US Pharm. 2017;42(4):29-31.
ABSTRACT: The CDC recently released updated guidelines for the use of antiretroviral postexposure prophylaxis after sexual contact, injection drug use, and other nonoccupational exposures to HIV. Nonoccupational postexposure prophylaxis (nPEP) is recommended only for HIV-negative persons who present within 72 hours after an exposure that has a substantial risk for HIV transmission. The preferred antiretroviral regimen for nPEP in adult and adolescent patients aged >13 years is tenofovir disoproxil fumarate/emtricitabine (Truvada) plus either raltegravir or dolutegravir. Renal function and other patient-specific factors that require alternative regimens are also included in these guidelines. Pharmacists are in a key position to work with healthcare providers to select the most appropriate nPEP regimen.
Occupational exposure to HIV refers to HIV exposure that occurs in a healthcare setting, while nonoccupational exposure refers to an isolated exposure to blood, genital secretions, or other potentially infectious body fluids outside of healthcare settings.1 Exposures that have the highest estimated per-act risks for HIV transmission from an infected source include blood transfusions, mother-to-child transmission, receptive anal intercourse, needle sharing during injection drug use, and needlesticks.1,2 Guidelines for the use of antiretroviral postexposure prophylaxis after sexual contact, injection drug use, and other nonoccupational exposures to HIV were recently updated by the CDC.1 This article focuses on these guidelines and reviews the epidemiology of nonoccupational exposure, risks for HIV transmission, and current recommendations for treatment and monitoring.
Epidemiology and Transmission
The updated guidelines for nonoccupational exposure focus on preventing HIV after sexual transmission (consensual or nonconsensual) or from injection drug use with an infected individual.1 In 2014, approximately 44,073 people were diagnosed with HIV in the United States.3 Of these cases, 67% of estimated HIV diagnoses were in gay and bisexual men, 24% were from heterosexual contact, and 6% were attributed to injection drug use.4 Within the U.S., the South had the highest rates of HIV diagnoses in 2014 (18.5 per 100,000 people), followed by the Northeast (14.2 per 100,000 people), the West, and the Midwest.4
The per-act risk of acquiring HIV is estimated to be 1.4% for engaging in receptive anal intercourse with a person who is infected with HIV and 0.6% for needle sharing during injection drug use with an HIV-positive person.1,2 The per-act risk of acquiring HIV from an infected person is lower with other types of sexual exposure. The estimated risk is 0.1% for insertive anal intercourse, 0.08% for receptive penile-vaginal intercourse, 0.04% for insertive penile-vaginal intercourse, and is considered negligible from biting or sharing sex toys.1,2 Victims of sexual assault are at increased risk for contracting HIV, as most sexual assaults occur in people aged 18 to 25 years; this is the same age group in which HIV infections are increasing rapidly as compared with other populations.5
Factors that may increase the risk of HIV transmission include the presence of another sexually transmitted disease and a high HIV viral load in the blood.1,2 Viral load is typically higher during acute HIV infection (approximately 2 to 4 weeks postinfection) and in later stages of the disease, when CD4+ T-cell counts drop below 200 cells/mm3. This is especially problematic during the acute phase, when many people are unaware of their infection status and therefore are more likely to spread the virus unknowingly.6
Prevention
The best way to prevent HIV infection is to avoid exposure to the virus. Sexual abstinence from oral, vaginal, and anal sex is 100% effective at preventing HIV.7 Other protective methods include using latex condoms appropriately during each sexual encounter and avoiding reuse of needles and sharing of injection drug equipment.7 Victims of sexual assault and/or abuse or other persons who believe they have been potentially exposed to someone infected with HIV are encouraged to seek care promptly.1
Treatment Guidelines
