which causes an increase of CysLTs receptor expression in the bronchial muscles, as well as the endothelial and epithelial cells. This can further lead to the induction or exacerbation of respiratory and/or cutaneous reactions.2,3,9
Nonallergic NSAID reactions are known to be cross-reactive, and therefore, patients who experience them can have a reaction to any NSAID that strongly inhibits the COX-1 enzyme, including aspirin (Table 2). On the other hand, highly selective cyclooxygenase-2 enzyme (COX-2) inhibiting NSAIDs and weak COX-1 inhibiting NSAIDs are usually well tolerated in this patient population.1

There are three types of nonallergic NHRs. NSAIDs-exacerbated cutaneous disease (NECD) and NSAIDs-induced urticaria/angioedema (NIUA) reactions induce cutaneous symptoms; NSAIDs-exacerbated respiratory disease (NERD) induces respiratory symptoms.1
NECD: NECD reactions occur in patients with underlying chronic spontaneous urticaria (CSU) who either develop urticaria or have an urticaria exacerbation following the ingestion of an NSAID. NECD reactions make up approximately 8% of all NHR cases, and it has been reported that about 10% to 30% of all patients with CSU will experience an exacerbation of their underlying disease following NSAID consumption.2,9 Symptoms are primarily limited to the dermis and subcutaneous layer of the skin with no respiratory involvement.9,10
Symptoms normally appear within 30 minutes to 4 hours after ingestion of the NSAID. However, delayed symptoms occurring up to 24 hours after ingestion have been reported.2,11,12 The magnitude of NSAID-induced symptoms is dose-dependent and is greater when chronic urticaria symptoms are already active.1 Symptoms usually subside within a few hours but may last up to several days. Repeated exposure to strongly inhibiting COX-1 NSAIDs, including aspirin, can lead to uncontrollable CSU.1 Therefore, all COX-1 inhibitors should be avoided in patients with NECD history. The selective COX-2 inhibitor celecoxib is usually well tolerated but should be initiated in a clinically supervised setting to confirm tolerability.1,2
The effects of NECD reactions have previously been reported to be dose-dependent.1 Therefore, patients who require aspirin for primary or secondary cardiovascular protection and have a history of NECD tend to respond better to the lower (cardio-protective) doses of aspirin (81 mg) than they do to the higher doses.5,13 Although data are limited, the use of pretreatment antihistamine therapy to help reduce the effects of NECD reactions has also been reported.5,14 In one retrospective study, patients who experienced recurrent pain and had a known history of NECD were pretreated with a long-acting H1-antihistamine 30 minutes prior to the administration of an NSAID.14 The study concluded that pretreatment with antihistamines may help improve tolerability of low-dose COX-1 inhibitors.
NIUA: NIUA reactions occur in otherwise healthy individuals (those without an underlying history of CSU) and manifest with the development of urticaria and/or angioedema after the administration of at least two chemically unrelated NSAIDs. An estimated 40% of all NHRs and 60% of nonimmunologic type reactions are thought to be NIUA reactions.9
Although delayed reactions have been reported, symptoms usually develop within 30 to 90 minutes following ingestion.11,15 Symptoms will often manifest as simultaneous angioedema and urticaria; however, patients who develop NIUA reactions can present with either alone. Almost without fail, all strongly COX-1–inhibiting NSAIDs will cause symptoms in patients who develop NIUA reactions. High doses of acetaminophen ( ≥1,000 mg), which weakly inhibits COX-1, may also cause NIUA in up to 25% of patients.1,11 However, the highly selective COX-2 inhibitor celecoxib is generally well tolerated.1
Initially, to prevent cross-reactivity and recurrence, it is recommended to avoid all NSAIDs with strong COX-1 inhibition in patients with a history of NIUA. If NSAID therapy is required for analgesic or anti-inflammatory treatment, low-dose weak COX-1 inhibitors or highly selective COX-2 inhibitors can be tried.1
Patients with a history of NIUA who require chronic aspirin for its cardiovascular benefit may receive aspirin treatment after desensitization (ATAD).13,16,17 Aspirin desensitization occurs by slowly increasing aspirin dosages given to the patient in an attempt to eliminate a pharmacologic and immunologic reaction. Several protocols for aspirin desensitization have been published and should be performed under the supervision of an experienced practitioner.2,13,18,19 Depending on urgency and clinical setting, desensitization could be attempted over several hours to days. Desensitization should only be performed for patients requiring chronic use because daily administration is necessary to maintain aspirin desensitization.
