US Pharm.
2008;33(5):HS-4-HS-9.
Seldom do patients in the
acute-care setting present with the sole issue of pain management. By
definition, pain is an indicator of disease, and its function is to detect and
localize tissue-damaging processes.1 Pain may present as a function
of an underlying disease or as a process separate from the primary diagnosis.
The purpose of this article is
to review acute illnesses that interact with the management of pain, where
misuse of analgesic agents would be detrimental to the patient. Two broad
etiologies can be applied to most of the discussion: increased drug toxicity
due to acute illness and worsened acute illness due to improper analgesic
selection.
Managing Pain in Liver
Disease
Because the liver
is responsible for the metabolism of many drugs, concerns about pain
management in liver disease are similar regardless of the cause of compromise,
whether alcoholic liver disease, hepatitis, cirrhosis, or carcinoma. The
elevation of liver biomarkers such as total and direct bilirubin, alanine
aminotransferase, and aspartate aminotransferase is a red flag for the
alteration of drug therapy.2
Acetaminophen may come to mind
in this setting. The acetaminophen content of several opioid combination
products (e.g., Vicodin, Percocet) must be considered with regard to the
following discussion.
A 50% to 75% dose reduction
from the recommended 4-g maximum daily dose is warranted in hepatic
insufficiency or alcohol use, where the potential for liver damage is already
present.3 In chronic alcohol users, however, the data conflict with
clinical wisdom and what is known about the metabolism of acetaminophen.4
Acetaminophen is metabolized
via several pathways, the most common one being glucuronide and sulfate
conjugation to inactive metabolites. A small portion of the acetaminophen is
eliminated unchanged in the urine; another portion is metabolized by the
CYP450 system--one being isoenzyme 2E1 (CYP2E1)--to a toxic metabolite, N
-acetyl-p-benzoquinone imine (NAPQI). In therapeutic doses, NAPQI is
conjugated with glutathione and rendered nontoxic. When increased NAPQI
concentrations are present, however, glutathione stores are depleted, and the
unconjugated NAPQI causes oxidative hepatocellular injury, liver failure, and
death.5
With acute ingestion, even in
chronic alcohol use, ethanol inhibits CYP2E1 activity, theoretically
minimizing the formation of NAPQI. Chronic use of alcohol induces CYP2E1
activity, theoretically increasing the formation of NAPQI. It has been
suggested that this induction is modest and short-lived, and therefore
clinically insignificant. Acetaminophen should be used with extreme discretion
in these situations.4
Principles of pharmacokinetics
such as first-pass effect and bioavailability account for the necessity of
adjusting the doses of analgesic agents in moderate-to-severe liver disease
(TABLE 1).6 First-pass effect occurs when an oral agent is
absorbed from the gastrointestinal (GI) tract and enters the hepatic
circulation, where an amount of the drug may be metabolized before reaching
the systemic circulation. The ratio of the amount entering the circulation to
the total dose is the drug's bioavailability. When liver disease is present,
the lack of hepatic blood flow results in increased bioavailability and the
need to lower the dose. Opioids with increased bioavailability due to this
phenomenon are morphine and hydromorphone to a large degree, and oxycodone and
fentanyl to a lesser degree.6
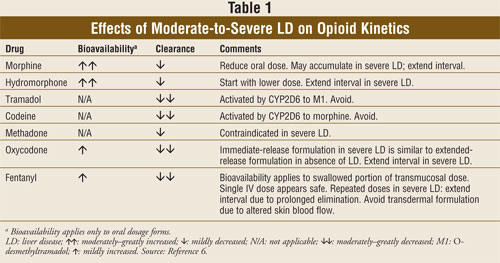
The principle of hepatic
clearance mirrors that of first-pass effect, but here the concern is removal
of an agent from the blood rather than the agent's introduction to the body.
In a fashion similar to that of first-pass effect, when hepatic blood flow is
compromised because of disease, clearance is decreased. This occurs with oral
and parenteral administration of the drug. Extending the drug interval may
help overcome the accumulation associated with decreased clearance. Several of
the opioids are dependent on hepatic clearance: morphine, hydromorphone, and
methadone to a small degree, and tramadol, codeine, oxycodone, and fentanyl to
a greater degree.6
Codeine and tramadol are
unique agents because of their hepatic conversion to their active forms.