Initiation of Therapy: Factors for the clinician to consider when deciding whether to initiate nonoccupational postexposure prophylaxis (nPEP) include the HIV status of the potentially exposed person, the HIV status of the source person (if available), the amount of time since the exposure, the characteristics of the exposure, and the frequency of possible HIV exposures.1 nPEP is recommended only for HIV-negative individuals who are seeking care within 72 hours after exposure. Additionally, the risk for HIV acquisition needs to be determined. nPEP is not recommended for exposures of the vagina, rectum, eye, mouth/mucous membrane, or skin to urine, nasal secretions, saliva, sweat, or tears that are not visibly contaminated with blood, as these types of exposures are considered to have a negligible risk for HIV acquisition. Exposure of the vagina, rectum, eye, mouth/mucous membrane, or skin to blood, semen, vaginal/rectal secretions, breast milk, or any body fluid visibly contaminated with blood when the source is known to be HIV-positive is considered to pose a substantial risk for HIV acquisition. nPEP is recommended in these situations when the affected person presents within 72 hours after the exposure. If the HIV status of the source patient is unknown, whether there has been a substantial risk exposure should be evaluated on a case-by-case basis.1
Baseline HIV antibody testing should be performed to determine if the exposed individual has a preexisting HIV infection; in these cases, the individual requires an antiretroviral regimen for the treatment of HIV and is not a candidate for nPEP.1 If there is a delay in test results such that they are not available during the initial evaluation, and nPEP is indicated, the exposed person should be started on prophylaxis. If necessary, nPEP can be discontinued later and transitioned to a full antiretroviral regimen if the patient is found to have HIV.1 Antibody testing should be performed with an FDA-approved rapid antibody test or a combination immunoassay that detects HIV antibodies and antigens.1 Combination tests detect HIV-1 and HIV-2 antibodies as well as the HIV-1 p24 antigen, and they can detect the presence of HIV within 6 weeks of an individual’s exposure.8,9 Because it can take up to 3 months after exposure to HIV for a person to produce antibodies, antibody tests (including most rapid and home tests) may have a false-negative result within this time frame.8,9
Duration of Therapy: The updated guidelines recommend that a three-drug regimen be used routinely as nPEP for a 28-day course of therapy.1
Selection of Specific Agents: The preferred regimen of nPEP for otherwise healthy adults and adolescents aged >13 years, including pregnant women, is tenofovir disoproxil fumarate/emtricitabine (available coformulated as Truvada) plus either raltegravir or dolutegravir. The preferred regimen for adults and adolescents aged >13 years with renal dysfunction (defined as a creatinine clearance 59 mL/min) is zidovudine plus lamivudine (both renally dose-adjusted) in combination with either raltegravir or dolutegravir. Dosing, common or serious adverse effects, and therapeutic considerations for use of each of the preferred first-line agents are included in Table 1.1,10 For situations in which a first-line agent cannot be used or is not tolerated, the updated guidelines include alternative regimens (Table 2).1,10 The guidelines also include preferred and alternative regimens for pediatric patients.1
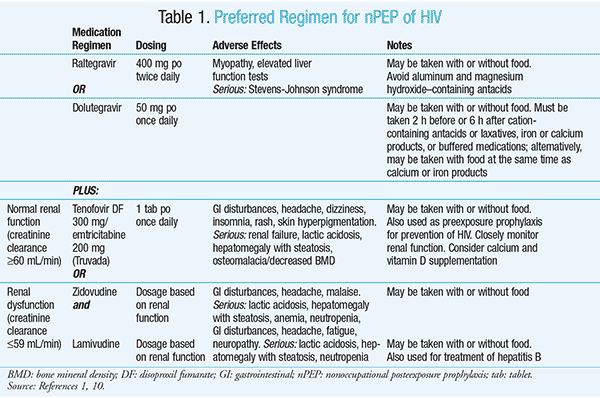

The clinician should be aware that a different salt form of tenofovir—tenofovir alafenamide—was approved by the FDA in combination with emtricitabine under the brand name Descovy in April 2016.11 Although tenofovir alafenamide has been shown to have more favorable effects on markers of renal and bone health as compared with tenofovir disoproxil fumarate, this agent has not been approved for use as preexposure prophylaxis, and it is not included in the updated guidelines for nPEP.1,11,12
Monitoring and Follow-Up
HIV prevention counseling, including risk-reduction behaviors, should be provided to patients seeking care after a possible HIV exposure. Because nPEP does not entirely eliminate the risk for HIV infection, patients receiving nPEP should be counseled to practice protective behaviors during and after therapy, including consistent condom use with sex partners and avoidance of sharing injection equipment with drug-use partners. Psychological counseling and support should be provided for all patients, particularly sexual-assault survivors.1
