US Pharm. 2012;37(9):37-43.
The National Osteoporosis Foundation (NOF) defines adequate amounts of calcium and vitamin D to be at least 1,200 mg and 800 to 1,000 IU per day, respectively, for men and postmenopausal women 50 years and older.1 Men and women who are not receiving adequate amounts of calcium and vitamin D supplementation are at an increased risk of osteoporosis and fractures.1 Despite an established recommended daily amount of adequate supplementation intake, many women do not understand the importance of such supplementation. A meta-analysis by Sunyecz and Weisman concluded that women did not take the recommended amounts of calcium and vitamin D despite advancements in osteoporosis treatments and medication.2
Additional issues with calcium supplementation exist due to the type of calcium salt utilized. Calcium supplementation is available as two salt forms, calcium carbonate and calcium citrate, and the pharmacokinetic profile differs for each. The citrate formulation is best absorbed in a basic environment; thus, it is the preferred salt if the patient is on either an H2-antagonist or a proton pump inhibitor (PPI).
Vitamin D sources exist inside and outside the human body. Vitamin D is a fat-soluble vitamin obtained from a few plant sources and supplementation and is also synthesized in the skin from sun exposure.3 Ergocalciferol (D2) is made from plant sterols and yeast, whereas cholecalciferol (D3) is synthesized in the skin by ultraviolet rays via 7-dehydrocholesterol.4 In both instances, vitamin D is acquired in its inactive form and has to be activated once in the body. The liver converts vitamin D to 25-hydroxyvitamin D (25[OH]D), known as calcidiol. Further activation then occurs in the kidney, where it forms the physiologically active formulation 1,25-dihydroxyvitamin D (1,25-[OH]2D), or calcitrol.3 Without this activation process, any formulation of calcium cannot be absorbed.
Osteoporosis screening and diagnosis are conducted by measuring bone mineral density (BMD). This procedure tests the risk of bone fracture; it can also indicate improvements or deterioration in BMD and may be used in monitoring osteoporosis therapy.1 There are two areas where BMD is measured: central (spine and hip) and peripheral (forearm, heel, fingers) skeletal sites. This test is accurate and highly reproducible when performed according to accepted standards. According to the World Health Organization (WHO) diagnostic classification, peripheral dual-energy x-ray absorptiometry (pDXA) is not appropriate for monitoring BMD after treatment.1 However, recent data suggest that “peripheral bone density testing in the primary care setting can also identify postmenopausal women who have a higher risk for fracture over the short term (1 year).”5 BMD testing may be needed for a minimum of 2 years to measure any change. Repeated screenings can be used in older women, those with lower BMD at baseline, and those with other risk factors for fracture.5
Research
Previous studies have indicated that community pharmacists have the opportunity for, and are beneficial to the community in, screening, educating, and referring women at risk of osteoporosis.6,7 A recent article published in Osteoporosis International reported that interventions by health care professionals improved patients’ quality of life, medication compliance, and calcium intake, but the data were less conclusive of the effects of such intervention on BMD. It concluded that further research is needed to assess the outcomes of nondrug interventions by health care professionals on certain osteoporotic women.8
The following study intends to show the positive impact a pharmacist can make when asking questions and counseling patients on calcium and vitamin D supplementation in the community pharmacy setting.
Objectives
The primary objective of this study was to identify women at risk for osteoporosis or on bisphosphonate therapy who were not taking recommended amounts of calcium and vitamin D supplementation. Secondary objectives included: 1) assessing efficacy of appropriate supplementation by measuring BMD at baseline, 3 months, and 6 months; and 2) participant satisfaction and the value of a community pharmacist’s role in increasing or modifying supplementation intake and thus increasing BMD.
Methods
The Institutional Review Board (IRB) approved this study, and informed consent was obtained from each participant. Participants were identified at two community pharmacies and two senior centers located in multiple regions of Massachusetts. Participants in each community pharmacy were identified using a real-time computer profile search and then approached by the use of an informational flyer explaining the study that was stapled on each prescription for a bisphosphonate or prednisone. Participants were also asked, upon prescription pick-up, if they would like to participate in the research study by the primary investigator (PI) or PharmD candidate. Another method of recruitment was by questioning women during counseling for any prescription or OTC products, in particular those seeking calcium and vitamin D supplementation. The PI also recruited participants during a one-on-one medication review session at two area senior centers. Participants were identified if on bisphosphonate treatment and/or long-term corticosteroid therapy based on a “medication brown bag” event.