Codeine is metabolized to morphine and tramadol to O-desmethyltramadol,
both by CYP2D6; in advanced liver disease, analgesia with these agents can be
unpredictable due to decreased enzyme activity and therefore should not be
used. Transdermal fentanyl also can be unpredictably absorbed due to altered
skin blood flow and should be avoided.6 The other aforementioned
opioids should be used cautiously (TABLE 1).6
Managing Pain in Kidney
Disease
As with the liver,
the kidneys are responsible for clearing many drugs from the body. Insult to
the kidney warrants an examination of drug therapy regardless of etiology.
7,8 With an acute kidney injury, the choice of analgesic drug and dose
becomes extremely important due to the potential for accumulation of certain
analgesics and their metabolites as well as the potential for increased damage
to the kidney. Markers of organ function such as serum creatinine and urine
protein play a major role in signaling a damaging event; however, acute
changes in mental status may be the first indicator of acute kidney failure in
patients receiving chronic opioid therapy for the management of malignant or
nonmalignant pain secondary to decreased drug elimination.9
Due to their lack of active
metabolites and lack of formation of metabolites with weak opioid activity,
the ideal opioids for use in the presence of kidney disease are fentanyl,
hydromorphone, methadone, and oxycodone. Caution is advised when these agents
are used in severe kidney dysfunction, however, as a portion of the parent
drug may be eliminated renally without hepatic deactivation. Opioids that
should be avoided or used with much caution are morphine and codeine (which is
hepatically metabolized to morphine), due to the formation of the active
metabolite morphine-6-glucuronide from both parent drugs. Propoxyphene forms
norpropoxyphene, which accumulates in the presence of kidney failure and can
cause profound central nervous system (CNS) and respiratory depression.9
Meperidine should be avoided at all costs due to the formation of the active
metabolite normeperidine, which accumulates in the presence of kidney disease
and causes seizures.9 Normeperidine has been shown to cause
seizures even in the absence of kidney failure.10
Nonsteroidal anti-inflammatory
drugs (NSAIDs) are widely discouraged in both acute and chronic kidney failure
due to blocking of the prostaglandin-mediated effect of renal vasodilation and
the altering of renal blood flow, along with sodium retention and volume
overload.11 While it was formerly hypothesized that
cyclooxygenase-2 (COX-2) inhibitors would circumvent this problem, it has been
shown that COX-2 inhibitors also adversely affect renal blood flow and
vascular tone.12
Adverse Effects of
Analgesic Agents
Cardiovascular (CV)
System: Patients with
atherosclerotic disease, recent coronary-artery bypass (CAB) graft, unstable
angina, myocardial infarction (MI), or ischemic stroke may be at greater risk
for increased CV risk.13 In a meta-analysis, the CV risk of
selective and nonselective COX inhibitors was reviewed.14
Rofecoxib, which was associated with the highest relative risk, has been
removed from the U.S. market; valdecoxib, which was contraindicated in
patients undergoing CAB surgery, was removed from the U.S. market as well.
14,15 Diclofenac was associated with the highest risk of CV events.
Naproxen and celecoxib at doses <=
200 mg per day were not associated with an increased risk of CV events.14
In the management of acute
pain, acetaminophen should be tried initially unless it is contraindicated. An
opioid-acetaminophen combination or an opioid alone should be considered if
acetaminophen alone is insufficient. The decision to use a selective COX-2
inhibitor or nonselective NSAID must be carefully weighed against the
potential harm associated with its use. Patients undergoing cardiac-bypass
surgery or those at risk for CV events should avoid these medications.13
The use of ibuprofen has been shown to decrease the effectiveness of
concurrent aspirin therapy and thus should be avoided.13 It is
unclear whether the increased risk of CV effects could be attenuated with the
use of daily aspirin. The use of naproxen with a proton-pump inhibitor may be
safer and more cost-effective for the treatment of pain in patients at high
risk for CV events who are at greater risk for GI bleeding.16 Both
selective and nonselective NSAIDs can worsen blood pressure control through
sodium retention, with resultant decreased kidney perfusion.
GI System:
One of the most common side effects of opioid analgesics is constipation. In
acute illness, constipation can contribute to or worsen urinary retention; it
also can lead to nausea, which can further weaken the patient or delay
accurate differential diagnosis of the nausea or vomiting. Tolerance is the
body's reaction to decreased response after continued exposure to a
medication. In most patients taking opioids, tolerance usually develops to the
side effects of nausea, vomiting, and fatigue within five to seven days.
