US Pharm. 2019;44(6):19-22.
ABSTRACT: Despite the increased use of statins as a result of expanding indications and guideline recommendations, lack of adherence remains a pervasive issue in medication management. Nonadherence to statins can lead to poor health outcomes, including cardiovascular disease–related emergency department visits, hospitalizations, healthcare costs, and mortality. Medication adherence is multifactorial, and a comprehensive approach is essential for optimization. Potential barriers to statin adherence include costs and adverse effects; interventions that pharmacists can employ include counseling and simplifying regimens.
Cardiovascular disease (CVD) is the leading cause of death globally. In 2016, an estimated 17.9 million people died from CVD, accounting for 31% of global deaths, of which 85% were due to myocardial infarction (MI) and stroke.1 Statins, as 3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitors are known, are commonly prescribed medications that are pivotal for preventing CVD and related events.2
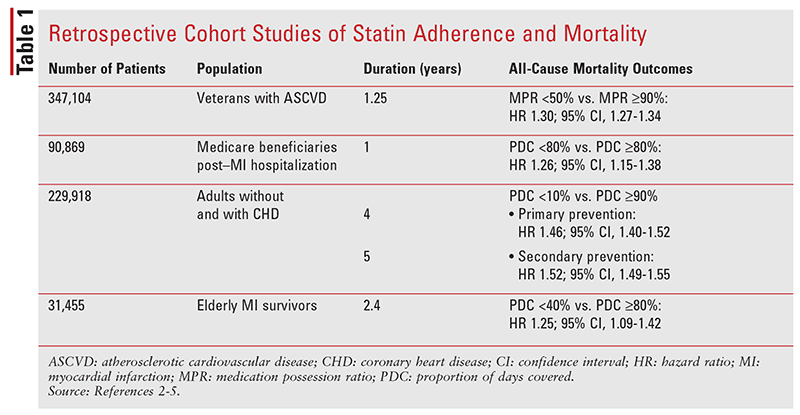
Medication adherence directly influences the extent to which statins prevent CVD events, including mortality (TABLE 1).2-7 Poor statin adherence can also increase CVD-related emergency department visits, hospitalizations, and healthcare costs.6,7 A 10% reduction in statin-medication possession ratio is associated with a 5% increased risk for CVD-related hospitalizations.7 Furthermore, nonadherence over 1 year is associated with a $400 to $900 per patient higher total healthcare cost in the subsequent 18 months. Despite well-documented benefits, statins are underused, and adherence remains suboptimal. Approximately 50% of patients discontinue statin therapy within 1 year, and adherence decreases over time.4,6
Predictors of Statin Nonadherence
Experience with, or apprehension about, adverse events such as muscle-related effects, cognitive or memory problems, and new-onset diabetes are major causes of statin discontinuation.8,9 Patient demographics also influence adherence. Adults aged 50 years or younger and aged 70 years or older, females, African American or Hispanic patients, low-income patients, those with low health literacy, and patients without coronary heart disease (CHD) have displayed lower statin adherence.10-13 In contrast, factors such as middle age, male sex, white ethnicity, higher income, CVD, comorbidities, follow-up monitoring, and cardiologist care were predictors of adherence. In older patients, diabetes and mental health comorbidities have been linked to statin nonadherence.14-16
Financial barriers can also predict nonadherence. In retrospective cohort studies, veterans with no copays were more likely to adhere to statin therapy, and copays were negatively associated with adherence for middle-income and lower-middle-income patients.17 A retrospective study among CHD patients showed more than one-third of patients were nonadherent to statin therapy after initiation because of high copays. Patients with copays of $20 or more were significantly less likely to be adherent compared with copays of $10 or less.18 Choudhry and colleagues found that eliminating copays for CVD medications (statins, beta-blockers, angiotensin-converting enzyme inhibitors, and angiotensin-receptor blockers) improved adherence and reduced the rate of a first major vascular event (11% vs. 12.8%; hazard ratio [HR], 0.86; 95% CI, 0.74-0.99).19
Managing Statin Intolerance