Inclusion criteria were based on the following: 1) women on bisphosphonate treatment (i.e., alendronate sodium, risedronate sodium, ibandronate sodium); and/or 2) women on long-term corticosteroid therapy (daily dose >5 mg prednisone or equivalent for >3 months). Women over the age of 18 years with documented history of long-term prednisone therapy were included in the study due to the association of low bone mass and risk of osteoporosis. Participants under 50 years of age were included in the study to potentially benefit from osteoporosis prevention education.
Exclusion criteria were based on the following: 1) women under the age of 18 years; 2) any allergies or contraindications to either supplement; 3) a history of calcium-based kidney stones; and 4) previous or current gastrointestinal problems with taking supplementation and/or bisphosphonates because of the possibility of worsening or recurrence of such issues. Participants were also excluded and referred to their primary care physician if their initial BMD test resulted in a T-score of ≤-2.5 without prior diagnosis of osteoporosis (TABLE 1).

All participants were asked to provide demographic information (telephone number, age, gender), current and past medical history, and medication history, including specific information on calcium and vitamin D intake (TABLE 2). At the initial visit, recruited participants were provided with educational material on osteoporosis and were reviewed by the PI. Participants were then advised of the appropriate amounts and types of calcium and vitamin D based on the participant’s age and medications. This document was used to record the initial BMD result and was used again at 3 months and 6 months to record supplementation intake as well as BMD measurement (TABLE 3). A BMD measurement was taken at peripheral skeletal sites (heel) using the Sahara Clinical Bone Sonometer (Hologic, Inc.).
Last, participants were prompted to assess their overall satisfaction with the service provided through a short survey at 6 months. Participants were asked to use a Likert (five-level) scale in answering 6 questions (FIGURE 1).
Data Analysis
Sample size was determined using the primary outcome involving the percentage of identified participants who were not appropriately using calcium and vitamin D supplementation. Since this percentage was unknown for the different sites, the assumed value of 50% was used. It was believed that this rate could be reduced to 25% as a result of education from the pharmacist-participant interaction. The number of participants required to detect this 25% difference at the 5% level of significance with 80% power was 30 participants.9 A total of 36 participants were to be recruited to account for possible dropouts.
Participant demographics, other health-related variables, and satisfaction survey results were summarized using descriptive statistics (including means and percentages). The McNemar test for categorical variables and paired t-tests for numerical variables were used to examine the effect of education from the pharmacist-participant interaction on appropriate calcium/vitamin D supplementation and BMD results at baseline, 3 months, and 6 months. Results were considered statistically significant if the observed level of significance is P <.05. All analyses were conducted using the statistical analysis computer program Number Cruncher Statistical System (NCSS).9
Results
Participant Eligibility: Of the four study sites, five of the 43 participants (12%) questioned for eligibility into the study were excluded. Reasons for exclusion were that three participants had an initial BMD measurement (T-score) of less than −2.5 without prior diagnosis of osteoporosis; one participant had a history of kidney stones; and one participant had a history of bone spurs (calcium was not recommended per participant’s primary care physician). Of the 38 eligible participants, 37 (97%) were 50 years of age or older and 1 (3%) was <50 years.
Assessment of Initial Supplementation Intake: Seventy-four percent of all eligible participants (28 out of 38) were either not taking any calcium and vitamin D supplementation, were taking inappropriate amounts of supplementation (TABLE 2), or were using a less effective calcium salt due to an acid reducer (i.e., PPI) or salt causing constipation. Thus, a community pharmacist intervention was required for 28 (74%) of the eligible participants.

Assessment of Supplementation Compliance After Pharmacist Intervention: Of the 28 participants who initially required a pharmacist’s intervention, 15 returned for follow-up at 3 months. Nine of these participants (60%) reported compliance with supplementation after the intervention compared to none (0%) at the initial visit, a statistically significant increase (P = .003). At 6 months, 8 participants returned for follow-up. Four of these participants (50%) continued to be compliant with what was originally recommended compared to none (0%) at the initial visit, a statistically significant increase (P = .046).