Opioids and NSAIDs can cause anorexia, and NSAIDs can aggravate peptic ulcer
disease or gastroesophageal reflux disease. The NSAID ketorolac is frequently
overlooked as the cause of GI bleeding or renal impairment. Another
complication seen in acute illness is diarrhea. Severe diarrhea can cause
electrolyte disturbances and dehydration, possibly leading to acute renal
insufficiency. Postoperative paralytic ileus, particularly after abdominal
surgery, can be associated with the use of opioid analgesics.17
The choice of opioid analgesic
is usually based on patient-specific factors. Transdermal fentanyl is an
option in nonopioid-naive patients who are unable to swallow oral
formulations.
CNS:
NSAIDs and opioids can contribute to an increased risk of falls, possibly due
to centrally mediated dizziness.18 Propoxyphene has been associated
with an increased risk of falls and hip fractures and is not recommended for
routine use in elderly patients.19 Falls in older patients can lead
to an increased risk of hip fracture and the accelerated downward spiral of
morbidity and mortality.20,21 In older patients with delirium or
dementia, worsened cognition may decrease functional status. The use of
fentanyl, sufentanil, or alfentanil in critically ill patients with baseline
increased intracranial pressure (ICP) can cause a transient increase in ICP.
22
Hematologic System:
Several types of hematologic events can arise in the acute-care setting.
Rates of deep venous thrombosis (DVT) and resulting complications such as
pulmonary embolism, MI, and stroke are important measures of quality in most
health care institutions. In an immobilized patient receiving pain control for
chronic or acute conditions, masking of pain caused by DVT can occur, leading
to one of the sequelae described above.23
Particularly in the
postoperative setting, acute blood loss is quite common among inpatients.
24,25 The use of NSAIDs should be avoided in these instances,
particularly when a patient's hemoglobin is below values considered to be
adequate (e.g., 10 mg/dL).24,25 Nonselective NSAIDs have been shown
to decrease platelet function and increase the propensity for bleeding;
meloxicam and nabumetone have less of an impact on platelet activity, however.
26 COX-2 inhibitors have not been shown to decrease platelet function.
26 Patients predisposed to bleeding, such as those with
thrombocytopenia, or patients on warfarin should avoid the use of NSAIDs.
27,28 Acetaminophen at doses of 2 to 4 g per day has been shown to
increase the international normalized ratio in patients on warfarin.29
Integumentary System:
Transdermal fentanyl is a good option for pain control when oral formulations
are inappropriate or cannot be tolerated in nonopioid-naive patients.
Medication absorption can be less than optimal in patients with increased body
temperature, however.30 Increases in body temperature can occur
through direct application of heat, fever, physical exertion, or application
of the patch to warm skin immediately after a shower or bath.30 Few
cases exist of fentanyl toxicity secondary to an increase in body temperature;
none have been secondary to fever.31-33 Serum fentanyl
concentration can vary in patients for reasons not well understood; one
possibility is that this is due to pharmacogenomic or pharmacokinetic
variability in the population.
Psychiatric Conditions:
While depression is not an acute illness, it is prevalent in patients
experiencing pain, and vice versa; pain coexisting with depression is harder
to manage.34 Treatment of the depression before analgesic therapy
is introduced greatly improves the patient's response to the analgesic.34
Antidepressants have consistently shown benefits in patients with various
types of pain and pain syndromes with or without depression.34 The
preferred agent for neuropathic pain with or without depression is a serotonin
norepinephrine reuptake inhibitor.35 Tricyclic antidepressants are
an option, but they have fallen out of favor due to their adverse-effects
profile and the availability of safer agents.35
One of the fears concerning
the use of opioid analgesics is the potential for dependence and addiction.