Statin intolerance may occur at any time during therapy, with an incidence of 10% to 15%.20 Myopathy can occur in 3% to 5% of patients, and severe cases (e.g., rhabdomyolysis) are less common (<1%).6 Statin-intolerant patients with a history of MI have a 50% higher risk of recurrent MI, CVD events, and hospitalizations than patients adherent to high-intensity statins.21
Given the benefits of statin therapy, it is prudent to identify “true” intolerance. Placebo-controlled, randomized trials showed that most statin-attributed adverse events were not actually caused by the statin.19 A retrospective cohort study found 92% of rechallenged patients able to tolerate statins long-term.22
The American College of Cardiology provides an app to guide statin intolerance evaluation and management; TABLE 2 summarizes prevention and management strategies.23-28
Rosuvastatin and pravastatin may be more tolerable, owing to hydrophilicity compared with lipophilicity with simvastatin and atorvastatin.29 Dose reduction, alternate-day, or once-weekly administration can improve lipid concentrations, though not to the extent of daily dosing, and are well-tolerated.21 Rosuvastatin’s potency and 20-hour elimination half-life (compared with 14 hours for atorvastatin and 1-2 hours for pravastatin and simvastatin) make it appealing for these dosing methods.29 However, CVD outcomes may decline with these methods, keeping in mind a 20% to 25% reduction in CVD mortality per 38.7 mg/dL LDL cholesterol reduction.30 Therefore, daily dosing should be encouraged as tolerated.
Pharmacist Adherence Interventions
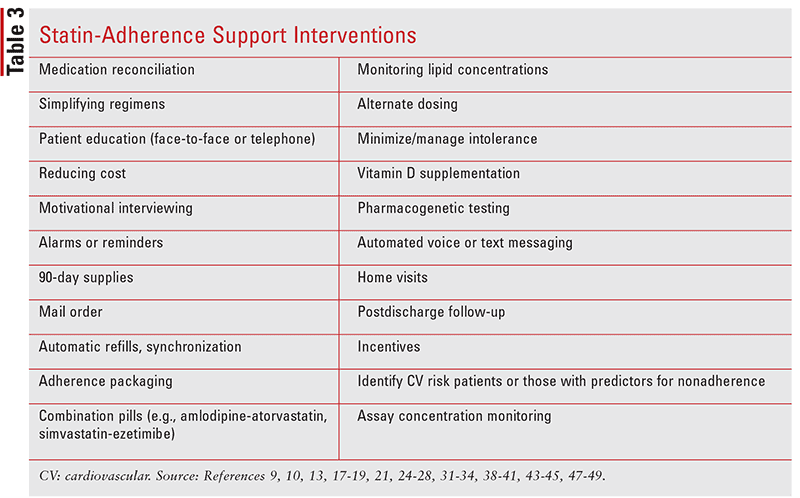
Pharmacists interact frequently with patients and can intervene to promote positive medication-taking behaviors.31-33 TABLE 3 lists examples of strategies to support statin adherence. Providing face-to-face counseling, phone calls, and home visits, and simplifying regimens and reviewing medication-refill information have shown significant adherence improvements.9,34 Pharmacist counseling can lead to the most remarkable improvements in medication use, especially considering that patients with lower perceived CVD risk are less likely to adhere to statins.8,35,36 Hyperlipidemia can be symptomless, rendering the benefits of statin therapy difficult to notice.8 A statin decision aid available on the Mayo Clinic website supports clinical assessments, provides educational visual aids, and can be embedded into the electronic health record (EHR).37
A retrospective cohort study evaluating counseling programs in community pharmacies revealed that patients who received brief counseling sessions conducted by a pharmacist upon statin initiation demonstrated significantly greater medication adherence and persistence than patients who did not receive counseling.34 In-person pharmacist interventions held at the site of medication dispensing showed one of the highest rates of success among various modes of delivery to improve adherence, according to a systematic review.38 For patients with CVD, postdischarge care has been found to improve adherence following MI.39,40
Indirect communication can also improve statin adherence.38 One meta-analysis concluded that text messaging doubled the odds of medication adherence (odds ratio, 2.11; P <.001).41 For patients unreachable by phone, a 6-month pre-post study mailed letters to 460 patients with a late refill for an oral antidiabetic, statin, ACE inhibitor, or angiotensin receptor blocker; 24.1% of patients became adherent after the mailing (P <.001).42 Derose and colleagues combined automated telephone calls with a mailed letter for patients who discontinued statins the prior year.43 The group that received communication had better adherence compared with the group with no outreach (relative risk, 1.63; 95% CI, 1.50-1.76).
