US Pharm. 2014;39(11):56-62.
ABSTRACT: Anxiety disorders are one of the most frequently occurring mental disorders. They are the result of abnormal neurotransmitter function within the central nervous system. Treatment options for anxiety disorders include lifestyle modifications, psychotherapy, and pharmacotherapy. While several drug classes may be employed for the treatment of anxiety disorders, antidepressants such as the selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors are considered first-line therapies. Pharmacists should be aware of adverse drug reactions and drug interactions for the various medications used in the treatment of anxiety disorders.
Anxiety disorders are a collection of psychological disorders characterized by excessive fear and anxiety that interfere with normal daily activities. Patients may demonstrate maladaptive behaviors including avoidance strategies. Anxiety is one of the most frequently occurring mental disorders, encompassing approximately 18% of adults aged >18 years in the United States, with women being 60% more likely than men to experience an anxiety disorder in their lifetime.1 Additionally, about 8% of adolescents aged 13 to 18 years have a diagnosed anxiety disorder.1
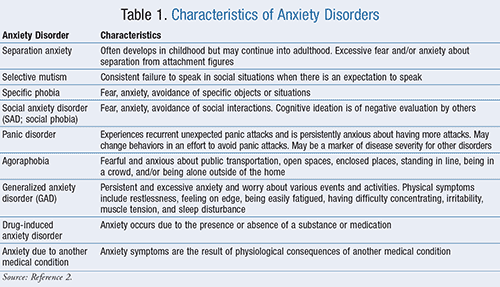
The Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) was released in May 2013.2 TABLE 1 lists the disorders outlined in the DSM-5 as well as the main characteristics of those disorders. The DSM-5 reclassifies obsessive-compulsive disorder (OCD) in the new obsessive-compulsive and other related disorders chapter, while posttraumatic stress disorder (PTSD) is included in the trauma- and stressor-related disorders chapter. These chapters follow the one on anxiety disorders and are closely related.2
Pathophysiology
Various structures within the brain as well as the abnormal function of several neurotransmitters are associated with the development of anxiety disorders. The locus coeruleus, which is the primary norepinephrine (NE)-containing site within the brain, projects NE to other areas of the brain in order to modulate mood, cognition, and sleep. Fear is processed by the amygdala, which also stores emotional memories. The hippocampus is responsible for encoding memories and is involved in fear conditioning. Stress response is mediated by the hypothalamus.3,4
The neurotransmitters involved in the pathogenesis of anxiety disorders include NE, serotonin (5-HT1), dopamine, glutamate, and gamma-aminobutyric acid (GABA). Compared to the general population, patients with anxiety disorders display increased NE and decreased 5-HT1, making these neurotransmitters main targets for pharmacologic therapy. Dopamine is also upregulated during states of anxiety, thus dopamine-D2 blockade has exhibited anxiolytic effects. However, dopaminergic signaling may also produce feelings of confidence. Therefore, while some patients do respond well to prodopa-minergic agents, others may report an increase in anxiety symptoms. The glutamate-NMDA (N-methyl-d-aspartate) receptor substrate mediates learning and memory including fear consolidation. GABA is decreased in anxiety disorders. Therefore, drugs that increase GABA neurotransmission produce anxiolytic effects in these patients.3,4
Treatment
Treatment options for anxiety disorders include both pharmacotherapy and psychotherapy. These treatment options may be implemented together or alone based on the patient’s needs and the specific anxiety disorder. At the initiation of pharmacotherapy, the patient should be monitored frequently to assess for symptom improvement and emergence of adverse effects. Patients should be counseled on all new drug therapies, and the lowest effective dosage should be used. Pharmacotherapy should be continued for at least 12 months after symptom improvement to decrease the risk of relapse.5 The duration of pharmacotherapy should be individualized, as some patients may be titrated off their medications while others will require lifelong maintenance therapy.
Nonpharmacologic Therapy
Cognitive behavioral therapy (CBT) is an integral part of the therapeutic plan for anxiety disorders. The purpose of CBT is to identify and correct dysfunctional cognitions. This therapy helps patients to control their emotional distress and decrease their use of maladaptive behaviors such as avoidance strategies.6 For some patients, CBT alone has been shown to effectively control anxiety symptoms, while others require the addition of pharmacotherapy.7
Lifestyle modifications may also be effective in decreasing anxiety symptoms. These lifestyle modifications include limiting the consumption of caffeine, drugs of abuse, and stimulants. Caffeine in high doses has been shown to exacerbate anxiety, and patients with anxiety disorders may be more sensitive and reactive to the effects of caffeine compared to the general population.6 Exercise, both aerobic and nonaerobic, has shown benefit in decreasing anxiety disorder symptoms.8 Lifestyle modifications should be utilized as adjunctive therapy with CBT, pharmacotherapy, or both.
