US Pharm. 2016;41(2):30-34.
ABSTRACT: Implantable cardiac devices are a mainstay in treating cardiac diseases and include pacemakers, cardioverter defibrillators, and cardiac resynchronization therapy. Some drugs can interfere with these devices, while certain medications can prevent inappropriate shocks from the devices. Care should be taken to treat and prevent infections from the devices appropriately. Cardiac device implantation should be done cautiously in patients on anticoagulants. Pharmacists should be aware of the different types of implantable cardiac devices and how medications can affect their efficacy and safety.
Implantable cardiac devices are a mainstay in treating cardiac diseases and preventing sudden cardiac death. There are several types of these devices, including pacemakers (PMs), implantable cardioverter defibrillators (ICDs), and cardiac resynchronization therapy (CRT).
Implantable cardiac devices, which consist of a generator and wires (leads), are implanted via a minimally invasive procedure in a cardiac catheterization laboratory or operating room. The leads are inserted into a vein and guided to their destination in the heart. They are attached to both the heart muscle and the generator, which is placed in a pocket under the skin in the upper chest.1-3 These devices require batteries to function. Lithium batteries allow an average battery life span up to 5 to 10 years before a replacement is needed.4,5
A population-based analysis of cardiac device implantation found that yearly implants of ICDs increased 145% between 1997 and 2004.6 PMs had an increased implantation of 24% during that time. In 2004, 67,000 ICDs and 179,000 PMs were implanted. CRT was approved in 2001. By 2004, approximately 68,000 CRTs had been implanted in patients. Of patients who received a device, 60% of ICD recipients, 85% of PM recipients, and 70% of CRT recipients were aged ≥65 years, and 75% of them had more than one comorbidity.6
In 2004, ICDs were the most expensive of the devices at almost $115,000.6 From 2007 to 2011 the cost of CRT and PMs decreased approximately 26% each, while ICD prices decreased about 24%.7 Currently, almost 400,000 cardiac devices are implanted yearly.8 The purpose of this article is to review the types of implantable cardiac devices and their association with medications.
Pacemakers
PMs are implantable devices that send electrical shocks to the heart in response to an abnormal heart rate. When a patient’s pulse exceeds or drops below the PM’s programmed rate, the device will send electrical currents to stimulate the heart and return the pulse to a normal rate.9 There are various types of PMs available with different indications.9-11
Single-chamber PMs have one lead, which is connected to the right atrium or right ventricle.9 When the lead is placed in the right atrium, it is indicated for sinus node dysfunction. When placed in the right ventricle, it is indicated for sinus node dysfunction, chronic atrial fibrillation, atrial block, carotid sinus hypersensitivity, or neutrally mediated syncope.10
A dual-chamber PM has two leads with one connected to the right atrium and one to the right ventricle, allowing it to mimic a more natural heart stimulation.9 Dual-chamber PMs are indicated for sinus node dysfunction with abnormal atrioventricular (AV) conduction, carotid sinus hypersensitivity, or neutrally mediated syncope.10
Triple-chamber PMs have three leads with the first lead connected to the right atrium, the second lead to the right ventricle, and the third lead to the left ventricle. Biventricular PMs and CRT are other terms for triple-chamber PMs.9 The lead placement allows these devices to coordinate the heart muscles to contract at the same time, leading to improved efficiency of the heart and its pumping capabilities, which is especially beneficial for patients with heart failure.10,12
Implantable Cardioverter Defibrillators
ICDs deliver electrical currents to the heart, stopping action potentials responsible for abnormal rhythms.13 ICDs can cardiovert, defibrillate, and pace the heart, and are used in patients at high risk of sudden cardiac death due to ventricular arrhythmias. Older ICDs delivered monophasic shocks, while newer devices deliver biphasic shocks. A monophasic shock is sent from one electrode to another. Biphasic shocks start like monophasic shocks, but the direction of the current is reversed by changing the polarity of the electrodes. Biphasic shocks are more effective and use less energy than monophasic shocks.14
Cardiac Resynchronization Therapy