Switching a multipill combination (MPC) to a single-pill combination (SPC) can reduce pill burden and result in fewer copays if accessible and/or covered by insurance. A retrospective study compared adherence to SPC versus MPC lipid-modifying medications. The SPC patients were 32% more likely to be adherent to treatment than were the MPC patients.44 Although not available in the United States, an SPC of aspirin, statin, and two blood pressure medications increased adherence by 21% in patients with or at risk of CVD compared with usual care.45
Since socioeconomically challenged patients are prone to lower medication adherence and are at higher risk for CVD mortality, incentives may improve outcomes in these patients.46 A randomized trial demonstrated increased statin adherence rates in patients who each received $172 over 1 year compared with those who did not (34% vs. 27%; P = .01).47 Providing financial incentives to both physicians and patients led to the most significant effect on adherence rates (39% vs. 27%; P <.001) and LDL-cholesterol reduction (8.5 mg/dL; 95% CI, 3.8-13.3; P = .002) compared to controls.
Although financial incentives may not seem feasible in every setting, promoting medication adherence, whether through incentives or other strategies, can improve clinical outcomes, thereby averting subsequent costs associated with poor health. Medication adherence initiatives can also increase revenue (e.g., clinic visits for therapy monitoring, prescriptions being filled on schedule, pay-for-performance reimbursement).31 The EHR can be maximized to allocate resources to patients who would benefit most from these interventions (those with CVD risk and/or predictors of statin nonadherence).48
Conclusion
Although the number of patients indicated to receive statin therapy continues to increase, use remains suboptimal owing to concerns about tolerability and adherence. Meanwhile, CVD remains the leading cause of morbidity and mortality. Pharmacists play a crucial role in reducing CVD risk by improving adherence to statins.
REFERENCES
1. World Health Organization. Fact sheet; 2017. Cardiovascular diseases (CVDs). www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds). Accessed March 23, 2019.
2. Rodriguez F, Maron DJ, Knowles JW, et al. Association of statin adherence with mortality in patients with atherosclerotic cardiovascular disease. JAMA Cardiol. February 13, 2019. Epub ahead of print.
3. Korhonen MJ, Robinson JG, Annis I E, et al. Adherence trade-off to multiple preventive therapies and all-cause mortality after acute myocardial infarction. J Am Coll Cardiol. 2017;70(13):1543-1554.
4. Shalev V, Chodick G, Silber H, et al. Continuation of statin treatment and all-cause mortality: a population-based cohort study. Arch Intern Med. 2009;169(3):260-268.
5. Rasmussen JN, Chong A, Alter DA. Relationship between adherence to evidence-based pharmacotherapy and long-term mortality after acute myocardial infarction. JAMA. 2007;297(2):177-186.
6. Birtcher K. When compliance is an issue—how to enhance statin adherence and address adverse effects. Curr Atheroscler Rep. 2015;17(1):471.
7. Pittman DG, Chen W, Bowlin SJ, Foody JM. Adherence to statins, subsequent healthcare costs, and cardiovascular hospitalizations. Am J Cardiol. 2011;107(11):1662-1666.
8. Rosenson RS. Statin non-adherence: clinical consequences and proposed solutions. F1000Res. 2016;5.
9. Lansberg P, Lee A, Lee ZV, et al. Nonadherence to statins: individualized intervention strategies outside the pill box. Vasc Health Risk Manag. 2018;14:91-102.
10. Mann DM, Woodward M, Muntner P, et al. Predictors of nonadherence to statins: a systematic review and meta-analysis. Ann Pharmacother. 2010;44(9):1410-1421.
