US Pharm. 2015;40(12):HS5-HS11.
ABSTRACT: Cirrhosis is a heterogeneous diagnosis that impacts liver function, including the metabolism and clearance of medications, but the exact effect remains unclear. Misconceptions and significant practice variability exist among healthcare professionals regarding analgesic use in patients with liver dysfunction. Based on limited safety and efficacy data, acetaminophen is the preferred analgesic in patients with liver disease who are not actively drinking, and it may be dosed up to 2 to 3 g/day. Nonsteroidal anti-inflammatory drugs (NSAIDs) should be avoided due to their adverse effects of renal impairment, fluid retention, and increased bleeding risk. Opioids should be used cautiously and initiated with immediate-release products at low doses with extended intervals and close monitoring. All pain medications should be titrated carefully to achieve safe and adequate pain relief in patients with hepatic impairment.
Cirrhosis is defined as permanent liver fibrosis secondary to damage or injury. It is a heterogeneous diagnosis, resulting in varying degrees of nodule formation, organ dysfunction, complications, and significant morbidity and mortality. Causes of cirrhosis include alcohol abuse, hepatitis B and C, cancer, and nonalcoholic steato-hepatitis.1 “Chronic liver disease and cirrhosis” as the primary diagnosis accounted for more than 101,000 hospital admissions in 2010.2 In 2013, it was reported as a leading cause of death in people ages 25 to 64 years, and the 12th leading cause of death for all ages, resulting in over 36,000 deaths.2
Unfortunately, these numbers may be underestimated due to diagnostic criteria limitations of International Classification of Diseases (ICD) codes, which lack consideration for cirrhosis-related complications. One study suggests that liver-related deaths may be as much as twice as high as what is currently reported by the CDC.3
Healthcare providers are familiar with the liver’s vital role in the metabolism and clearance of many drugs. Cirrhosis can lead to the development of portohepatic shunts, ultimately decreasing the first-pass metabolism that occurs with many oral medications. Decreased albumin production can also result in increased free-drug concentrations of highly protein-bound medications. It is generally thought that as liver dysfunction progresses, drug elimination is impaired; however, patients with chronic hepatitis or hepatic cancer, in the absence of cirrhosis, may only have a small change in drug clearance.4 It remains unclear to what extent cirrhosis affects drug disposition.
While certain medications have specific renal adjustment parameters, hepatic dose adjustments may not be readily available. Many studies have evaluated pharmacokinetics and patients’ analgesic serum levels, which do not necessarily correlate with patient response. Other available studies have focused primarily on hepatitis C, although many patients develop liver dysfunction and cirrhosis secondary to other causes. Child-Pugh scores, which are not commonly calculated in practice, often direct dosing, but resultant recommendations may still be vague without clear guidance for clinical application. The management of patients may be further complicated by concomitant renal impairment, whether related to liver disease, such as hepatorenal syndrome, or another cause.4
Analgesic Use in Cirrhosis
Pain management is often a challenge for healthcare professionals, but it remains a very important component of providing quality patient care and is a common factor in patient satisfaction.5 A high prevalence of pain has been found among patients with chronic liver disease, reported between 32% and 77%.6-8 Pain and opioid-based pain regimens have been found to be significant predictors of healthcare utilization among liver disease patients.8
One retrospective review found that 77% of preliver transplant patients (n = 108) reported bodily pain, with more than one-third indicating multiple sites.7 The pain reportedly affected work, sleep, mobility, appetite, enjoyment, and mood. Approximately 90% reported receiving pharmacologic therapy; however, only 33% perceived relief. Short-acting opioids were prescribed 40% of the time, and 32% of patients reported being prescribed five or more pain medications. The authors concluded that pain in end-stage liver disease (ESLD) patients is very common, impacts functionality, and is often undertreated, despite the numerous pharmacologic therapies prescribed.7
Polypharmacy was suggested as a common approach to pain management, possibly due to overcautious prescribers using very low doses of several medications rather than careful titration and optimization of fewer agents.7 Polypharmacy increases costs, complicates the understanding of agent efficacy, creates therapeutic duplications, and increases the inherent risk of drug-drug interactions and adverse effects. Pain management in cirrhosis presents a complex area of pharmacotherapy with unique considerations of altered liver pathophysiology and a fine balance between adequate pain relief and avoidance of significant potential adverse effects.7
Acetaminophen
It is commonly believed that acetaminophen should be avoided in liver dysfunction patients due to its well-known hepatotoxicity risk in overdose. This risk is directly related to the saturation of other metabolic pathways and depletion of glutathione (GSH) stores.5 Approximately 5% of acetaminophen is metabolized by CYP450 enzymes (predominantly 2E1, as well as 1A3 and 3A4) to the reactive metabolite N-acetyl-p-benzoquinone imine (NAPQI). In the presence of GSH, NAPQI is conjugated and excreted renally. When GSH is unavailable, NAPQI accumulates and causes hepatocyte necrosis and apoptosis.