36 Dependence and addiction are distinctly different states. Dependence
is a physical reliance on an agent without which a withdrawal syndrome would
be present. For opioids, the withdrawal syndrome includes agitation, insomnia,
increased pain, nausea, vomiting, and palpitations, among other things.37
Addiction is psychological; it combines a craving for opioids with the
existence of impaired control over use, compulsive use, or continued use
despite harm. In the treatment of an opioid-dependent patient in an acute-pain
situation, it is important to keep in mind that the opioid requirement will be
higher.37 A scheduled maintenance dose should be given to prevent
withdrawal, with breakthrough doses given in addition.37 If
increasing the doses of an opioid does not control pain, opioid rotation is
recommended.37 Tramadol appears to have a lower risk of dependence
and addiction; however, in patients previously dependent, it may cause
reemergence of the addiction.38
In pseudoaddiction, a patient
whose pain is inadequately managed engages in drug-seeking behavior to acquire
more drugs for the pain. Superficially, it is nearly impossible to distinguish
pseudoaddiction and true addiction; however, Lusher et al found that behaviors
associated with pseudoaddiction were disputes between patients and staff over
analgesia and the use of OTC analgesics, and behaviors associated with true
addiction were illicit drug use and physiologic dependence.39
Opioids should be prescribed
cautiously in depressed patients with suicidal ideation, particularly on an
outpatient basis.40 The quantity dispensed should be limited, as
intentional overdose can lead to respiratory depression and death.40
Multiple organ failure and subsequent death have been noted in cases of
tramadol overdose.41
Conclusion
The selection of analgesics in the
acute-care setting requires a careful consideration of the coexisting disease
states and the potential for interaction between drug and disease. Care should
be taken to ensure that the changes occurring during an acute illness do not
compromise the safety of the selected analgesic and that the selected
analgesic does not promote the severity of the illness.
REFERENCES
1. Fields HL,
Martin JB. Pain: pathophysiology and management. In: Brunwald E, Fauci AS,
Kasper DL, et al, eds. Harrison's Principles of Internal Medicine. 15th
ed. New York, NY: McGraw-Hill; 2001:55-60.
2. Tegeder I, Lotsch J,
Geisslinger G. Pharmacokinetics of opioids in liver disease. Clin
Pharmacokinet. 1999;37:17-40.
3. AGS Panel on
Persistent Pain in Older Persons. The management of persistent pain in older
persons. J Am Geriatr Soc. 2002;50(suppl 6):S205-S224.
4. Prescott LF.
Paracetamol, alcohol and the liver. Br J Clin Pharmacol.
2000;49:291-301.
5. Chyka PA. Clinical
toxicology. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy:
A Pathophysiologic Approach. 5th ed. New York, NY: McGraw-Hill;
2002:99-121.
6. Davis M. Cholestasis
and endogenous opioids: liver disease and exogenous opioid pharmacokinetics.
Clin Pharmacokinet. 2007;46:825-850.
7. Mueller BA. Acute
renal failure. In: DiPiro JT, Talbert RL, Yee GC, et al, eds.
Pharmacotherapy: A Pathophysiologic Approach. 5th ed. New York, NY:
McGraw-Hill; 2002:771-795.
8. Pai AB, Pai MP.
Acute renal disease. In: Tisdale JE, Miller DA, eds. Drug-Induced Diseases:
Prevention, Detection, and Management. Bethesda, MD: American Society of
Health-System Pharmacists; 2005:583-598.
9. Murphy EJ. Acute
pain management pharmacology for the patient with concurrent renal or hepatic
disease. Anaesth Intensive Care. 2005;33:311-322.
10. Marinella MA.
Meperidine-induced generalized seizures with normal renal function. South
Med J. 1997;90:556-558.
11. Brater DC. Effects
of nonsteroidal anti-inflammatory drugs on renal function: focus on
cyclooxygenase-2 selective inhibition. Am J Med. 1999;107:65S-70S.
12. Cheng HF, Harris
RC. Renal effects of non-steroidal anti-inflammatory drugs and selective
cyclooxygenase-2 inhibitors. Curr Pharm Des. 2005;11:1795-1804.
13. Bennett JS,
Daugherty A, Herrington D, et al. The use of nonsteroidal anti-inflammatory
drugs (NSAIDs): a science advisory from the American Heart Association.
Circulation. 2005;111:1713-1716.
14. McGettigan P, Henry
D. Cardiovascular risk and inhibition of cyclooxygenase: a systematic review
of the observational studies of selective and nonselective inhibitors of
cyclooxygenase. JAMA. 2006;296:1633-1644.
15. Nussmeier NA,
Whelton AA, Brown MT, et al. Complications of the COX-2 inhibitors parecoxib
and valdecoxib after cardiac surgery. N Eng J Med. 2005;352:1081-1091.
16. Velentgas P, West
W, Cannuscio CC, et al. Cardiovascular risk of selective cyclooxygenase-2
inhibitors and other non-aspirin non-steroidal anti-inflammatory medications.
Pharmacoepidemiol Drug Saf. 2006;15:641-652.
17. Goettsch WG, Sukel
MP, van der Peet DL, et al. In-hospital use of opioids increases rate of coded
postoperative paralytic ileus. Pharmacoepidemiol Drug Saf.