11. Colantonio LD, Rosenson RS, Deng L, et al. Adherence to statin therapy among US adults between 2007 and 2014. J Am Heart Assoc. 2019;8(1):e010376.
12. Hope HA, Binkley GM, Fenton S, et al. Systematic review of the predictors of statin adherence for the primary prevention of cardiovascular disease. PLoS One. 2019;14(1):e0201196.
13. Campione JR, Sleath B, Biddie AK, Weinberger M. The influence of physicians’ guideline compliance on patients’ statin adherence: a retrospective cohort study. Am J Geriatr Pharmacother. 2005;3(4):229-239.
14. Morotti K, Lopez J, Vaupel V, et al. Adherence to and persistence with statin therapy in a veteran population. Ann Pharmacother. 2019;53(1):43-49.
15. Ofori-Asenso R, Ilomäki J, Tacey M, et al. Predictors of first-year nonadherence and discontinuation of statins among older adults: a retrospective cohort study. Br J Clin Pharmacol. 2019;85(1):227-235.
16. Katzmann JL, Mahfoud F, Böhm M, et al. Association of medication adherence and depression with the control of low-density lipoprotein cholesterol and blood pressure in patients at high cardiovascular risk. Patient Prefer Adherence. 2018;13:9-19.
17. Kazerooni R, Bounthavong M, Watanabe JH. Association of copayment and statin adherence stratified by socioeconomic status. Ann Pharmacother. 2013;47(11):1463-1470.
18. Ye X, Gross C, Schommer J, et al. Association between copayment and adherence to statin treatment initiated after coronary heart disease hospitalization: a longitudinal, retrospective, cohort study. Clin Ther. 2007;29(12):2748-2757.
19. Choudhry NK, Avorn J, Glynn RJ, et al. Full coverage for preventive medications after myocardial infarction. N Engl J Med. 2011;365:(22):2088-2097.
20. Banach M, Rizzo M, Toth PP, et al. Statin intolerance—an attempt at a unified definition. Position paper from an International Lipid Expert Panel. Arch Med Sci. 2015;11(1):1-23.
21. Awad K, Mikhailidis DP, Toth PP, et al. Efficacy and safety of alternate-day versus daily dosing of statins: a systematic review and meta-analysis. Cardiovasc Drugs Ther. 2017;31(4):419-431.
22. Zhang H, Plutzky J, Skentzos S, et al. Discontinuation of statins in routine care settings: cohort study. Ann Intern Med. 2013;158(7):526-534.
23. American College of Cardiology. Statin Intolerance App. 2016. http://tools.acc.org/statinintolerance/#!/subpages/alternateCauses. Accessed March 21, 2019.
24. Backes JM, Ruisinger JF, Gibson CA, Moriarty PM. Statin-associated muscle symptoms—managing the highly intolerant. J Clin Lipidol. 2017;11:24-33.
25. Grundy SM, Stone NJ, Bailey AL, et al. AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: executive summary. J Am Coll Cardiol. 2018. November 3, 2018. Epub ahead of print.
26. Wu Z, Camargo CA Jr, Khaw KT, et al. Effects of vitamin D supplementation on adherence to and persistence with long-term statin therapy: secondary analysis from the randomized, double-blind, placebo-controlled ViDA study. Atherosclerosis. 2018;273:59-66.
27. Kang JH, Nguyen QN, Mutka J, Le QA. Rechallenging statin therapy in veterans with statin-induced myopathy post vitamin D replenishment. J Pharm Pract. 2017;30(5):521-527.
28. Li JH, Joy SV, Haga SB, et al. Genetically guided statin therapy on statin perceptions, adherence, and cholesterol lowering: a pilot implementation study in primary care patients. J Pers Med. 2014;4(2):147-162.
29. McTaggart F. Comparative pharmacology of rosuvastatin. Atheroscler Suppl. 2003;4(1):9-14.
30. MRC/BHF Heart Protection Study Investigators. MRC/BHF Heart Protection Study of cholesterol lowering with simvastatin in 20,536 high-risk individuals: a randomised placebo-controlled trial. Lancet. 2002;360:7-22.