Alcoholics may develop a CYP2E1 induction due to chronic alcohol ingestion and have decreased GSH levels from malnutrition. Such physiological changes may cause these patients to be at increased risk of acetaminophen-induced liver injury. Consequently, concern exists that all cirrhosis patients have lower GSH levels and increased hepatotoxicity risk with acetaminophen. Although a prolonged half-life has been documented in cirrhosis patients, no prospective, long-term studies are available to evaluate the safety of acetaminophen. Some studies suggest hepatic injury is rare with appropriate dosing and that acetaminophen is well-tolerated in stable chronic liver disease up to 4 g/day, with no evidence of increased CYP activity or critically depleted GSH stores.5,9,10
Total daily acetaminophen doses of 2 to 3 g are recommended for both short- and long-term use in cirrhotic patients who are not actively drinking alcohol, based on expert opinion.4,5 Contrary to common belief, acetaminophen is considered the preferred agent in liver dysfunction when kept within a total daily dosing limit, but should be avoided in patients who actively drink alcohol.4,11,12 IV acetaminophen is contraindicated in severe hepatic impairment and severe active liver disease.13
Nonsteroidal Anti-inflammatory Drugs (NSAIDs)
In cirrhotic patients, alterations in metabolism and bioavailability are expected with NSAIDs because of significant CYP metabolism and protein binding.5 However, the concern for cirrhosis patients primarily relates to the physiologic effects of NSAIDs. These drugs inhibit prostaglandin synthesis leading to decreased renal blood flow and glomerular filtration rate (GFR) and impaired renal sodium and water excretion, which may worsen ascites and increase the risk of hepatorenal syndrome in cirrhosis patients.4 NSAIDs are known to cause gastrointestinal irritation, ulceration, and perforation and increase patients’ overall risk of bleeding due to inhibition of platelet thromboxane A2 produc-tion.4,5 Due to thrombocytopenia, decreased synthesis of clotting factors, and esophageal varices associated with portal hypertension, cirrhosis patients may be at a higher risk of bleeding, which can be life-threatening.4 For these reasons, NSAIDs should be avoided in cirrhosis.
