US Pharm. 2012;37(4):HS-4-HS-7.
Melanoma, the fifth most common cancer in the United States, accounts for fewer than 5% of skin cancer cases but is the most serious form of the disease, causing up to 75% of skin cancer–related deaths.1-3 The incidence of cutaneous melanoma increased tremendously from 1970 to the late 1990s; however, rates have remained relatively stable since 2000.3,4 Five-year survival rates for melanoma increased from 82% in 1975 to 92% in 2004, but the overall mortality rate remains unchanged.3 Although melanoma is curable if detected in its early localized form, metastatic melanoma continues to be a therapeutic challenge.4
A melanoma is a malignant tumor that arises from melanocytes, dendritic cells that produce melanin, a pigment that protects the body from damaging ultraviolet (UV) radiation. Melanocytes use tyrosine to synthesize melanin. A cluster of melanocytes form nevi (pigmented lesions or moles), and melanoma results when these melanocytes undergo a malignant transformation.3,4 Melanocytes may be found in various areas of the body; however, they are primarily located in the epidermis, and more than 90% of all melanomas are cutaneous.
Incidence Rates and Epidemiology
The incidence of melanoma varies worldwide, with the highest rates in Northern Europe, New Zealand, Australia, and North America.3 Melanoma has one of the fastest-growing incidence rates in the U.S. The U.S. incidence rate increased from 7.9 to 17.7 per 100,000 persons between 1975 and 2000.4 The American Cancer Society (ACS) estimates that, in 2012, approximately 76,250 new melanoma cases (44,250 men, 32,00 women) will be diagnosed and about 9,180 individuals (6,060 men, 3,120 women) will die from the disease.2 Unfortunately, these rates may be underestimated because superficial and in situ melanomas are often managed in outpatient settings, which do not routinely report cases to the cancer registries.4
Unlike many other cancers, melanoma affects people of all ages. Incidence increases with age, and people in their 80s have the highest rate of occurrence; however, melanoma is one of the most common cancers in young adults and can occur in those under 30 years of age.2 Melanoma is rare in children and adolescents; only about 2% of cases are diagnosed in patients younger than 20 years.3 Overall, the median age of diagnosis is 53 years.4 Men are affected slightly more than women, with a ratio of 1.3 to 1, but melanoma remains the leading cause of cancer in women aged 20 to 29 years.1,4 Although mortality rates have declined in younger populations, men aged older than 65 years continue to have the highest mortality rate.3 Melanoma mortality rates vary from state to state, with the highest rates seen in the northwestern U.S. (FIGURE 1).5
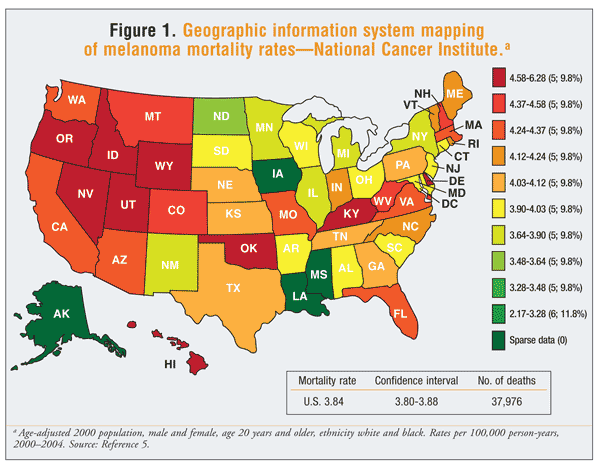
Melanoma is predominantly a malignancy of light-skinned, fair-complexioned people. White populations are most often affected, accounting for 98% of cases.1,4 People of darker complexion, such as East Asian and Indian individuals, develop melanoma at a rate 10 to 20 times lower than that for white individuals.1 In the U.S., approximately 1 in 50 (2%) white, 1 in 200 (0.5%) Hispanic, and 1 in 1,000 (0.1%) African American individuals are at risk for developing melanoma in their lifetime.2
Etiology and Risk Factors
The etiology of melanoma is not fully understood, but many intrinsic and extrinsic factors have been identified that contribute to the occurrence of cutaneous melanoma. Physical characteristics such as blue or green eyes, red or blond hair, pale complexion, high degree of freckling, and tendency to sunburn put one at greater risk for developing melanoma. The number of melanocytic nevi on the body and the presence of atypical melanocytic nevi increase the risk of melanoma.3
Family history and personal history of melanoma are two important factors for diagnosis. If a patient has at least one family member who was diagnosed with melanoma, the risk of diagnosis increases 2.2-fold. The risk is 100% if the patient has two or more family members with dysplastic nevi and/or melanoma.6 Immunosuppression is also a risk factor for melanoma. Maternal–fetal transfer of melanoma is rare but should be noted, as melanoma is the cancer most likely to metastasize from the placenta to the fetus. Congenital melanocytic nevi are also considered precursors for melanoma. External risk factors include geographic location and increased exposure to sunlight and UV radiation.3