31. Kim J, Combs K, Downs, J, Tillman F. Medication adherence: the elephant in the room. US Pharm. 2018;43(1):30-34.
32. Bosworth HB, Brown JN, Danus S, et al. Evaluation of a packaging approach to improve cholesterol medication adherence. Am J Manag Care. 2017;23(9):e280-e286.
33. Leslie RS, Gilmer T, Natarajan L, Hovell M. A multichannel medication adherence intervention influences patient and prescriber behavior. J Manag Care Spec Pharm. 2016;22(5):526-538.
34. Taitel M, Jiang J, Rudkin K, et al. The impact of pharmacist face-to-face counseling to improve medication adherence among patients initiating statin therapy. Patient Prefer Adherence. 2012;6:323-329.
35. Gatwood J, Bailey JE. Improving medication adherence in hypercholesterolemia: challenges and solutions. Vasc Health Risk Manag. 2014;10:615-625.
36. Fung V, Graetz I, Reed M, Jaffe MG. Patient-reported adherence to statin therapy, barriers to adherence, and perceptions of cardiovascular risk. PLoS One. 2018;13(2):e0191817.
37. Statin choice decision aid. Mayo Clinic website. http://statindecisionaid.mayoclinic.org/. Accessed March 24, 2019.
38. Cutrona SL, Choudhry NK, Fischer MA, et al. Modes of delivery for interventions to improve cardiovascular medication adherence. Am J Manag Care. 2010;16(12):929-942.
39. Hickson RP, Robinson JG, Annis IE, et al. Changes in statin adherence following an acute myocardial infarction among older adults: patient predictors and the association with follow-up with primary care providers and/or cardiologists. J Am Heart Assoc. 2017;6(10).
40. Colantonio LD, Huang L, Monda KL, et al. Adherence to high-intensity statins following a myocardial infarction hospitalization among Medicare beneficiaries. JAMA Cardiol. 2017;2(8):890-895.
41. Thakkar J, Kurup R, Laba TL, et al. Mobile telephone text messaging for medication adherence in chronic disease: a meta-analysis. JAMA Intern Med. 2016;176(3):340-349.
42. Mann A, Esse TW, Serna O, et al. Effectiveness of mailed letters to improve medication adherence among Medicare Advantage Plan participants with chronic conditions. Patient Prefer Adherence. 2018;13:37-46.
43. Derose SF, Green K, Marrett E, et al. Automated outreach to increase primary adherence to cholesterol-lowering medications. JAMA Intern Med. 2013;173(1):38-43.
44. Kamat SA, Bullano MF, Chang CL, et al. Adherence to single-pill combination versus multiple-pill combination lipid-modifying therapy among patients with mixed dyslipidemia in a managed care population. Curr Med Res Opin. 2011;27(5):961-968.
45. Thom S, Poulter N, Field J, et al. Effects of a fixed-dose combination strategy on adherence and risk factors in patients with or at high risk of CVD: the UMPIRE randomized clinical trial. JAMA. 2013;310(9):918-929.
46. Sidney S, Quesenberry C, Jaffe M, et al. Recent trends in cardiovascular mortality in the United States and public health goals. JAMA Cardiol. 2016;1:594-599.
47. Asch DA, Troxel AB, Stewart WF, et al. Effect of financial incentives to physicians, patients, or both on lipid levels: a randomized clinical trial. JAMA. 2015;314(18):1926-1935.
48. Lucas JE, Bazemore TC, Alo C, et al. An electronic health record based model predicts statin adherence, LDL cholesterol, and cardiovascular disease in the United States Military Health System. PLoS One. 2017;12(11):e0187809.
49. Vethe NT, Munkhaugen J, Andersen AM, et al. A method for direct monitoring of atorvastatin adherence in cardiovascular disease prevention: quantification of the total exposure to parent drug and major metabolites using 2-channel chromatography and tandem mass spectrometry. Ther Drug Monit. 2019;41(1):19-28.
To comment on this article, contact rdavidson@uspharmacist.com.