Current guidelines recommend CRT in patients with left ventricular ejection fraction ≤35%, sinus rhythm, QRS ≥150 ms, and New York Heart Association Class II to IV (ambulatory) on appropriate heart failure medications.12 A prospective cohort heart failure study comparing treatment with medication only versus medication plus CRT in patients ≥75 years of age and an ejection fraction ≤35%, with a mean follow-up of about 2 years, resulted in mortality rates of 46% and 21%, respectively.15 Another heart failure study comparing CRT to right ventricular (RV) pacing found that CRT improved left ventricle end-diastolic volume, end-systolic volume, NT-proBNP (N-terminal pro-brain natriuretic peptide), left ventricular ejection fraction, oxygen consumption, and quality of life significantly more than RV pacing.16 Drug therapy for heart failure should be optimized to get maximum results from CRT.12
Device-Medication Interactions
Up to 20% to 40% of patients will experience inappropriate shocks from their ICD.17 These shocks can severely reduce quality of life. Patients describe shocks as being “kicked by a mule,” and they can lead to increased anxiety.17 The first step in preventing inappropriate shocks is to treat the underlying cause. Medications, metabolic abnormalities, and ischemic events are underlying causes of shocks. Drugs that increase the defibrillation threshold can cause defibrillation to fail, and a higher voltage shock would be delivered from the device for the second shock. Medications that lower the threshold allow the electrical current from the device to continue to affect the heart and potentially create new arrhythmias.18
Antiarrhythmics, which decrease excitation of the heart, can alter the effectiveness of implantable defibrillators by affecting the threshold for the device to successfully defibrillate the heart. The threshold is the lowest amount of energy required to successfully defibrillate the heart and restore normal sinus rhythm. Antiarrhythmics can increase or decrease the threshold (TABLE 1).14,17,19
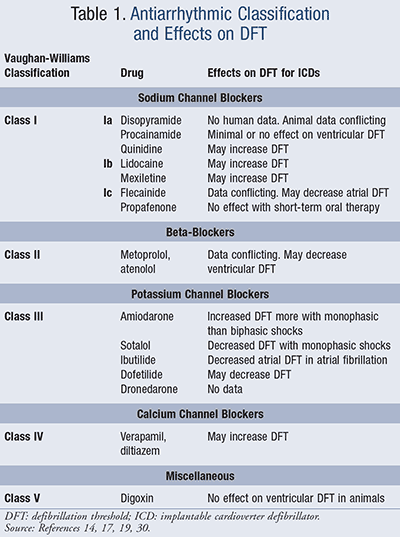
Several agents used in the treatment of heart failure can induce inappropriate shocks. Beta-blockers can decrease the threshold. When beta-blockers are paired with amiodarone, the threshold can slightly increase. ACE inhibitors, angiotensin receptor blockers, and aldosterone antagonists can cause hyperkalemia, which can lead to T-wave oversensing and a false tachycardia reading by the ICD, resulting in a shock.14
Calcium channel blockers (CCBs) work on the voltage sensitive channels and affect the cytosolic calcium, which is present after a defibrillating shock and remains elevated during the postshock phase. The length of this phase and level of cytosolic calcium are indications for unsuccessful defibrillation and the cause of refibrillation. CCBs increase the time the heart spends in the postshock phase, but decrease the cytosolic calcium. These effects are thought to be caused by the blocking of calcium, leading to a decrease in calcium-activated potassium channels.13 In limited studies, verapamil increased the defibrillation threshold, but the clinical significance is unclear.14
Prevention of Inappropriate Shocks
Antiarrhythmics can be used to prevent inappropriate shocks from ICDs. In underlying atrial fibrillation, beta-blockers or CCBs can prevent inappropriate shocks. Amiodarone is an alternative. Sotalol is the preferred agent in patients with underlying ventricular tachycardia or fibrillation. Amiodarone plus a beta-blocker is an alternative therapy. Ranolazine and azimilide decrease premature shocks in ventricular arrhythmias, but azimilide is not available in the United States.14,18,20
Anxiety and depression are found in 84% of patients with implantable cardiac devices, and over one-third of these patients meet diagnostic criteria for anxiety disorders.17,18 Anxiety and depression are predictors of arrhythmic events and long-term regression of cardiac function, which lead to inappropriate and appropriate shocks. Treating anxiety and depression appropriately may prevent this cause of inappropriate shocks.17,18
For end-of-life patients, deactivation of ICDs and pacemakers is recommended, as most receive multiple shocks in their remaining time. This decreases the quality of life not only for the patient but also for the family.18
Prevention and Treatment of Infections
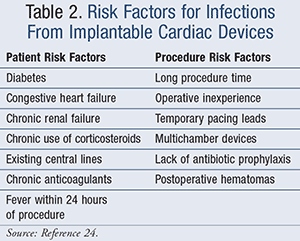
The exact prevalence of infections at the site of cardiac device implantation is unknown.21 Patients undergoing procedures to replace or upgrade devices are at a higher risk for infection (1%-7%) compared to patients receiving their first device (0.5%).22 A population-based cohort study found 1-, 3-, and 6-month mortality rates to be 1.4%, 3.2%, and 5.5%, respectively, after development of a local device infection.23 Patient and procedure factors can influence a patient’s risk for developing an infection (TABLE 2).24
Approximately 90% of infections involving implantable cardiac devices include a single organism, with the most common being Staphylococcus species (80%). Half of these are methicillin-resistant.21,22 Other potential organisms include Corynebacterium species, Propionibacterium acnes, enterococci, gram-negative bacilli, fungi, anaerobes, Candida species, and mycobacteria.22,24