Although NIUA reactions have likely been documented for many years as part of an allergy profile, reassessment after a time may be considered. In one study, approximately 63% of patients with a true, confirmed NIUA reaction tolerated NSAIDs within 72 months after the previous reaction, even without desensitization.20
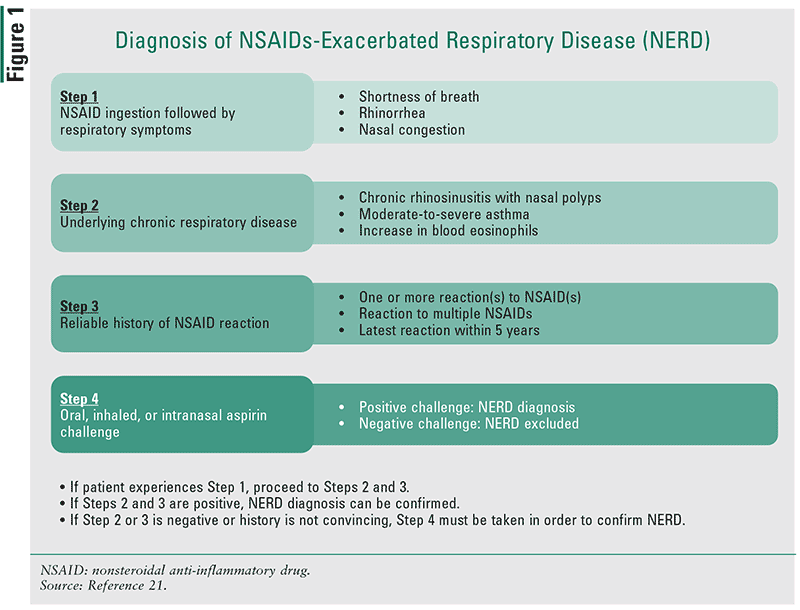
NERD: NERD reactions are characterized by the development of respiratory symptoms such as chest tightness, bronchospasms, rhinitis, nasal polyposis, and/or nasal congestion after the ingestion of one or more NSAIDs. This condition occurs in patients with underlying lower or upper chronic respiratory diseases (Figure 1).1,21 Since NERD is also considered a chronic eosinophilic inflammatory disorder, these patients will often have higher-than-normal blood eosinophils––following an exacerbation. The prevalence of NERD varies from 4% to 20% depending on the study population, with patients having severe asthma disease and those of female gender having higher prevalence rates.21,22 Symptoms generally occur within 30 minutes to 3 hours after ingestion of NSAIDs.
As with NIUA, avoidance of strongly COX-1–inhibiting NSAIDs after the development of NERD is recommended to prevent cross-reactivity. Low-dose, weak COX-1–inhibiting agents (e.g., acetaminophen ≤1,000 mg) and highly selective COX-2 inhibitors are generally well tolerated; however, a small percentage (<2%) of patients with unstable asthma may have a reaction.1 In patients who need chronic aspirin or NSAID therapy, ATAD may be considered as a therapeutic option in the management of NERD patients (Table 3). ATAD has been associated with a reduction in chronic respiratory symptoms, intranasal corticosteroid use, nasal polyps, and asthma exacerbations.23 Asthma exacerbations and allergic rhinosinusitis in patients who develop NERD reactions should be managed as directed by international guidelines.24,25
Allergic NSAID-Induced
Hypersensitivity Reactions
Allergic NHRs are non–cross-reactive reactions, immunologically mediated based on immunoglobulin E (IgE) or T-cell response. These reactions are not dependent on COX-1 inhibition and can be induced by a single NSAID or by a class of NSAIDs with similar chemical structures. There are two predominant types of allergic NHRs, which include single NSAID-induced urticaria/angioedema, anaphylaxis, or both (SNIUAA) reactions and single NSAID-induced delayed reactions (SNIDRs). These reactions differ based on timing (acute or delayed) and are not associated with any underlying chronic disease states.1
SNIUAA: SNIUAA reactions make up more than 20% of all NHRs and are characterized by urticaria, angioedema, or anaphylaxis following the ingestion of a single NSAID or NSAIDs with similar chemical structures.9 Symptoms of SNIUAA generally occur acutely, within seconds to minutes. The timing and spectrum of symptoms associated with SNIUAA reactions are suggestive of a type 1 immunologic reaction, and specific IgE antibodies can usually be detected in skin tests of some patients.1,26 All NSAIDs can cause SNIUAA reactions. Diclofenac and aspirin have been the most implicated NSAIDs in the U.S.27
Diagnosis of SNIUAA is determined in patients without underlying cutaneous disease who have immediate reactions following ingestion of a single NSAID. To differentiate SNIUAA reactions from nonallergic reactions (e.g., NIUA), these patients can be given an oral challenge with a chemically unrelated strong COX-1 inhibitor. Strict avoidance of the culprit agent and agents with similar structures is highly recommended (Table 2). Patients with SNIUAA will be able to safely tolerate other NSAIDs with different chemical structures. Desensitization in this type of reaction is not well documented and is not recommended.1,2