Relaxation breathing techniques may be beneficial in situations that trigger anxiety. Recommending that patients keep a journal may increase awareness regarding anxiety triggers and improve self-awareness of personal coping behaviors. Last, massage therapy and acupuncture may improve relaxation and decrease anxiety in patients with generalized anxiety disorder (GAD).9
Pharmacologic Therapy
Newer Antidepressants: Selective serotonin reuptake inhibitors (SSRIs) are considered a first-line treatment for anxiety disorders (TABLE 2).7,10 These agents selectively inhibit the presynaptic reuptake of serotonin by the serotonin transporter, which increases the concentration available to bind to postsynaptic receptors.9 Studies have shown comparable efficacy between SSRIs.5 When initiating therapy, SSRIs should be started at a low dose and titrated up to achieve a therapeutic dose. The anxiolytic effect may take as little as 2 weeks or up to 8 weeks to manifest. SSRIs have a black box warning for an increased risk of suicidal thoughts and behaviors in children and young adults. Therefore, patients should be carefully monitored for suicidality, especially at the initiation of therapy.10
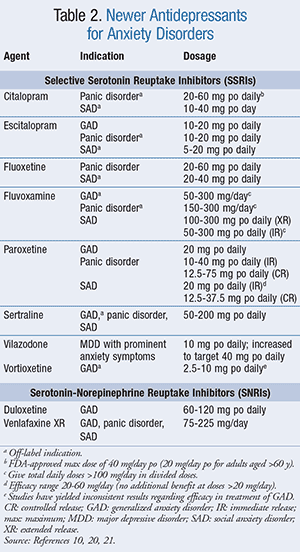
The most common adverse drug reactions (ADRs) are gastrointestinal (GI) in nature, with nausea being the most common, especially in the first 2 weeks of therapy. Other ADRs include sexual dysfunction, impaired platelet aggregation with an increased risk of bleeding, and hyponatremia. Citalopram may prolong the QT interval, especially at doses >40 mg/day or when combined with other medications that have this effect.11 Additionally, patients should be monitored for concomitant therapy with other serotonergic agents due to an increased risk of serotonin syndrome and neuroleptic malignant syndrome. In these emergency situations, abrupt discontinuation is acceptable. However, during routine discontinuation, SSRIs should be gradually tapered to avoid withdrawal symptoms.12 The agents should be used with caution in pregnant patients, as most SSRIs are Pregnancy Category C, with the exception of paroxetine, which is Category D.10
Serotonin-norepinephrine reuptake inhibitors (SNRIs) (TABLE 2) are often initiated after failure of, or inadequate response to, an SSRI, as those agents may be less well tolerated.12 SNRI agents inhibit both serotonin and norepinephrine reuptake.10 While these drugs are a potentially effective therapy, some patients actually experience an increase in physiological anxiety symptoms due to the increased norepinephrine-mediated signaling.4 As with SSRIs, SNRIs carry a black box warning regarding the increased risk of suicidal thoughts and behavior in children and young adults, and the most common ADRs are GI-related. Other ADRs include central nervous system (CNS) effects such as headache and sleep disturbances, impaired platelet aggregation with increased risk of bleeding, hyponatremia, sexual dysfunction, blood pressure elevations, and orthostatic hypotension. Venlafaxine is noted to have the most effect on sustained blood pressure elevations.7,10 SNRIs may cause hepato-toxicity. The dose of venlafaxine should be adjusted in patients with existing hepatic impairment, while duloxetine should be avoided entirely in this population.10 The anxiolytic effect of SNRIs has a latency period of at least 2 weeks.
Older Antidepressants: The mechanism of tricyclic antidepressants (TCAs) (TABLE 3) is not fully understood, though they are believed to interfere with norepinephrine and serotonin reuptake, subsequently increasing their activity at the postsynaptic receptors.10 This class of medications has a comparable efficacy to SSRIs and SNRIs, yet are less commonly employed in the treatment of anxiety disorders because of the increased frequency of adverse effects.7,12 Furthermore, TCAs are potentially lethal in the event of an overdose, and their use should be avoided in patients who are considered at risk for suicide.12 TCAs should be initiated at a low dose and titrated up to achieve a therapeutic dose.