Local and systemic infections should be treated with empirical antibiotics and device removal. IV vancomycin is the preferred empirical therapy, with daptomycin being second-line. One study found a 71% reinfection rate and an increased rate of endocarditis and death in patients without device removal.21 Antimicrobial therapy should last 10 to 14 days for an infection of the pocket, 14 days for bacteremia, and 4 to 6 weeks for endocarditis. Oral antibiotics for 7 to 10 days can be used for superficial or incisional infections, and the device would not need to be removed. Long-term antibiotics could be considered in patients with device infections who are not candidates for complete device removal.21,24
A randomized, double-blind study found prophylactic cefazolin initiated immediately before the procedure and continued for 5 days to be effective at decreasing the incidence of device-related infections.25 For penicillin-allergic patients, vancomycin, daptomycin, and linezolid are alternatives. Antimicrobial agents can be imbedded into the implantation area along with the device, allowing them to be released locally around the device, but more research is needed in this approach.21 Guidelines do not recommend antimicrobial prophylaxis for dental procedures to prevent implantable cardiac device–related infections.24
Anticoagulation
Recent anticoagulation guidelines from the Anti-coagulation Forum recommend that warfarin be held for 5 days prior to device implantation. They recommend to consider bridging with a low-molecular-weight heparin (LMWH) if the patient is at a high thromboembolic (TE) risk and to restart anticoagulants 2 days after the procedure if hemostasis has been achieved.26 The recommendations for the new oral anticoagulants (e.g., apixaban, rivaroxaban) for cardiac device implantation depend on patient renal function.27
A retrospective cohort study for cardiac device implantation compared a new protocol that bridged with heparin in patients with low TE risk and continued warfarin in patients with high TE risk.28 Researchers examined rates of thromboembolism, pocket hematoma, cost, and hospitalization with patients from a retrospective cohort treated according to previous guidelines where high TE–risk patients were treated with pre- and postoperative LMWH and low TE–risk patients were treated with postoperative low-dose LMWH. The results showed that the new protocol resulted in significantly fewer bleeding complications, less hospitalizations, and lower costs, along with adequate protection against thromboembolism compared to the control group.28
A randomized, controlled trial of patients with an annual TE risk of ≥5% compared continued warfarin to bridging with heparin for cardiac device implantation. The trial was stopped early due to a significant risk of device-pocket hematoma in the heparin group (16%) compared to the warfarin group (3.5%).29
Conclusion
The use of implantable cardiac devices continues to increase and improve patients’ quality of life. Pharmacists should be aware of the different types of devices and their association with medications. Antiarrhythmics can cause problems with devices, but can also be used to control inappropriate effects. Infection is a serious device risk, and proper treatment and prophylactic antibiotics should be used. Antibiotic prophylaxis is not necessary for dental procedures in patients with implanted cardiac devices. Appropriate management of oral anticoagulants around cardiac device implantation depends on factors such as a patient’s TE risk, the procedure’s bleeding risk, and a patient’s renal function.
REFERENCES
1. Biventricular pacemaker. Cleveland Clinic. http://my.clevelandclinic.org/services/heart/services/arrhythmia-treatment/biventricular-pacemaker. Accessed October 1, 2015.
2. Implantable cardioverter defibrillator (ICD). Cleveland Clinic. http://my.clevelandclinic.org/services/heart/services/arrhythmia-treatment/implantable-cardioverter-defibrillator-ICD. Accessed October 1, 2015.
3. Permanent pacemaker. Cleveland Clinic. http://my.clevelandclinic.org/services/heart/services/arrhythmia-treatment/permanent-pacemaker. Accessed October 1, 2015.
4. Implantable cardioverter-defibrillators (ICDs). Results. Mayo Clinic. www.mayoclinic.org/tests-procedures/implantable-cardioverter-defibrillator/basics/results/prc-20015079. Accessed October 5, 2015.
5. Pacemaker. Results. Mayo Clinic. www.mayoclinic.org/tests-procedures/pacemaker/basics/results/prc-20014279. Accessed October 5, 2015.
6. Zhan C, Baine WB, Sedrakyan A, Steiner C. Cardiac device implantation in the United States from 1997 through 2004: a population-based analysis. J Gen Intern Med. 2007;23(1):13-19.
7. Long G, Mortimer R, Sanzenbacher G. Recent average price trends for implantable medical devices, 2007-2011. AvaMed. September 21, 2013. http://advamed.org/res/365/recent-average-price-trends-for-implantable-medical-devices-2007-2011. Accessed October 10, 2015.