2007;16:668-674.
18. Vestergaard P,
Rejnmark L, Mosekilde L. Fracture risk associated with the use of morphine and
opiates. J Intern Med. 2006;260:76-87.
19. Kamal-Bahl SJ,
Stuart BC, Beers MH. Propoxyphene use and risk for hip fractures in older
adults. Am J Geriatr Pharmacother. 2006;4:219-226.
20. Haentjens P, Autier
P, Barette M, et al. Survival and functional outcome according to hip fracture
type: a one-year prospective cohort study in elderly woman with an
intertrochanteric or femoral neck fracture. Bone. 2007;41:958-964.
21. Vestergaard P,
Rejnmark L, Mosekilde L. Has mortality after a hip fracture increased? J Am
Geriatr Soc. 2007;55:1720-1726.
22. Albanese J, Viviand
X, Potie F, et al. Sufentanil, fentanyl, and alfentanil in head trauma
patients: a study on cerebral hemodynamics. Crit Care Med.
1999;27:407-411.
23. Meyer GS, Eagle KA.
Patient-controlled analgesia masking pulmonary embolus in a postoperative
patient. Crit Care Med. 1992;20:1619-1621.
24. Eauno P, Petersen
KD, Husted SE. Increased blood loss after preoperative NSAID. Retrospective
study of 186 hip arthroplasties. Acta Orthop Scand. 1993;64:522-524.
25. Robinson CM,
Christie J, Malcolm-Smith N. Non-steroidal anti-inflammatory drugs,
perioperative blood loss, and transfusion requirements in elective hip
arthroplasty. J Arthroplasty. 1993;8:607-610.
26. van Kraaij DJ,
Hovestad-Witterland AH, de Metz M, Vollaard EJ. A comparison of the effects of
nabumetone vs meloxicam on serum thromboxane B2 and platelet function in
healthy volunteers. Br J Clin Pharmacol. 2002;53:644-647.
27. Kenny GNC.
Potential renal, haematological and allergic adverse effects associated with
nonsteroidal anti-inflammatory drugs. Drugs. 1992;44:31-37.
28. Wells PS, Holbrook
AM, Crowther NR, et al. Interactions of warfarin with drugs and food. Ann
Intern Med. 1994;121:676-683.
29. Parra D, Beckey NP,
Stevens GR. The effect of acetaminophen on the international normalized ratio
in patients stabilized on warfarin therapy. Pharmacotherapy.
2007;27:675-683.
30. Duragesic (fentanyl
transdermal system) package insert. Titusville, NJ: Ortho-McNeil-Janssen
Pharmaceuticals, Inc; February 2008.
31. Frolich M,
Giannotti A, Modell JH, Frolich M. Opioid overdose in a patient using a
fentanyl patch during treatment with a warming blanket. Anesth Analg.
2001;93:647-648.
32. Newshan G.
Heat-related toxicity with the fentanyl transdermal patch. J Pain Symptom
Manage. 1998;16:277-278.
33. Rose PG, Macfee MS,
Boswell MV. Fentanyl transdermal system overdose secondary to cutaneous
hyperthermia. Anesth Analg. 1993;77:390-391.
34. Bair MJ, Robinson
RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review.
Arch Intern Med. 2003;163:2433-2445.
35. Jann MW, Slade JH.
Antidepressant agents for the treatment of chronic pain and depression.
Pharmacotherapy. 2007;27:1571-1587.
36. Drayer RA,
Henderson J, Reidenberg M. Barriers to better pain control in hospitalised
patients. J Pain Symptom Manage. 1999;17:434-440.
37. Mehta V, Langford
RM. Acute pain management for opioid dependent patients. Anaesthesia.
2006;61:269-276.
38. Ultram (tramadol
HCl tablets) package insert. Raritan, NJ: Ortho-McNeil Pharmaceutical, Inc;
February 2007.
39. Lusher J, Elander
J, Bevan D, et al. Analgesic addiction and pseudoaddiction in painful chronic
illness. Clin J Pain. 2006;22:316-324.
40. CDC. Toxicology
testing and results for suicide victims--13 states, 2004. MMWR.
2006;55:1245-1248.
41. De Decker K,
Cordonnier J, Jacobs W, et al. Fatal intoxication due to tramadol alone: case
report and review of the literature. Forensic Sci Int. 2008;175:79-82.
To comment on this article, contact
rdavidson@jobson.com.





