US Pharm. 2021;46(10):HS-11-HS-15.
ABSTRACT: Nonalcoholic fatty liver disease (NAFLD) is a condition that affects nearly a quarter of the U.S. population. Nonalcoholic steatohepatitis, the more severe form of NAFLD, is associated with an increased risk for cirrhosis and poor health outcomes. NAFLD is a common comorbidity in patients with type 2 diabetes and/or obesity, but it often remains undiagnosed. There are currently no FDA-approved medications for the treatment of NAFLD. Weight loss is the cornerstone of management through lifestyle interventions and the use of pharmacologic and/or surgical treatments to promote weight loss in appropriate patients. Research with select glucose-lowering medications is ongoing.
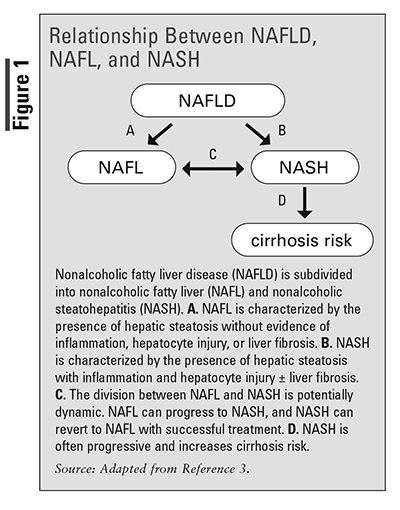
Nonalcoholic fatty liver disease (NAFLD), characterized by excess fat accumulation in the liver, is estimated to occur in approximately 24% of adults in the U.S., making NAFLD one of the most common causes of liver disease in the country.1,2 NAFLD is defined by the presence of hepatic steatosis (fatty liver) in the absence of heavy alcohol use and thus differs from alcohol-associated liver disease.2 NAFLD is an umbrella term encompassing two forms of fatty liver disease: nonalcoholic fatty liver (NAFL) and nonalcoholic steatohepatitis (NASH).3 NAFL is consistent with the presence of excess fat in the liver but with little to no inflammation or liver cell damage present.2 NASH, in contrast, is considered when both excess fat and inflammation/hepatocyte damage are present in the liver.2 In both cases, no significant alcohol consumption should exist, defined as >21 drinks/week for men and >14 drinks/week for women.4 FIGURE 1 further describes the relationship between NAFLD, NAFL, and NASH.3
NAFLD is a common comorbidity in patients with type 2 diabetes mellitus (T2D) and/or components of the metabolic syndrome, and it is associated with notable morbidity and mortality. Recent estimates suggest that over 18 million people in the U.S. are living with T2D and NAFLD, with over 6 million having NASH.5 It is predicted that over the next 20 years, patients with comorbid NASH and T2D will require 65,000 liver transplants and will experience 1.37 million cardiovascular-related deaths and 812,000 liver-related deaths.5 Given the importance of NAFLD as a comorbidity in patients with T2D, the American Diabetes Association (ADA) began offering recommendations related to NAFLD screening in 2019.6 In the most recent 2021 ADA Standards of Medical Care in Diabetes, the ADA recommends that patients with T2D or prediabetes with elevated liver enzymes (e.g., elevated alanine aminotransferase [ALT]) or evidence of fatty liver on ultrasound be further evaluated for the presence of NASH and liver fibrosis.7
Pathophysiology and Associated Comorbidities
While the pathogenesis of NAFLD is not entirely understood, insulin resistance is widely considered a key mechanism contributing to the development and progression of hepatic steatosis.8 Insulin resistance, impaired lipid and glucose metabolism, and altered insulin secretion all play a role in both NAFLD and T2D progression and may be indicators as to why the two disease states are so intimately linked.9 Similar genetic predispositions and environmental factors, such as obesity, may also play a role in the progression of both NAFLD and T2D.9 In patients with T2D and NAFLD, hepatic glucose production is inadequately suppressed due to hepatic insulin resistance.10 This causes a variety of metabolic alterations that increase plasma triglyceride (TG) and free fatty acid (FFA) levels, which further impairs insulin signaling.9 Increased FFA levels can additionally overload the fat-metabolism capacity of the liver, leading to activation of inflammatory pathways and deposition of fat in nonadipose tissues (often referred to as ectopic fat deposition).11 Chronic hyperglycemia also contributes to oxidative stress and accumulation of reactive oxygen species capable of damaging a myriad of tissues, including the liver.9 Overall, insulin resistance, lipid accumulation, and excess inflammation are all believed to contribute to the development and progression of NAFLD.