SNIDRs: Unlike SNIUAA reactions, SNIDRs are delayed reactions occurring 24 to 48 hours after the exposure to a single oral or topical NSAID. The exact prevalence of SNIDRs is unknown but is thought to be less than 5%.1,9 The most common symptoms associated with this reaction are maculopapular eruptions, contact dermatitis, and photosensitivity reactions. Other severe cutaneous reactions—such as Stevens-Johnson syndrome/toxic epidermal necrolysis and drug reaction with eosinophilia—and systemic symptoms can occur but are also less frequently induced in SNIDRs.28 Clinical symptoms vary depending on the involved agent. For example, ibuprofen and naproxen are most frequently associated with maculopapular eruptions, while diclofenac and ketoprofen are more commonly associated with contact dermatitis.3 Selective COX-2 inhibitors are more associated with severe cutaneous reactions.29,30 Patients who experience SNIDRs should avoid the culprit NSAID and agents with similar chemical structures.
Pharmacist’s Role
NSAIDs, whether prescription or OTC, are one of the most frequently consumed classes of medications. In addition to the other adverse reactions associated with them, pharmacists should be aware of NHRs and understand the differences between the classifications. Since NHRs can occur among NSAIDs with similar chemical structures or similar mechanisms of action, it is essential for pharmacists to educate patients about the various NSAIDs that can be consumed. Patients should be provided written information that includes potentially cross-reactive agents and safe alternatives. They should be encouraged to carry this written information with them at all times. Pharmacists can play a key role in educating and directing patients to consult their providers about desensitization if chronic NSAID or aspirin therapy is needed.
REFERENCES
1. Kowalski ML, Asero R, Bavbek S, et al. Classification and practical approach to the diagnosis and management of hypersensitivity to nonsteroidal anti-inflammatory drugs. Allergy. 2013;68(10):1219-1232.
2. Laidlaw TM, Cahill KN. Current knowledge and management of hypersensitivity to aspirin and NSAIDs. J Allergy Clin Immunol Pract. 2017;5(3):537-545.
3. Kulthanan K, Jiamton S, Boochangkool K, Jongjarearnprasert K. Angioedema: clinical and etiological aspects. Clin Dev Immunol. 2007;26438.
4. Leeyaphan C, Kulthanan K, Jongjarearnprasert K, Dhana N. Drug-induced angioedema without urticaria: prevalence and clinical features. J Eur Acad Dermatol Venereol. 2010;24(6):685-691.
5. Kowalski ML, Woessner K, Sanak M. Approaches to the diagnosis and management of patients with a history of nonsteroidal anti-inflammatory drug-related urticaria and angioedema. J Allergy Clin Immunol. 2015;136(2):245-251.
6. Doeglas HM. Reactions to aspirin and food additives in patients with chronic urticaria, including the physical urticarias. Br J Dermatol. 1975;93(2):135-144.
7. Moore-Robinson M, Warin RP. Effect of salicylates in urticaria. Br Med J. 1967;4(5574):262-264.
8. Blumenthal KG, Lai KH, Huang M, Wallace ZS. Adverse and hypersensitivity reactions to prescription nonsteroidal anti-inflammatory agents in a large health care system. J Allergy Clin Immunol Pract. 2017;5(3):737-743.
9. Doña I, Salas M, Perkins JR, et al. Hypersensitivity reactions to non-steroidal anti-inflammatory drugs. Curr Pharm Des. 2016;22(45):6784-6802.
10. Sánchez-Borges M, Caballero-Fonseca F, Capriles-Hulett A, González-Aveledo L. Aspirin-exacerbated cutaneous disease (AECD) is a distinct subphenotype of chronic spontaneous urticaria. J Eur Acad Dermatol Venereol. 2015;29(4):698-701.