Opioids
Opioids are the most common drug class for analgesia, particularly moderate and severe pain or pain not relieved by acetaminophen and NSAIDs. Opioids can contribute to significant complications with cirrhosis, including precipitating encephalopathy, warranting caution with their use.5 It is reasonable to keep in mind that opioid effects may be reversed with naloxone, while complications of untreated or undertreated pain may be significant.14 Opioids may be necessary after appropriate nonpharmacologic and nonopioid treatments have produced inadequate results.7
Oxidation is the major metabolic pathway for opioids (often via CYP enzymes), except for morphine, and is considered to be affected by liver dysfunction.4,15 Oxidation is reduced in liver disease, leading to decreased drug clearance and/or increased oral bioavailability with reduced first-pass metabolism. While glucuronidation is believed to be less affected by mild-to-moderate liver dysfunction, it can be significant with severe disease.4 With these considerations in mind, certain opioids are considered to be safe, effective, and preferred to NSAIDs for moderate-to-severe pain management with the use of reduced doses and extended intervals. However, due to vague recommendations, the decision is often left to the practitioner’s clinical judgment.15 Long-acting agents may be considered only after safe and effective dosing of short-acting agents is determined.5
Tramadol: Tramadol undergoes more than 80% hepatic metabolism.4 While it has been expected to provide less pain relief in cirrhotic patients due to decreased biotransformation, this has not been seen in clinical studies.4 One study noted a significant difference in the peak concentration (Cmax) and peak time (Tmax), clearance reduced by half, and an elimination half-life of up to 3 times control with 20 hepatic malignancy patients compared to 10 healthy controls.16 The authors recommend prolonging the dosing interval, considering tramadol 50 mg every 12 hours safe and effective. Others have recommended 25 mg every 8 hours in liver dysfunction.5 Drug information resources, such as Lexicomp, recommend 50 mg every 12 hours for cirrhosis patients and state that extended-release product should not be used in patients with severe (Child-Pugh class C) hepatic dysfunction.17
Tramadol may be beneficial in intractable pain due to its partial inhibition of serotonin reuptake and activity on peripheral pain pathways.5 Caution should be used with tramadol in patients with seizure history and in combination with other serotonergic medications, due to its effect of lowering seizure threshold and possible development of serotonin syndrome, respectively. The dose and frequency of tramadol should also be reduced according to renal function.17
Hydrocodone and Oxycodone: Hydrocodone and oxycodone rely on CYP2D6 and 3A4 for metabolism to their more potent metabolites, hydromorphone and oxymorphone, respectively.5 The analgesic effect of these medications may be less potent in liver dysfunction patients due to decreased conversion to their active metabolites, while decreased clearance and prolonged half-life may produce more unwanted effects.4,5 It is recommended to initiate therapy with lower doses and extended intervals, allowing for adequate time between doses to avoid accumulation and titrating according to patient response. Caution should be used with acetaminophen combination products (e.g., Vicodin, Percocet), ensuring the total daily acetaminophen dose is within the recommended 2 to 3 g/day limit from all sources.5
Morphine: Morphine is considered to have a high hepatic extraction on first-pass metabolism, making it only 30% to 40% bioavailable following oral dosing in healthy patients.4,5 Morphine is metabolized by the liver to morphine-6-glucuronide (active metabolite) and morphine-3-glucuronide (inactive metabolite with potential neurotoxic effects), which are then cleared renally. With reduced first-pass metabolism in cirrhotic patients, morphine is likely more bioavailable and lower oral doses are recommended.4,5,15 Studies have consistently shown decreased clearance and a prolonged half-life for morphine in cirrhotic patients. It has been recommended to increase the dosing interval by as much as double to avoid drug accumulation, in both IV and oral administration.5 Morphine should be avoided in renal impairment, including hepatorenal syndrome, due to significant accumulation of its metabolites and risk for neurotoxicity.4
Hydromorphone: Some studies report a Cmax increased up to four times following a single immediate-release dose of hydromorphone in patients with moderate liver disease, due to decreased first-pass metabolism in liver dysfunction and high hepatic extraction.4 It has been recommended to start with lower doses but maintain a similar dosing interval.4,5 Pharmacokinetics for hydromorphone have not been studied in patients with severe liver disease. Hydromorphone’s clearance appears to be relatively unaffected, particularly by renal dysfunction. However, it has been suggested that hydromorphone not be used in hepatorenal syndrome due to the observed risk of accumulation of its neuroexcitatory metabolite, hydromorphone-3-glucuronide.4
Fentanyl: Although fentanyl is heavily protein bound, it is thought to be unaffected by cirrhosis.4,5,15 Fentanyl may be better tolerated in cirrhotic patients, as it has no toxic metabolites.4 However, fentanyl may not be the most appropriate agent due to its very high potency and other clinical scenarios (e.g., acute pain, outpatient management, and the need for oral administration).15
The use of transdermal fentanyl patches has been recommended as a first-line option for intractable pain by some authors, but this is controversial as cirrhosis patients have been found to have elevated Cmax concentrations and area under the curve (AUC) when using the patches, 35% and 73%, respectively.4,5,18 It is important to remember that the use of a fentanyl patch should only be considered once opioid requirements have been determined by the titration of short-acting opioids.15 It is recommended to initiate at 50% of the usual dose in mild-to-moderate hepatic impairment and avoid use in severe hepatic impairment.18 Fentanyl is considered a good option in renal impairment, and IV administration may be considered in hepatorenal syndrome, as it is one of the opioids least affected by renal dysfunction.4,5
Meperidine: In current practice, meperidine is rarely used for analgesia due to the risk of seizures with accumulation of its normeperidine metabolite. Although normeperidine concentrations may be less due to decreased CYP activity in these patients, there is increased bioavailability related to decreased protein binding and delayed clearance, and the metabolite can still accumulate.4,5,15 Meperidine should be avoided in cirrhosis.