Several gene mutations that relate to melanoma risk have been identified. Cyclin-dependent kinase inhibitor 2A and melanocortin-1 receptor gene have been associated with an increased risk of hereditary melanoma.1 Familial atypical multiple mole melanoma syndrome (also known as dysplastic nevus syndrome), a hereditary disease that increases the risk of melanoma 400- to 1,000-fold, is characterized by a predisposition to develop dysplastic nevi and cutaneous melanoma.3 The mitogen-activated protein kinase pathway is a major signaling pathway that has been associated with the development of melanoma. A high prevalence of BRAF gene mutations appears to be an epidemiologic link between UV radiation and melanoma. Alterations in melanoma-specific pathways such as NEDD9, MITF, and NRAS also play a role in the development of melanoma. Studies continue to explore the relationship between molecular genetics and melanoma risk in the hope of better understanding the disease and identifying new targets for pharmacologic therapy.3
Pathophysiology
The developmental pathway of melanoma involves a series of distinct steps; however, not all steps must occur in order for melanoma to develop. Morphologic stages include melanocytic atypia, atypical melanocytic hyperplasia, radial growth phase, primary melanoma in the vertical growth phase with or without in-transit metastases, regional lymph node metastatic melanoma, and distant metastatic melanoma.3
The main goal is to diagnose melanoma early to prevent tumor invasion and metastasis. The classic clinical presentation of melanoma varies by type. Early detection may be facilitated by using the ABCDE mnemonic to assess a mole: Asymmetry; Border irregularity; Color variation or recent color change; Diameter (increasing or >6 mm); Evolving lesion, including surface changes (i.e., any change in shape, size, color, or elevation) and the development of new symptoms (e.g., bleeding, itching). Several characteristics are used to help distinguish benign nevi from atypical nevi, including size, shape, color, location, and number. If any abnormality is suspected, a dermatologist should perform a comprehensive skin examination, and a biopsy may be warranted.3 This step is critical for evaluating a patient with multiple or atypical nevi, a history of excessive sun exposure, or melanoma.4
Classification and Staging
Melanoma is classified into four major types: superficial spreading melanoma, lentigo maligna melanoma, acral lentiginous melanoma, and nodular melanoma. The types differ in appearance, site, and population affected. Superficial spreading, lentigo maligna, and acral lentiginous melanomas have a period of superficial growth and, if identified early, may be cured by surgical excision. However, nodular melanoma usually presents as a deeply invasive lesion that is highly capable of early metastasis and is difficult to cure.3
Melanoma staging is important to help determine prognosis and treatment options. The American Joint Committee on Cancer TNM staging system for melanoma contains three major components that define the extent of disease: Tumor size, lymph Node involvement, and Metastasis. A stage 0 melanoma is defined as a melanoma in situ that has not yet spread from the epidermis to the dermis. Stages I and II are localized to a specific area. Stage III encompasses lymph nodes, but there is no distant spread. Stage IV indicates metastasis to distant sites. Tumor thickness, level of tumor invasion, and ulceration are powerful predictors of survival, and TNM status and stage are used to determine prognosis and survival. Other predictors include serum lactate dehydrogenase, presence and site of distant metastases, evidence of nodular involvement, and regression of primary melanoma.1-4
Prevention