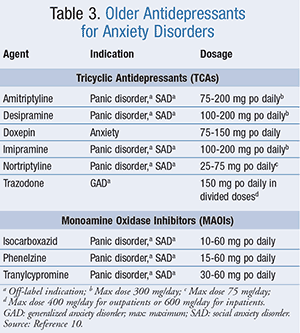
The most common ADRs for TCAs are anticholinergic and antihistaminic. Other ADRs include orthostatic hypotension, increased intraocular pressure, weight gain, and seizures. TCAs may also cause cardiac rhythm abnormalities and are contraindicated in patients recovering from a myocardial infarction.10 Furthermore, due to the risk of QT prolongation, patients with cardiac conduction defects should not receive TCAs. Abrupt discontinuation should be avoided in order to prevent withdrawal symptoms and cholinergic rebound effects. As with other anti-depressants, TCAs may have a latency period of at least 2 weeks.10
In the treatment of anxiety disorders, monoamine oxidase inhibitors (MAOIs) (TABLE 3) are used mainly for patients who are refractory to other therapies, especially when they experience phobic reactions or have mixed anxiety-depressive states.7,12 MAOIs exert their effects by binding to MAO, which decreases its activity and subsequently increases the concentration of NE, dopamine, and 5-HT1.10 These drugs also carry the black box warning for increased risk of suicidality in children and young adults. MAO inhibition in the GI tract and liver enhances tyramine absorption, which may cause severe hypertension and potentially fatal hypertensive crisis as tyramine displaces NE from the adrenergic storage site.10
Patients should be counseled on avoiding foods high in tyramine, and the pharmacist should review the patient’s full medication profile to avoid coadministration of MAOIs. Serotonin syndrome may also occur if MAOIs are administered concomitantly with serotonergic agents. Other ADRs include orthostatic hypotension, sleep disturbances, drowsiness, fatigue, headache, dizziness, and, rarely, psychosis, mania, and toxic delirium. In order to avoid insomnia, patients should have their drug administered in the morning or afternoon.7 MAOIs should be tapered prior to discontinuation to avoid symptoms of withdrawal such as nausea, vomiting, psychosis, convulsions, and vivid nightmares with agitation.
Benzodiazepines: Benzodiazepines (TABLE 4) act on the GABA-benzodiazepine receptor complex to enhance the inhibitory effects of GABA.10 This class of medications is effective for managing acute anxiety exacerbations, or may be utilized at the initiation of SSRI or SNRI therapy until a patient is established on a therapeutic dose. Benzodiazepines are not recommended for chronic use due to the adverse-effect profile associated with long-term use ( >2 months). These adverse effects include dependence and potentially fatal withdrawal. Alprazolam has demonstrated significant rebound anxiety, withdrawal symptoms, and propensity for abuse.13 Therefore, agents with a longer half-life and slow onset of action, such as clonazepam, are less susceptible to misuse and may be preferred.10,13
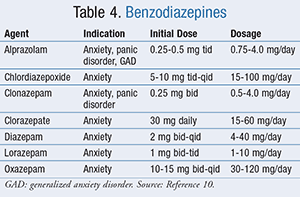
The use of benzodiazepines in patients with a history of substance abuse is a relative contraindication because of the increased risk of addiction and abuse. In addition to the risk of dependence and withdrawal syndrome, other ADRs include respiratory depression and overdose. Due to the risk of respiratory depression, these agents should be avoided whenever possible in patients with respiratory diseases. Small doses of benzodiazepines may be utilized long term in patients with refractory anxiety who have failed multiple therapies.7
Buspirone: Buspirone’s mechanism of action is not fully understood, but it is believed to suppress serotonergic activity while enhancing noradrenergic and dopaminergic actions.14 It has a high affinity for 5-HT1A receptors and a moderate affinity for dopamine D2 receptors but does not affect the benzodiazepine-GABA receptors. The recommended initial dose is 7.5 mg twice daily with a maximum dose of 60 mg/day. Buspirone is not known to cause drug dependence.7 Therefore, it may be considered in place of benzodiazepines in patients with a history of substance abuse.5,7 Dose titration should occur in 5 mg increments every 2 to 3 days. The onset of anxiolytic effects may take up to 2 weeks. The most common ADRs are CNS effects such as dizziness, nervousness, insomnia, drowsiness, and lightheadedness. Additionally, concomitant use of buspirone with MAOIs is not recommended because of the risk of elevated blood pressure.14
Gabapentin and Pregabalin: Gabapentin and pregabalin are calcium channel modulators that are used off-label for the treatment of social anxiety disorder (SAD).10,15 They bind to the 2--subunit of the voltage-gated calcium channels in the CNS tissues, which subsequently modulates the release of excitatory neurotransmitters.16 Neither directly binds to the GABA-benzodiazepine receptors. However, prolonged exposure to these agents does increase the rate of functional GABA transport. The main ADRs are dizziness, sedation, dry mouth, weight gain, impaired coordination, and impaired psychomotor and cognitive function. Randomized, controlled trials have demonstrated the efficacy of pregabalin in acute treatment and relapse prevention in GAD.12,17 The usual dose of pregabalin is 600 mg/day. Gabapentin 3,600 mg/day has shown a favorable risk-benefit ratio in comparison to placebo for the treatment of SAD.18
Hydroxyzine: Hydroxyzine is a first-generation antihistamine that blocks the histamine-1 (H1) receptors and, to a lesser extent, the muscarinic and serotonergic receptors. Its use for the treatment of GAD is off-label. Common ADRs include sedation, xerostomia, tremor, hypotension, and tachycardia.10 Caution is advised when using hydroxyzine in combination with other agents that may cause CNS depression. Hydroxyzine is listed as Category X for use in the first trimester of pregnancy and as Category C for the second and third trimesters. A meta-analysis concluded that hydroxyzine showed “comparable efficacy versus active medication.”19 Due to a lack of comparative data regarding first-line options (SSRIs and SNRIs), hydroxyzine is not recommended first-line, but may be considered an effective second-line option.19 The usual daily dose is 50 mg orally in divided doses.10
Conclusion
The treatment of anxiety disorders can be challenging. Pharmacists can help prepare patients during the pharmacotherapy initiation by counseling them on therapeutic onset, possible ADRs, and drug interactions. Furthermore, the pharmacist should monitor for compliance and work collaboratively with physicians to ensure that the patient receives individualized, effective, and safe therapy.