Codeine: In healthy patients, codeine’s analgesic effect is variable due to CYP2D6 phenotypic variation, affecting conversion to morphine. In liver dysfunction patients, codeine’s serum levels may be more variable due to decreased CYP activity, causing diminished analgesic effect.4,5,15 Due to a general lack of studies in cirrhotic patients, codeine is not recommended.
Methadone: For the management of opioid dependence, methadone is often used. The drug presents many challenges with its significant interindividual variability in bioavailability, protein binding, and long half-life.4,5,15 It is predominantly metabolized by CYP3A4.15 Accumulation may occur with repeated dosing in severe liver dysfunction, and plasma concentrations may be elevated by acute alcohol ingestion.4 The use of methadone for analgesia in patients with liver disease has not been studied; thus, it is not recommended for this use in these patients.4 However, the benefits of monitored methadone maintenance programs for opioid dependence, such as heroin abuse, may outweigh potential risks.5
Buprenorphine: Oral administration of buprenorphine, a partial mu opioid agonist, results in extensive first-pass metabolism and poor analgesic effect, whereas sublingual administration allows for about 50% to 55% of the drug to avoid inactivation by CYP3A4.4,15 Unfortunately, studies on buprenorphine are not currently available to determine whether dose adjustments are needed or if the pharmacokinetics remain unchanged in hepatic impairment. Hepatitis has been reported with buprenorphine use in patients with preexisting hepatic dysfunction. For these reasons, buprenorphine should be used cautiously in patients with liver disease.
Miscellaneous
Lidocaine: Topical lidocaine patch use is a common consideration for localized pain control. Although oral lidocaine is considered to have a high hepatic extraction with first-pass metabolism, no literature was found discussing the pharmacokinetics or use of lidocaine patches for pain management in cirrhotic patients.19 Pain in liver dysfunction may be diffuse and/or associated with ascites, limiting the indication for topical lidocaine patches.7 Due to the lack of evidence and possible toxicity, lidocaine should be used cautiously in liver dysfunction patients and only for localized pain.
Tricyclic Antidepressants (TCAs): TCAs have long been the mainstay of neuropathic pain therapy.5 TCAs undergo first-pass metabolism and renal excretion. These agents are well-known for their anticholinergic effects, to which cirrhotic patients may be more susceptible. Constipation is a significant concern, which may precipitate hepatic encephalopathy in these patients. These medications should be used cautiously and only if necessary. If used, nortriptyline and desipramine may be preferred due to lower potency and less sedation, tachycardia, and orthostatic hypotension. It is recommended to begin at low doses in liver disease and titrate slowly based on patient response.4
Anticonvulsants: Anticonvulsants also play a role in the neuropathic pain management (via neurotransmitter modulation), but these drugs are commonly metabolized by the liver and excreted renally.5 Lower doses and prolonged intervals may be necessary in cirrhotic patients. Carbamazepine is associated with hepatotoxicity and should be avoided in patients with liver dysfunction. Gabapentin is the preferred agent, as it is not metabolized hepatically nor protein bound, but should be adjusted according to renal function.4
Summary and Recommendations
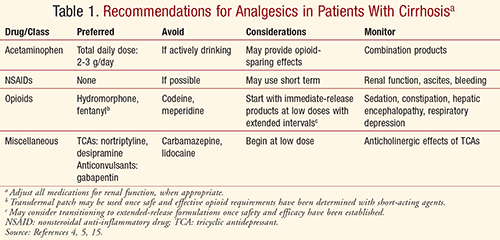
With all patients, pain management must be individualized based on benefits versus risks with close monitoring of pain relief and adverse effects. Pain management should involve a multidisciplinary approach of pharmacologic, behavioral, psychological, and physical interventions to achieve the best results and optimize quality of life, safely and effectively. Practitioners must always be mindful of increased potential for drug-induced liver injury in patients with preexisting liver disease, including cirrhosis.5 With the pharmacokinetic and adverse-effect profiles of various analgesic medications in mind, practitioners should consider the recommendations summarized in TABLE 1 to achieve safe and adequate pain control in cirrhotic patients.