As discussed above, NAFLD and T2D share common pathophysiologic pathways. Not surprisingly, NAFLD has been linked with several additional comorbidities associated with the metabolic syndrome such as obesity, chronic kidney disease (CKD), and cardiovascular disease (CVD).3 While these comorbidities are common, they are not necessarily present in all cases of NAFLD (e.g., NAFLD can occur in nonobese patients). Of note, NAFLD is likely an independent risk factor for the development of heart disease.3
Diagnosis
While serum liver function tests (such as aminotransferase and ferritin) are often abnormal in patients with NAFLD, abnormal laboratory tests are not required or sufficient to make a diagnosis since laboratory tests can be normal in some cases of NAFLD.12 Most patients with NAFLD also do not present with specific symptoms.13 NAFLD is therefore a diagnosis of exclusion. Recommendations from the American Association for the Study of Liver Diseases (AASLD) state that the diagnosis of NAFLD requires each of the following criteria be met: demonstration of hepatic steatosis by imaging or biopsy; exclusion of significant alcohol consumption; exclusion of other causes of hepatic steatosis; and absence of coexisting chronic liver disease.12
As noted above, excluding other causes of hepatic steatosis is important in making an accurate diagnosis of NAFLD. TABLE 1 provides a list of alternative causes of hepatic steatosis.12

Treatment of NAFLD
Per guidance from AASLD, the management of NAFLD should consist of treating liver disease as well as managing associated metabolic comorbidities, such as obesity, insulin resistance, T2D, and hyperlipidemia.12 Importantly, patients with NAFLD are not at higher risk for statin-associated liver injury in the absence of decompensated cirrhosis, and statins should therefore be used as indicated to mitigate CVD risk. To avoid worsening of hepatic steatosis, patients with NAFLD should also be advised against excessive alcohol consumption.12 Achieving weight loss through lifestyle modifications, pharmacologic interventions, and/or surgical approaches can be employed to improve hepatic steatosis (see section on Lifestyle Interventions below). While not a focus of this review, all stages of NAFLD are more common in patients meeting candidacy criteria for bariatric surgery.12
There are currently no randomized, controlled trials that specifically evaluate bariatric surgery as an intervention to treat NAFLD, although there are a few published studies comparing liver histology before and after bariatric surgery.12 One study enrolled 381 severely obese adult patients and examined liver histology 1 and 5 years following bariatric surgery.14 Of the 381 participants, 99 had probable or definite NASH prior to surgery. Patient follow up showed significant improvement of steatosis and resolution of probable or definite NASH at 1 and 5 years postsurgery.14 Given the importance of weight loss as a strategy to reduce hepatic steatosis, these findings may not be surprising, yet the AASLD states it is premature to consider bariatric procedures as an established approach to treat NASH. The AASLD does state, however, that bariatric surgery can be considered in otherwise eligible obese individuals with NAFLD.12
Lifestyle Interventions
Lifestyle interventions involving dietary modification, increased physical activity, and weight loss are recommended in patients with NAFLD.12 Weight loss of ≥5% of total body weight can improve hepatic steatosis, with weight reductions of ≥7% demonstrating improvements in histological disease activity.15 AASLD offers the following guidance related to lifestyle interventions in patients with NAFLD12:
Weight loss generally reduces hepatic steatosis, achieved either by hypocaloric diet alone or in conjunction with increased physical activity. A combination of hypocaloric diet (daily reduction by 500-1,000 kcal) and moderate-intensity exercise is likely to provide the best likelihood of sustaining weight loss over time.