8. Prevention and management of device infection. Mayo Clinic. www.mayoclinic.org/medical-professionals/clinical-updates/cardiovascular/prevention-management-device-infection. Accessed October 13, 2015.
9. Olshansky B, Hayes DL. Patient information: pacemakers (beyond the basics). UptoDate. Waltham, MA: UptoDate; 2015. www.uptodate.com/contents/pacemakers-beyond-the-basics. Accessed October 12, 2015.
10. Epstein AE, DiMarco JP, Ellenbogen KA, et al. 2012 ACCF/AHA/HRS focused update incorporated in the ACCF/AHA/HRS 2008 guidelines for device-based therapy of cardiac rhythm abnormalities. J Am Coll Cardiol. 2013;61(3):e6-e75.
11. Samii SM. Indications for pacemakers, implantable cardioverter-defibrillator, and cardiac resynchronization devices. Med Clin North Am. 2015;99(4):795-804.
12. Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA guideline for the management of heart failure. Circulation. 2013;128:e240-e327.
13. Krauthamer V. The pharmacology of electrical stimulation in the heart: where devices meet drugs. Drug Discov Today Technol. 2007;4(2):63-67.
14. Dopp AL, Miller JM, Tisdale JE. Effect of drugs on defibrillation capacity. Drugs. 2008;68(5):607-630.
15. de la Cruz E, Cortés M, Farré J, et al. Comparison of pharmacological treatment alone versus treatment combined with cardiac resynchronization therapy in patients over 75 years. J Interv Card Electrophysiol. 2015;43(1):13-20.
16. Kindermann M, Hennen B, Jung J, et al. Biventricular versus conventional right ventricular stimulation for patients with standard pacing indication and left ventricular dysfunction. J Am Col Cardiol. 2006;47(10):1927-1937.
17. Singh S, Murawski MM. Implantable cardioverter defibrillator therapy and the need for concomitant antiarrhythmic drugs. J Cardiovasc Pharmacol Ther. 2007;12(3):175-180.
18. Lampert R. Managing with pacemakers and implantable cardioverter defibrillators. Circulation. 2013;128(14):1576-1585.
19. Kanjwal K, Mainigi SK. Defibrillation threshold testing (DFT): a primer. J Innov Cardiac Rhythm Manage. 2012;3(2012):5-15.
20. Connolly SJ. Comparison of beta-blockers, amiodarone plus beta-blockers, or sotalol for prevention of shocks from implantable cardioverter defibrillators. JAMA. 2006;295(2):165-171.
21. Nof E, Epstein LM. Complications of cardiac implants: handling device infections. Eur Heart J. 2013;34(3):229-236.
22. Tarakji KG, Wilkoff BL. Management of cardiac implantable electronic device infections: the challenges of understanding the scope of the problem and its associated mortality. Expert Rev Cardiovasc Ther. 2013;11(5):607-616.
23. Kirkfeldt RE, Johansen JB, Nohr EA, et al. Complications after cardiac implantable electronic device implantations: an analysis of a complete, nationwide cohort in Denmark. Eur Heart J. 2013;35(18):1186-1194.
24. Baddour LM, Epstein AE, Erickson CC, et al. Update on cardiovascular implantable electronic device infections and their management. Circulation. 2010;121:458-477.
25. De Oliveira JC, Martinelli M, Nishioka SA, et al. Efficacy of antibiotic prophylaxis before the implantation of pacemakers and cardioverter-defibrillators: results of a large, prospective, randomized, double-blinded, placebo-controlled trial. Circ Arrhythmia Electrophysiol. 2009;2(1):29-34.
26. Witt DM, Clark NP, Kaatz S, et al. Guidance for the practical management of warfarin therapy in the treatment of venous thromboembolism. J Thromb Thrombolysis. 2016;41:187-205.
27. Burnett AE, Mahan CE, Vazquez SR, et al. Guidance for the practical management of the direct oral anticoagulants (DOACs) in VTE treatment. J Thromb Thrombolysis. 2016;41:206-232.
28. Cano O, Munoz B, Tejada D, et al. Evaluation of a new standardized protocol for the perioperative management of chronically anticoagulated patients receiving implantable cardiac arrhythmia devices. Heart Rhythm. 2012;9(3):361-367.
29. Birnie DH, Healey JS, Wells GA. Pacemaker or defibrillator surgery without interruption of anticoagulation. N Engl J Med. 2013;368: 2084-2093.
30. Antiarrhythmic agents. Facts & Comparisons [online database]. http://online.factsandcomparisons.com. St. Louis, MO: Wolters Kluwer Health, Inc; 2015. Accessed October 30, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