REFERENCES
1. Understanding cirrhosis of the liver. American Gastroenterological Association. Updated 2015. www.gastro.org/info_for_patients/2013/6/6/understanding-cirrhosis-of-the-liver. Accessed November 9, 2015.
2. CDC. Chronic liver disease and cirrhosis. February 6, 2015. www.cdc.gov/nchs/fastats/liver-disease.htm. Accessed November 9, 2015.
3. Asrani SK, Larson JJ, Yawn B, et al. Underestimation of liver-related mortality in the United States. Gastroenterology. 2013;145(2):375-382.e1-2.
4. Bosilkovska M, Walder B, Besson M, et al. Analgesics in patients with hepatic impairment: pharmacology and clinical implications. Drugs. 2012;72:1645-1669.
5. Chandok N, Watt KD. Pain management in the cirrhotic patient: the clinical challenge. Mayo Clin Proc. 2010;85(5):451-458.
6. Rogal SS, Winger D, Bielefeldt K, Szigethy E. Pain and opioid use in chronic liver disease. Dig Dis Sci. 2013;58(10):2976-2985.
7. Madan A, Barth KS, Balliet WE, et al. Chronic pain among liver transplant candidates. Prog Transplant. 2012;22(4):379-384.
8. Rogal SS, Winger D, Bielefeldt K, et al. Healthcare utilization in chronic liver disease: the importance of pain and prescription opioid use. Liver Int. 2013;331497-1503.
9. Benson GD. Acetaminophen in chronic liver disease. Clin Pharmacol Ther. 1983;33(1):95-101.
10. Lauterburg BH. Analgesics and glutathione. Am J Ther. 2002;9(3):225-233.
11. Benson GD, Koff RS, Tolman KG. The therapeutic use of acetaminophen in patients with liver disease. Am J Ther. 2005;12(2):133-141.
12. De Ledinghen V, Heresbach D, Fourdan O, et al. Anti-inflammatory drugs and variceal bleeding: a case-control study. Gut. 1999;44:270-273.
13. Ofirmev (acetaminophen injection) package insert. Hazelwood, MO: Mallinckrodt Hospital Products Inc; 2014.
14. Swetz KM, Carey EC, Rho RH, et al. Safe use of opioids to manage pain in patients with cirrhosis. Mayo Clin Proc. 2010;85(10):959. www.ncbi.nlm.nih.gov/pmc/articles/PMC2947971/. Accessed February 6, 2015.
15. Tegeder I, Lötsch J, Geisslinger G. Pharmacokinetics of opioids in liver disease. Clin Pharmacokinet. 1999;37(1):17-40.
16. Kotb HI, Fouad IA, Fares KM, et al. Pharmacokinetics of oral tramadol in patients with liver cancer. J Opioid Manag. 2008;4(2):99-104.
17. Tramadol. Lexi-Drugs. Lexicomp. Hudson, OH: Wolters Kluwer Health, Inc. http://online.lexi.com. Accessed November 9, 2015.
18. Duragesic (fentanyl transdermal system) package insert. Titusville, NJ: Janssen Pharmaceuticals, Inc; 2009.
19. Lutwak N, Howland MA, Gambetta R, Dill C. Even ‘safe’ medications need to be administered with care. BMJ Case Rep. January 2, 2013. www.ncbi.nlm.nih.gov/pmc/articles/PMC3604254/. Accessed February 6, 2015.
To comment on this article, contact rdavidson@uspharmacist.com.