Weight loss of at least 3% to 5% of body weight appears necessary to improve steatosis, but a greater weight loss (7%-10%) is needed to improve the majority of the histopathological features of NASH, including fibrosis.
Exercise alone in adults with NAFLD may prevent or reduce hepatic steatosis, but its ability to improve other aspects of liver histology remains unknown.
Glucose-Lowering Medications
Although there are currently no approved medications for the treatment of NAFLD, there are several agents under study that target a variety of mechanisms and pathways to address insulin resistance, hepatic steatosis, and fibrosis.12 Notably, several medication classes approved for the treatment of T2D that improve insulin sensitivity and/or reduce total body mass have shown early promise in patients with NAFLD. Of currently available glucose-lowering medication classes, agents from the thiazolidinedione, glucagon-like peptide-1 (GLP-1) receptor agonist, and sodium-glucose cotransporter-2 (SGLT2) inhibitor classes have shown the most promise. A summary of evidence to date with agents from these medication classes is provided below.
Thiazolidinediones (TZDs): The ADA recommends TZDs as a second-line option for add-on to metformin in patients with T2D who are not meeting individualized glycemic targets, especially when cost is a major consideration or when there is a compelling need to minimize hypoglycemia.7 TZDs improve insulin sensitivity via activation of peroxisome proliferator-activated receptor (PPAR)-gamma nuclear receptors. Activation of PPAR-gamma has broad effects on lipid and glucose metabolism as well as inflammatory processes.16 Given these effects, TZDs have been explored as potential therapies in patients with NASH. One such study enrolled 55 patients with NASH and either T2D or prediabetes where participants were treated with a hypocaloric diet plus 45 mg of pioglitazone daily or a hypocaloric diet plus placebo for 6 months.17 The pioglitazone group, compared with the placebo group, experienced significant improvement in hepatic steatosis (P = .003) and markers of inflammation (P = .008).
In another study, participants with T2D or prediabetes were likewise treated with pioglitazone or placebo for a duration of 18 months, immediately followed by an additional 18-month open-label treatment phase with pioglitazone to assess longer-term safety.18 In this trial, 51% of patients treated with pioglitazone experienced resolution of NASH (P <.001), with additional improvements in fibrosis noted. Metabolic and histologic improvements were reportedly continued to improve over the 36-month duration of the trial, although pioglitazone treatment was associated with weight gain when compared with placebo.18
More recently, a study was conducted to determine if patients diagnosed with NASH and T2D achieve a more significant histologic response to pioglitazone compared with patients with NASH and prediabetes.19 Resolution of NASH was seen in 44% of T2D patients compared with 26% of patients with prediabetes. Study participants with T2D also experienced greater improvements in liver fibrosis and adipocyte insulin sensitivity when compared with participants with prediabetes.19 These results suggest that patients with T2D may realize a more pronounced benefit to pioglitazone when compared to patients with NASH who do not have diabetes and thus may be good candidates for TZD treatment.
`The other currently available TZD agent, rosiglitazone, has also shown encouraging results in multiple trials, but it is not commonly used in clinical practice. Although data to date with TZDs are promising, it is important to take into consideration potential adverse events associated with the medication class. Weight gain, fluid retention, and exacerbation of heart failure are all potential safety concerns that must be considered prior to TZD use.7
GLP-1 Receptor Agonists: GLP-1 receptor agonists are a growing class of glucose-lowering medications that offer a variety of benefits in the setting of T2D treatment. Notably, select agents from the GLP-1 receptor agonist class have been associated with CVD risk reduction and robust weight loss—benefits that have fueled particular interest in these agents for the treatment of NAFLD. The Liraglutide Safety and Efficacy in Patients with Non-alcoholic Steatohepatitis (LEAN) trial was a phase ll study that enrolled 52 overweight patients with biopsy-confirmed NASH.20 After 48 weeks of treatment with either liraglutide 1.8 mg daily or placebo, 39% (9/23) of participants in the treatment group experienced resolution of definite NASH (via liver biopsy) compared with 9% (2/22) of the placebo group (P = .019). Side effects were reported as mild and transient in both study groups.