Assessment of BMD After Pharmacist Intervention: Of the 28 participants who initially required a pharmacist’s intervention and whose BMD was measured at the initial visit, 15 returned for follow-up at 3 months (TABLE 3). Nine of these participants (60%) had an increase or no change in BMD compared to none (0%) at baseline, a statistically significant increase (P = .001). At 6 months, 8 participants returned for follow-up. Five of these (62.5%) had an increase in BMD from the 3-month follow-up, a statistically significant increase (P = .026); and 4 of these 8 participants (50%) had an increase in BMD compared to none (0%) at baseline (P = .077).
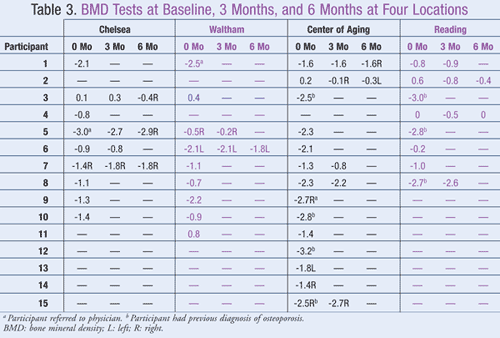
According to the U.S. Preventive Services Task Force, no studies have evaluated the optimal timing for repeated screening; however, BMD testing may be needed for a minimum of 2 years to measure any change.5 This current study indicates that 3 to 6 months may be a sufficient time to show a positive change in BMD; however, results should be interpreted with caution because of study limitations (i.e., small patient population, numerous drop-outs).
Assessment of Satisfaction of Overall Service Provided by the Community Pharmacist: Of the 38 eligible participants, 13 (34%) completed an exit survey (FIGURE 1).
Based on the survey results, most participants answered, “strongly
agree” for the following question: “I found this service beneficial to
me in the management of my osteoporosis.” Most participants answered
“agree” for the following questions: “I learned how to manage my
osteoporosis with this study;”
“I would recommend this service to someone I know;” “I would be
willing to pay for this service in the future;” and “I would be
interested in continuing osteo-porosis counseling, education, and
monitoring by a pharmacist.” In addition, most participants indicated
that they would be willing to pay “less than or equal to $10” for the
service provided.

Discussion
According to the NOF, men and women over the age of 50 years require 1,200 mg of calcium and 800 to 1,000 IU of vitamin D.1 All participants with the exception of one woman in this study fell into this age category. Participants’ intake of supplementation can be seen in TABLE 2. Initial findings of this study indicate that the majority of women were not taking the correct, if any, amount of calcium and vitamin D supplementation despite the proven necessity of such supplementation in reducing the risk of fractures.1 Some women were also unaware of the absorption issues with calcium supplementation. Absorption of calcium, depending on stomach acidity, is between 500 to 600 mg per dose. Of the 38 eligible participants, 9 participants were on an acid-reducer (e.g., PPI or H2-antagonist) at baseline. A number of women studied were taking 1,200 mg of calcium once daily, resulting in an inappropriate total amount absorbed. Women were also under the impression that they were receiving the full benefits of calcium from taking 1,200 mg in a single dose.
Through this study, participants who were not taking the recommended amounts of combination supplementation or who needed to separate their calcium supplementation intake into two doses were identified. Most of these women were not aware of the calcium salt differences and thus were not using the most appropriate formulation. A pharmacist’s intervention proved beneficial in identifying those participants on acid-suppressing medications and in modifying calcium supplementation to an alternate formulation.
A majority of participants (60% in the community pharmacies, 57% in the senior centers) who returned for follow-up at 3 and 6 months showed an increase in compliance to calcium and vitamin D supplementation after a community pharmacist’s intervention. This study supported previous results indicating that pharmacists have the opportunity for, and are beneficial to the community in, screening women at risk of osteoporosis as well as increasing medication compliance.
Study results showed participant compliance in more than half of the study population after a pharmacist intervention. Results also indicate that in about 67% of participants, BMD increase after 3 months may be due to compliance with supplementation. About 50% of participants who returned in 6 months showed an increase in overall BMD from the initial visit. Previous studies reported that more research is needed to correlate compliance of supplementation with the effects on BMD. Even though this study may have shown a correlation between calcium and vitamin supplementation compliance and increased BMD, more studies are warranted.