Melanoma has become a worldwide public health concern, and awareness is the key factor in early detection and prevention of the disease. ACS recommendations for prevention include limiting direct sun exposure between the hours of 10 AM and 4 PM, when UV rays are the most intense; wearing protective clothes that cover the arms, legs, and torso, sunglasses that protect the eyes, and a broad-brimmed hat that shields the face; avoiding tanning beds and sunlamps; and using sunscreen and lip balm on sun-exposed areas.7-9
The American Academy of Dermatology recommends using sunscreen with a sun protection factor of 30 or more. Sunscreen products labeled as broad spectrum shield against both UVA and UVB rays and are a good source of protection. One ounce (about a palmful) of sunscreen should be applied to the arms, legs, neck, and face every 2 hours, or sooner after sweating or swimming. Sunscreen should also be applied on cloudy days because UV rays are still present.3,7-9
Treatment
The treatment of melanoma depends upon the stage of disease. The stage of cutaneous melanoma is directly associated with the development of metastatic disease. If the melanoma is diagnosed at an earlier stage, surgical excision is the treatment of choice. For melanomas that have lymphatic involvement, surgical excision may not be enough, and lymph biopsy may be warranted.3,4
Surgery: The surgical excision margin is the most important determinant of outcome in early-stage melanoma. Thin (<1 mm) melanomas require an incision of less than 1 cm. The cure rate for melanomas smaller than 1 mm is as high as 98%.10 Midsize melanomas (1-2 mm) usually require an incision of 2 cm. Larger melanomas (>4 mm) may have lymphatic involvement and are associated with a high rate of recurrence. These tumors also require a maximum surgical incision margin of 2 cm.
Surgery is usually limited to patients with early-stage disease. Patients with stage III melanoma usually have lymph node involvement or in-transit metastasis. In-transit metastasis is when a tumor develops in the lymph vessel between the primary melanoma and the regional lymph node basin.11 In-transit metastases occur more than 2 cm from the original site and are more common in individuals with thick, ulcerated lesions. Surgery is also used to manage these distinct melanoma-associated lesions.
Immunotherapy: Melanoma is one of the most immunogenic solid tumors. Immunotherapy appears to be a viable treatment option, as more traditional cancer treatments do not always yield positive results in cases of melanoma. Complete response rates to immunotherapy vary in patients with melanoma, but generally they are low.
Interferon (IFN) alfa is approved for adjuvant therapy in metastatic melanoma. For patients who have undergone surgical resection for lesions larger than 2 mm, with or without regional lymph node metastases, the only effective adjuvant therapy is IFN.12 Various trials have evaluated different doses and schedules of IFN therapy for metastatic melanoma. The optimal IFN treatment modality has not been established for the management of metastatic melanoma. Some of the disadvantages of IFN therapy are high cost, unclear long-term benefit, and toxicities (e.g., liver toxicity). IFN is known to cause constitutional symptoms manifesting as fever, chills, myalgia, and fatigue. These symptoms have been treated with acetaminophen, nonsteroidal anti-inflammatory drugs, and meperidine. The most common dosages of IFN alfa therapy are listed in TABLE 1.13,14

Interleukin-2 (IL-2), a glycoprotein produced by activated lymphocytes, may be immunosuppressive. The role of immunosuppressive properties in melanoma is not fully understood. IL-2 does not have a direct effect on the tumor; rather, it works to regulate immunologic activity. Because of severe multiorgan toxicity associated with IL-2 therapy, treatment with higher doses of IL-2 is reserved for patients with good organ function who are being closely monitored by experienced clinicians. The high-dose regimen of recombinant IL-2 (aldesleukin) used to treat metastatic melanoma is 600,000 IU/kg/dose every 8 hours, for a maximum of 14 doses in a 5-day period, given for two cycles, with a 10- to 14-day rest period between cycles.11 Common side effects of IL-2 include hypotension, arrhythmias, severe infections, and shortness of breath; these effects are reversible.