A recently published 72-week, placebo-controlled trial with subcutaneous semaglutide enrolled 320 participants with biopsy-confirmed NASH.21 Overall, treatment with semaglutide resulted in a significantly higher percentage of patients experiencing NASH resolution when compared with those randomized to receive placebo.21 Another 24-week trial with dulaglutide showed that GLP-1 receptor agonist treatment significantly reduced liver fat content in patients with T2D and NAFLD.22 Overall, these findings are promising and support the continued investigation of GLP-1 receptor agonists as a viable treatment of NAFLD.
SGLT2 Inhibitors: SGLT2 inhibitors are another important class of glucose-lowering medications that contribute to weight loss and are recommended for use in a variety of populations to mitigate CVD risk, slow progression of CKD, and improve heart failure outcomes.7 Multiple proof-of-concept studies have been published with agents from the SGLT2 inhibitor class demonstrating reductions in liver fat and improvements in transaminase levels.23-28 Ongoing trials investigating the impact of SGLT2 inhibition on liver histology will further inform the potential future use of these agents in patients with NAFLD.
Additional Resources
While it remains to be seen if the glucose-lowering agents discussed above will play a future role in the treatment of NAFLD, there are several resources where pharmacists can direct patients for reliable information. Organizations such as the American Liver Foundation, the Global Liver Institute, and the National Institute of Diabetes and Digestive and Kidney Diseases offer a variety of educational resources as summarized in TABLE 2.

Conclusion
NAFLD is an important condition for pharmacists to be aware of, considering that it is associated with significant morbidity, mortality, and cost to the healthcare system. While there are currently no FDA-approved medications indicated for the treatment of NAFLD, ongoing research examining the role of glucose-lowering agents such as TZDs, GLP-1 receptor agonists, and SGLT2 inhibitors is underway. In the current absence of FDA-approved medications to treat NAFLD, however, pharmacists can play an important role in educating patients regarding the importance of moderation of alcohol intake, adequately managing cardiovascular risk factors, and reinforcing patient education regarding lifestyle modifications to promote weight loss and improve hepatic steatosis.
REFERENCES
1. Spengler EK, Loomba R. Recommendations for diagnosis, referral for liver biopsy, and treatment of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Mayo Clin Proc. 2015;90(9):1233-1246.
2. National Institute of Diabetes and Digestive and Kidney Diseases. Definition & facts of NAFLD & NASH. www.niddk.nih.gov/health-information/liver-disease/nafld-nash/definition-facts. Accessed August 15, 2021.
3. Budd J, Cusi K. Nonalcoholic fatty liver disease: what does the primary care physician need to know? Am J Med. 2020;133(5):536-543.
4. Bril F, Cusi K. Management of nonalcoholic fatty liver disease in patients with type 2 diabetes: a call to action. Diabetes Care. 2017;40(3):419-430.
5. Younossi ZM, Tampi RP, Racila A, et al. Economic and clinical burden of nonalcoholic steatohepatitis in patients with type 2 diabetes in the U.S. Diabetes Care. 2020;43(2):283-289.
6. American Diabetes Association. Standards of Medical Care in Diabetes–2019. Diabetes Care. 2019;42(Suppl 1):S1-S193.
7. American Diabetes Association. Standards of Medical Care in Diabetes–2021. Diabetes Care. 2021;44(Suppl 1):S1-S232.
8. Tomah S, Alkhouri N, Hamdy O. Nonalcoholic fatty liver disease and type 2 diabetes: where do diabetologists stand? Clin Diabetes Endocrinol. 2020;6:9.
9. Saponaro C, Gaggini M, Gastaldelli A. Nonalcoholic fatty liver disease and type 2 diabetes: common pathophysiologic mechanisms. Curr Diab Rep. 2015;15(6):607.
10. Gaggini M, Morelli M, Buzzigoli E, et al. Non-alcoholic fatty liver disease (NAFLD) and its connection with insulin resistance, dyslipidemia, atherosclerosis and coronary heart disease. Nutrients. 2013;5(5):1544-1560.