11. Doña I, Blanca-López N, Cornejo-García JA, et al. Characteristics of subjects experiencing hypersensitivity to non-steroidal anti-inflammatory drugs: patterns of response. Clin Exp Allergy. 2011;41(1):86-95.
12. Szczeklik A, Gryglewski RJ, Czerniawska-Mysik G. Clinical patterns of hypersensitivity to nonsteroidal anti-inflammatory drugs and their pathogenesis. J Allergy Clin Immunol. 1977;60(5):276-284.
13. Modena B, White AA, Woessner KM. Aspirin and nonsteroidal anti-inflammatory drugs hypersensitivity and management. Immunol Allergy Clin North Am. 2017;37(4):727-749.
14. Trautmann A, Anders D, Stoevesandt J. H1-antihistamine premedication in NSAID-association urticaria. J Allergy Clin Immunol Practice. 2016;4(6):1205-1212.
15. Doña I, Blanca-López N, Jagemann LR, et al. Response to a selective COX-2 inhibitor in patients with urticaria/angioedema induced by nonsteroidal anti-inflammatory drugs. Allergy. 2011;66(11):1428-1433.
16. McMullan KL, Wedner HJ. Safety of aspirin desensitization in patients with reported aspirin allergy and cardiovascular disease. Clin Cardiol. 2013;36(1):25-30.
17. Slowik SM, Slavin RG. Aspirin desensitization in a patient with aspirin sensitivity and chronic idiopathic urticaria. Ann Allergy Asthma Immunol. 2009;102(2):171-172.
18. White AA, Stevenson DD, Woessner KM, Simon RA. Approach to patients with aspirin hypersensitivity and acute cardiovascular emergencies. Allergy Asthma Proc. 2013;34(2):138-142.
19. Lee RU, White AA, Ding D, et al. Use of intranasal ketorolac and modified oral aspirin challenge for desensitization of aspirin-exacerbated respiratory disease. Ann Allergy Asthma Immunol. 2010;105(2):130-135.
20. Doña I, Barrionuevo E, Salas M, et al. Natural evolution in patients with nonsteroidal anti-inflammatory drug-induced urticaria/angioedema. Allergy. 2017;72(9):1346-1355.
21. Kowalski ML, Agache I, Bavbek S, et al. Diagnosis and management of NSAID-exacerbated respiratory disease (NERD): a EAACI position paper. Allergy. 2019:74(1)28-39.
22. Makowska JS, Burney P, Jarvis D, et al. Respiratory hypersensitivity reactions to NSAIDs in Europe: the global allergy and asthma network (GA-LEN) survey. Allergy. 2016;71(11):1603-1611.
23. White AA, Stevenson DD. Aspirin-exacerbated respiratory disease. N Engl J Med. 2018;379(11):1060-1070.
24. Brožek JL, Bousquet J, Agache I, et al. Allergic rhinitis and its impact on asthma (ARIA) guidelines-2016 revision. J Allergy Clin Immunol. 2017;140(4):950-958.
25. Bateman ED, Hurd SS, Barnes PJ, et al. Global strategy for asthma management and prevention: GINA executive summary. Eur Respir J. 2008;31:143-178.
26. Woessner KM, Castells M. NSAID single-drug-induced reactions. Immunol Allergy Clin North Am. 2013;33(2):237-249.
27. Sánchez-Borges M, Caballero-Fonseca F, Capriles-Hulett A, González-Aveledo L. Hypersensitivity reactions to nonsteroidal anti-inflammatory drugs: an update. Pharmaceuticals. 2010;3(1):10-18.
28. Mockenhaupt M, Kelly JP, Kaufman D, Stern RS. The risk of Stevens-Johnson syndrome and toxic epidermal necrolysis associated with nonsteroidal anti-inflammatory drugs: a multinational perspective. J Rheumatol. 2003;30(10):2234-2240.
29. LaGrenade L, Lee L, Weaver J, et al. Comparison of reporting of Stevens-Johnson syndrome and toxic epidermal necrolysis in association with selective COX-2 inhibitors. Drug Saf. 2005;28(10):917-924.
30. Ward KE, Archambault R, Mersfelder TL. Severe adverse skin reactions to nonsteroidal antiinflammatory drugs: a review of the literature. Am J Health Syst Pharm. 2010;67(3):206-213.
To comment on this article, contact rdavidson@uspharmacist.com.