Patients with stage IV malignant melanoma require cytotoxic, single- or double-agent, systemic therapy. Single-agent chemotherapy with dacarbazine, temozolomide, or fotemustine should be used in selected patients who are not candidates for treatment with high-dose IL-2. Poor response to chemotherapy or immunotherapy alone has led to the evolution of combination therapy. Biologic therapy has been combined to increase overall activity and, perhaps, response rates.3
Ipilimumab: Ipilimumab (Yervoy) was approved by the FDA in March 2011 to treat advanced or metastatic melanoma.15 Ipilimumab is a human immunoglobulin G1 monoclonal antibody that exerts its action by blocking cytotoxic T lymphocyte–associated antigen 4, thereby increasing T-cell activation and proliferation. Ipilimumab activates the immune system, causing antitumor action.11 The National Comprehensive Cancer Network recommended ipilimumab as a category 1 treatment option for metastatic melanoma following a phase III trial that showed an overall survival benefit.16 The usual dosage of ipilimumab in metastatic melanoma is 3 mg/kg IV every 3 weeks for four doses.
Summary
Melanoma is one of the few cancers whose incidence is on the rise. Older adults are mainly affected, with a higher incidence later in life. The primary treatment for melanoma is surgery with adjuvant IFN, depending upon the melanoma’s size and grade. Treatment for advanced melanoma is limited, and toxicities may limit its overall benefit. The key focus for the prevention of melanoma is sunscreen application and regular skin examinations for the detection of suspicious moles.
REFERENCES
1. Urba WJ, Washington CV, Nadiminti H. Cancer of the skin. In: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York, NY: McGraw-Hill Medical; 2012.
2. American Cancer Society. What are the key statistics about
melanoma?
www.cancer.org/Cancer/SkinCancer-Melanoma/DetailedGuide/melanoma-skin-cancer-key-statistics.
Accessed February 21, 2012.
3. O’Bryant CL, Poust JC. Melanoma. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th ed. New York: McGraw-Hill Medical; 2011.
4. Kazerooni R, Madden T. Melanoma. In: Schumock GT, Brundage DM, Richardson M, eds. Pharmcotherapy Self-Assessment Program: Hematology/Oncology I and II (Book 10). 5th ed. Kansas City, MO: American College of Clinical Pharmacy; 2006:55-80.
5. National Cancer Institute. Cancer mortality maps. http://ratecalc.cancer.gov. Accessed March 13, 2012.
6. Goldstein AM, Tucker MA. Genetic epidemiology of cutaneous melanoma: a global perspective. Arch Dermatol. 2001;137:1493-1496.
7. American Cancer Society. Melanoma skin cancer.
www.cancer.org/Cancer/SkinCancer-Melanoma/DetailedGuide/melanoma-skin-cancer-prevention.
Accessed February 18, 2012.
8. Robinson JK. Sun exposure, sun protection, and vitamin D. JAMA. 2005;294:1541-1543.
9. American Academy of Dermatology. Be Sun Smart.
www.aad.org/skin-care-and-safety/skin-cancer-prevention/be-sun-smart/be-sun-smart.
Accessed February 18, 2012.
10. Slinluff CL Jr, Flaherty K, Rosenberg SA, Read PW. Cutaneous melanoma. In: DeVita VT Jr, Lawrence TS, Rosenberg SA, eds. DeVita, Hellman, and Rosenberg’s Cancer: Principles & Practice of Oncology. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2008:1897-1951.
11. CDC. United States Cancer Statistics (USCS). 1999–2007 incidence
and mortality data. www.cdc.gov/uscs. Accessed February 18, 2012.
12. Molife R, Hancock BW. Adjuvant therapy of malignant melanoma. Crit Rev Oncol Hematol. 2002;44:81-102.
13. PegIntron (peginterferon alfa-2b) product information. Whitehouse Station, NJ: Schering Corp; December 2011.
14. Intron A (interferon alfa-2b, recombinant) product information. Whitehouse Station, NJ: Schering Corp; September 2011.
15. Eggermont AM, Robert C. New drugs in melanoma: it’s a whole new world. Eur J Cancer. 2011;47:2150-2157.
16. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology. Melanoma. Version 3.2012.
To comment on this article, contact rdavidson@uspharmacist.com.