11. Cusi K. Role of insulin resistance and lipotoxicity in non-alcoholic steatohepatitis. Clin Liver Dis. 2009;13(4):545-563.
12. Chalasani N, Younossi Z, Lavine JE, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67(1):328-357.
13. Bhatt HB, Smith RJ. Fatty liver disease in diabetes mellitus. Hepatobiliary Surg Nutr. 2015;4(2):101-108.
14. Mathurin P, Hollebecque A, Arnalsteen L, et al. Prospective study of the long-term effects of bariatric surgery on liver injury in patients without advanced liver disease. Gastroenterology. 2009;137(2):532-540.
15. Musso G, Cassader M, Rosina F, et al. Impact of current treatments on liver disease, glucose metabolism and cardiovascular risk in non-alcoholic fatty liver disease (NAFLD): a systematic review and meta-analysis of randomised trials. Diabetologia. 2012;55(4):885-904.
16. Soccio RE, Chen ER, Lazar MA. Thiazolidinediones and the promise of insulin sensitization in type 2 diabetes. Cell Metab. 2014;20(4):573-591.
17. Belfort R, Harrison SA, Brown K, et al. A placebo-controlled trial of pioglitazone in subjects with nonalcoholic steatohepatitis. N Engl J Med. 2006;355(22):2297-2307.
18. Cusi K, Orsak B, Bril F, et al. Long-term pioglitazone treatment for patients with nonalcoholic steatohepatitis and prediabetes or type 2 diabetes mellitus: a randomized trial. Ann Intern Med. 2016;165(5):305-315.
19. Bril F, Kalavalapalli S, Clark VC, et al. Response to pioglitazone in patients with nonalcoholic steatohepatitis with vs without type 2 diabetes. Clin Gastroenterol Hepatol. 2018;16(4):558-566.
20. Armstrong MJ, Gaunt P, Aithal GP, et al. Liraglutide safety and efficacy in patients with non-alcoholic steatohepatitis (LEAN): a multicentre, double-blind, randomised, placebo-controlled phase 2 study. Lancet. 2016;387(10019):679-690.
21. Newsome PN, Buchholtz K, Cusi K, et al. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021;384(12):1113-1124.
22. Kuchay MS, Krishan S, Mishra SK, et al. Effect of dulaglutide on liver fat in patients with type 2 diabetes and NAFLD: randomised controlled trial (D-LIFT trial). Diabetologia. 2020;63(11):2434-2445.
23. Choi DH, Jung CH, Mok JO, et al. Effect of dapagliflozin on alanine aminotransferase improvement in type 2 diabetes mellitus with non-alcoholic fatty liver disease. Endocrinol Metab (Seoul). 2018;33(3):387-394.
24. Shimizu M, Suzuki K, Kato K, et al. Evaluation of the effects of dapagliflozin, a sodium-glucose co-transporter-2 inhibitor, on hepatic steatosis and fibrosis using transient elastography in patients with type 2 diabetes and non-alcoholic fatty liver disease. Diabetes Obes Metab. 2018;21(2):285-292.
25. Eriksson JW, Lundkvist P, Jansson P-A, et al. Effects of dapagliflozin and n-3 carboxylic acids on non-alcoholic fatty liver disease in people with type 2 diabetes: a double-blind randomised placebo-controlled study. Diabetologia. 2018;61(9):1923-1934.
26. Itani T, Ishihara T. Efficacy of canagliflozin against nonalcoholic fatty liver disease: a prospective cohort study. Obes Sci Pract. 2018;4(5):477-482.
27. Inoue M, Hayashi A, Taguchi T, et al. Effects of canagliflozin on body composition and hepatic fat content in type 2 diabetes patients with non-alcoholic fatty liver disease. J Diabetes Investig. 2019;10(4):1004-1011.
28. Kuchay MS, Krishan S, Mishra SK, et al. Effect of empagliflozin on liver fat in patients with type 2 diabetes and nonalcoholic fatty liver disease: a randomized controlled trial (E-LIFT trial). Diabetes Care. 2018;41(8):1801-1808.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.