Limitations
The patient population within this study was relatively small, although a surplus of participants was included to show statistical significance (38 vs. 36). Additional and larger studies may be warranted to show the positive role a community pharmacist’s intervention has on supplementation compliance and increased BMD.
In addition, BMD data may have been skewed. Initial and follow-up BMD measurements were recorded; however, it was not recorded which heel, left or right, was measured by the Sahara Clinical Bone Sonometer. This could possibly have had an effect on reported data due to the fact that BMD in each heel may vary. Results could also have been skewed as a result of other recommendations made by the PI, such as modification of all life-style and diet risk factors associated with an increased risk of osteoporosis. Last, favorable BMD results could also have been caused by increased compliance with bisphosphonate therapy alone.
Compliance with supplementation at 3-month and 6-month follow-up was assessed based on a self-reported response. Participants were asked open-ended questions as to the amount of supplementation being taken. No standard compliance assessment tool was used, which might have been a more appropriate tool in assessing compliance.
Finally, data may have been biased because recruitment methods were not uniform across all settings. The recruitment methods differed, as there was no real-time computer system at each of the senior centers. These settings had to rely on different recruitment methods from the community pharmacy settings, such as one-on-one medication counseling. This may have biased the results because participants may have been more willing to follow-up with the pharmacist, since all members of the senior centers were provided other services outside of the study.
Conclusion
The majority (74%) of participants in the study were either not supplementing with calcium and vitamin D, were taking inappropriate amounts of supplementation, or were using a calcium salt with less absorption caused by other medications the participant was taking (i.e., PPI) initially. A community pharmacist proved beneficial in identifying and recommending appropriate amounts and types of supplementation for those at osteoporosis risk and on bisphosphonate therapy. Intervention by a community pharmacist proved beneficial for compliance with appropriate amounts of calcium and vitamin D supplementation, and appropriate intakes of supplementation showed increased BMD at 3 months and 6 months. Averaging each question on the survey that the participants completed, most participants believe that the service was beneficial and helped to manage their osteoporosis. Most participants would recommend the service to someone they know. Participants would be willing to pay $10 or less for the service in the future. More studies are warranted to demonstrate if OTC supplementation can increase BMD.
The authors would like to acknowledge Professor Gary Tataronis, Statistician, Massachusetts College of Pharmacy and Health Sciences, for his contribution to this study.
REFERENCES
1. National Osteoporosis Foundation. Osteoporosis: Clinician’s Guide to Prevention and Treatment of Osteoporosis. Washington, DC: National Osteoporosis Foundation; 2010.
2. Sunyecz JA, Weisman SM. The role of calcium in osteoporosis therapy. J Womens Health. 2005;14:180-192.
3. Office of Dietary Supplements, National Institutes of
Health. Dietary supplement fact sheet: vitamin D (health professional
version). June 24, 2011.
http://ods.od.nih.gov/factsheets/VitaminD-HealthProfessional/. Accessed
August 6, 2012.
4. Arvold DS, Odean MJ, Dornfeld MP, et al. Correlation of
symptoms with vitamin D deficiency and symptom response to
cholecalciferol treatment: a randomized controlled trial. Endocr Pract. 2009:15:203-212.
5. U.S. Preventive Services Task Force (USPSTF). Screening
for osteoporosis in postmenopausal women: recommendations and
rationale. Ann Intern Med. 2002;137:526-528.
6. Johnson JF, Koenigsfeld C, Hughell L, et al. Bone
health screening, education, and referral project in northwest Iowa:
creating a model for community pharmacies. J Am Pharm Assoc. 2008;48:379-387.
7. MacLaughlin EJ, MacLaughlin AA, Snella KA, et al.
Osteoporosis screening and education in community pharmacies using a
team approach. Pharmacotherapy. 2005;25:379-386.
8. Lai P, Chua SS, Chan SP. A systematic review of
interventions by healthcare professionals on community-dwelling
postmenopausal women with osteoporosis. Osteoporosis Int. 2010;21:1637-1636.
9. Hintze J. PASS: Power Analysis and Sample Size. Kaysville, UT: NCSS, LLC; 2008.
To comment on this article, contact rdavidson@uspharmacist.com.