REFERENCES
1. What is anxiety disorder? National Institute of Mental Health. www.nimh.nih.gov/health/topics/anxiety-disorders/index.shtml. Accessed July 1, 2014.
2. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Arlington, VA: American Psychiatric Publishing; 2013.
3. DiPiro JT, Talbert RL, Yee GC, et al. Pharmacotherapy: A Pathophysiologic Approach. New York, NY: McGraw-Hill Medical; 2002.
4. Bystritsky A, Khalsa SS, Cameron ME, Schiffman J. Current diagnosis and treatment of anxiety disorders. P T. 2013;38(1):30-57.
5. Koen N, Stein DJ. Pharmacotherapy of anxiety disorders: a critical review. Dialogues Clin Neurosci. 2011;13(4):423-437.
6. Hofmann SG, Smits JA, Asnaani A, et al. Cognitive enhancers for anxiety disorders. Pharmacol Biochem Behav. 2011;99(2):275-284.
7. Bandelow B, Zohar J, Hollander E, et al. World Federation of Societies of Biological Psychiatry (WFSBP) guidelines for the pharmacological treatment of anxiety, obsessive-compulsive and post-traumatic stress disorders, first revision. World J Biol Psychiatry. 2008;9(4):248-312.
8. Jayakody K, Gunadasa S, Hosker C. Exercise for anxiety disorders: systematic review. Br J Sports Med. 2014;48(3):187-196.
9. McPherson F, McGraw L. Treating generalized anxiety disorder using complementary and alternative medicine. Altern Ther Health Med. 2013;19(5):45-50.
10. Clinical Pharmacology [online database]. www.clinicalpharmacology.com. Tampa, FL: Gold Standard, Inc; 2014. Accessed July 1, 2014.
11. Celexa (citalopram) package insert. St. Louis, MO: Forest Pharmaceuticals, Inc; 2014.
12. Baldwin DS, Anderson IM, Nutt DJ, et al. Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol. 2014;28(5):403-439.
13. Moylan S, Staples J, Ward SA, et al. The efficacy and safety of alprazolam versus other benzodiazepines in the treatment of panic disorder. J Clin Psychopharmacol. 2011;31(5):647-652.
14. Buspirone HCL (buspirone hydrochloride) package insert. Cincinnati, OH: Patheon Pharmaceuticals Inc; 2013.
15. Cassano GB, Baldini Rossi N, Pini S. Psychopharmacology of anxiety disorders. Dialogues Clin Neurosci. 2002;4(3):271–285.
16. Lyrica (pregabalin) package insert. New York, NY: Pfizer Inc; December 2013.
17. Baldwin DS, Ajel K, Masdrakis VG, et al. Pregabalin for the treatment of generalized anxiety disorder: an update. Neuropsychiatr Dis Treat. 2013;9: 883-892.
18. Pande AC, Davidson JR, Jefferson JW, et al. Treatment of social phobia with gabapentin: a placebo-controlled study. J Clin Psychopharmacol. 1999;19(4):341-348.
19. Guaiana G, Barbui C, Cipriani A. Hydroxyzine for generalised anxiety disorder. Cochrane Database Syst Rev. 2010;(12):CD006815.
20. Bidzan L, Mahableshwarkar AR, Jacobsen P, et al. Vortioxetine (Lu AA21004) in generalized anxiety disorder: results of an 8-week, multinational, randomized, double-blind, placebo-controlled clinical trial. Eur Neuropsychopharmacol. 2012;22(12):847-857.
21. Mahableshwarkar AR, Jacobsen PL, Serenko M, Chen Y. A randomized, double-blind, fixed-dose study comparing the efficacy and tolerability of vortioxetine 2.5 and 10 mg in acute treatment of adults with generalized anxiety disorder. Hum Psychopharmacol. 2014;29(1):64-72.
To comment on this article, contact rdavidson@uspharmacist.com.