Patients receiving nPEP should be monitored for drug toxicity, with laboratory tests including CBC and renal and hepatic function performed at baseline and periodically thereafter. The importance of adherence should be emphasized, and information about potential drug interactions and side effects of the medication regimen should be provided. Because patients might be under significant emotional distress when initially seeking care after a potential HIV exposure, providers should consider scheduling a follow-up visit within 7 days of the first visit. The purpose of this visit is to provide additional patient counseling and support, assess medication tolerability and adherence, review any additional test results, and evaluate the need for an alternative nPEP regimen.1
Role of Preexposure Prophylaxis: For people who engage in behaviors that result in frequent or recurrent exposures to HIV, the use of preexposure prophylaxis (PrEP) should be considered after the 28-day nPEP regimen is completed.1 The fixed-dose combination of tenofovir disoproxil fumarate/emtricitabine (Truvada) given daily is approved for this indication.1,13 Patients taking PrEP should be counseled about the importance of adherence to daily medication and of following an integrated risk-reduction strategy, including behavioral modification. It is important for clinicians to exclude acute and chronic HIV infection by conducting HIV testing immediately before PrEP is prescribed and assessing for HIV infection at least every 3 months while the patient is on therapy. Additionally, testing for other sexually transmitted infections (including hepatitis B virus) and the patient’s renal-function status should be performed prior to the initiation of PrEP.13
Conclusion
Pharmacists can play an integral role in the management of nonoccupational exposures to HIV by educating patients about the importance of seeking care within 72 hours of exposure and the appropriate use of nPEP. Pharmacists can work with clinicians to determine if nPEP is appropriate for a patient and to recommend the most suitable nPEP regimen. Pharmacists can ensure that nPEP medications are readily available and provide counseling to patients on behavioral modifications and side effects of drug regimens. Pharmacists can work with patients and healthcare providers to ensure the proper management of nonoccupational exposures to decrease the risk of HIV transmission.
REFERENCES
1. CDC. U.S. Department of Health and Human Services. Updated guidelines for antiretroviral postexposure prophylaxis after sexual, injection drug use, or other nonoccupational exposure to HIV—United States, 2016. www.cdc.gov/hiv/pdf/programresources/cdc-hiv-npep-guidelines.pdf. Accessed October 28, 2016.
2. Patel P, Borkowf CB, Brooks JT, et al. Estimating per-act HIV transmission risk: a systematic review. AIDS. 2014;28:1509-1519.
3. CDC. HIV/AIDS: basic statistics. Updated November 30, 2016. www.cdc.gov/hiv/basics/statistics.html. Accessed October 28, 2016.
4. CDC. HIV in the United States: at a glance. www.cdc.gov/hiv/statistics/overview/ataglance.html. Accessed October 28, 2016.
5. Draughon JE. Sexual assault injuries and increased risk of HIV transmission. Adv Emerg Nurs J. 2012;34:82-87.
6. Fauci AS. Pathogenesis of HIV disease: opportunities for new prevention interventions. Clin Infect Dis. 2007;45(suppl 4):S206-S212.
7. CDC. HIV/AIDS: prevention. Updated December 21, 2016. www.cdc.gov/hiv/basics/prevention.html. Accessed October 28, 2016.
8. CDC. Laboratory testing for the diagnosis of HIV infection: updated recommendations. June 27, 2014. http://stacks.cdc.gov/view/cdc/23446. Accessed October 28, 2016.
9. CDC. HIV testing. Updated June 20, 2016. www.cdc.gov/hiv/testing/index.html. Accessed October 28, 2016.
10. Lexi-Comp Online [Internet database]. Hudson, OH: Lexi-Comp, Inc; 2016. www.lexi.com. Accessed October 28, 2016.
11. U.S. Food and Drug Administration approves Descovy® (emtricitabine, tenofovir alafenamide), Gilead’s third TAF-based HIV therapy. April 4, 2016. www.gilead.com/news/press-releases/2016/4/us-food-and-drug-administration-approves-descovy-emtricitabine-tenofovir-alafenamide-gileads-third-tafbased-hiv-therapy. Accessed October 28, 2016.
12. Panel on Antiretroviral Guidelines for Adults and Adolescents. U.S. Department of Health and Human Services. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-treatment-guidelines/0/. Accessed October 28, 2016.
13. U.S. Public Health Service. Preexposure prophylaxis for the prevention of HIV infection in the United States - 2014. www.cdc.gov/hiv/pdf/prepguidelines2014.pdf. Accessed October 28, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.





