US Pharm. 2013;38(1):19-22.
ABSTRACT: Autism spectrum disorders (ASDs) are a group of developmental disorders marked by significant social, communication, and behavioral impairments. The CDC estimates that 1 in 88 children has an ASD. While there is no cure for ASDs, there are both nonpharmacologic and pharmacologic therapies available to aid in the management of this condition. Management goals include minimizing the core characteristics through educational interventions, treating comorbidities commonly associated with ASDs, improving independence and quality of life, and easing familial distress through education and support. Pharmacists can assist patients and parents and/or caregivers in a variety of ways to manage ASDs.
Surveillance data indicate a rise in identified autism spectrum disorders (ASDs) from 1 in 150 children in 2000 to 1 in 88 children in 2008.1 ASDs, also known as pervasive developmental disorders, encompass a range of symptoms, skills, and levels of impairment, which explain their description as “spectrum” disorders.2 Within ASDs are autistic disorder; Asperger’s disorder (Asperger syndrome); pervasive developmental disorder–not otherwise specified (PDD-NOS); Rett’s disorder; and childhood disintegrative disorder.3
While ASDs occur in all racial, ethnic, and socioeconomic groups, current epidemiological data suggest that prevalence is highest among non-Hispanic white children.1 In addition, ASDs are more common among boys than girls, with an estimated prevalence of 1:54 compared to 1:252, respectively.1 With increasing prevalence, health care costs also rise. Annual medical costs (average of $10,700 per child) are 4 to 6 times higher for a child with an ASD compared to a child without.4,5
Etiology
While there is no definitive cause for ASD, research shows that both genetics and the environment play a role.2 Genetic causes include fragile X syndrome, which results from a single genetic mutation causing intellectual disability, and tuberous sclerosis, a rare genetic disorder that causes noncancerous tumor growth in the brain and vital organs.2,6 Additionally, the presence of a genetic link predisposes a child to ASD. In 9 out of 10 cases, if one identical twin has ASD, the other twin also has ASD.7,8 In addition, there is 35 times the risk for siblings of a child with ASD to develop the disorder. It is important to note that while there is an increased familial risk, development of ASD is not definite.7,8
Research continues to explore environmental risks, such as parental age, family medical conditions, complications during birth and pregnancy, and the potential for ASD development.2 Vaccines containing thimerosal are also at the center of the debate regarding ASD risk. However, no evidence supports this theory, and the American Academy of Pediatrics (AAP) supports childhood immunizations.9
Screening, Diagnosis, and Symptoms
A definitive diagnosis of ASD may be difficult to determine, as the manifestations of ASD vary greatly. Notable differences among children are onset of symptoms, symptom severity, and the nature of the symptoms.10 Core ASD symptoms include impaired social interaction skills, communication difficulties, and the presence of repetitive or stereotyped behaviors, interests, and activities.2,3
Children with an ASD do not achieve typical developmental skills in early childhood.2 Parents are often the first to detect such a delay, which may occur prior to a child’s first birthday. However, other children may show appropriate development until the second or third year of life. Loss or reversal of normal development can also occur.2,10
Early identification of ASD is critical, as evidence suggests that early identification and subsequent intervention may result in significantly better outcomes.11 The AAP surveillance algorithm for screening infants and young children for developmental disorders suggests that developmental surveillance be incorporated into every well-child visit though age 5 years.12 The five components of surveillance are eliciting and attending to parental concerns about the child’s development; documenting and maintaining a developmental history; making accurate observations of the child; identifying the presence of risk and protective factors; and documenting the process and findings.12
If concerns arise as a result of surveillance, further assessment with developmental screening tests is warranted. The use of screening tools to evaluate the child’s visual and hearing abilities, early communication and language skills, and motor skills are tailored to the child’s current age.12 In order to ensure that a child is meeting the appropriate developmental milestones (TABLE 1), screenings are administered at the 9-, 18-, and 24- or 30-month visits.12 If, at any time, a child fails to meet any milestone, further assessment is necessary, as this indicates a high likelihood for the presence of a developmental disability.13

The Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR) criteria for the diagnosis of autistic disorder are provided in TABLE 2. Other disorders within the spectrum have different diagnostic criteria per the DSM-IV-TR.3 Of note, the DSM-V, scheduled for official release in May 2013, may impose significant changes in the classification and diagnosis of ASDs. The most considerable change comes with the labeling of ASDs. The current DSM-IV-TR defines autistic disorder, Asperger’s disorder, and PDD-NOS and considers them “autism spectrum disorders.” The DSM-V is slated to replace the individual diagnostic labels of autistic disorder, Asperger’s disorder, and PDD-NOS with one diagnostic label of autism spectrum disorder. Further distinction within the diagnosis, will be based on severity and noted by level 1, 2, or 3.14

Nonpharmacologic Therapies
ASDs require lifetime management. The primary goals of nonpharmacologic management are to minimize the associated core characteristics, improve the child’s independence and quality of life, and ease family distress through education and support.11 Educational interventions are the foundation of ASD management and aim to enhance communication, social, daily-living, play, and leisure skills. Children should begin developmental interventions as soon as the diagnosis of autism is considered. The AAP guidelines recommend intensive educational interventions, such as active engagement of the child for at least 25 hours per week year-round. Specific interventional strategies are detailed in TABLE 3.11

Pharmacologic Therapies
Medications do not correct the core deficits associated with ASD, but rather treat medical conditions and psychiatric comorbidities that often accompany ASDs. Children with an ASD often have additional conditions including sleep or sensory problems, seizures, intellectual disability, mental disorders (anxiety, depression, or attention-deficit/hyperactivity disorder [ADHD]), and gastrointestinal (GI) disorders.2 One study reported that among children with ASDs, about half had at least one of the three commonly co-occurring conditions (ADHD, intellectual disability, or epilepsy).5
The prevalence of epilepsy among ASD patients ranges from 11% to 39%, with a higher prevalence in patients with comorbid mental retardation and motor deficits.11 The onset of epilepsy in ASDs is biphasic in distribution, peaking before 5 years of age and again in adolescence. Selection of anticonvulsant medication and treatment duration in children with epilepsy and an ASD does not differ from routine childhood epilepsy treatment.11
Sleep disturbances are also common. It is important to identify if the sleep disturbance has an identifiable etiology, and if so, to correct it. If there is no identifiable cause, behavioral interventions such as good sleep hygiene, restriction of daytime sleep, and positive bedtime routines may be effective. While medications such as antihistamines, alpha2-agonists, benzodiazepines, trazodone, and newer sedative hypnotic agents are occasionally used in the management of pediatric insomnia, the evidence for the use of these agents in children with an ASD is lacking.11 Additionally, some evidence supports melatonin dysregulation and disruption of the circadian rhythm.15 A systematic review of randomized trials, assessing the efficacy of oral melatonin versus placebo in children with developmental disorders, found that while melatonin did not provide any significant improvements in the total amount of sleep or the number of nighttime awakenings, there was a statistically significant benefit for the time to sleep onset.15 Therefore, it may be reasonable to try melatonin if sleep onset is the issue.
A range of behavioral symptoms often accompany ASDs, including hyperactivity, short attention span, impulsivity, aggressiveness, self-injurious behaviors, and temper tantrums.3 It is important to determine if the source of the behavior has an identifiable etiology, such as constipation, headache, dental pain, acute otitis media, or sinusitis, as this may instigate maladaptive behavior.11 With no identifiable cause, a trial of medication may be considered. While the use of psychoactive medications is common, there is little evidence comparing the use of various agents for symptom management associated with ASDs.11
Risperidone, an atypical antipsychotic, was the first medication to receive FDA approval for the treatment of irritability associated with autistic disorder in patients aged 5 to 16 years (TABLE 4). Risperidone demonstrated a 25% improvement on the aberrant behavior checklist (ABC) in 90% of patients in clinical trials.16 The ABC assesses drug and treatment effects using five scales, including: 1) irritability, agitation, and crying; 2) lethargy and social withdrawal; 3) stereotypical behavior; 4) hyperactivity and noncompliance; and 5) inappropriate speech.17 Adverse events associated with risperidone include somnolence, sedation, increased appetite, and nausea.16
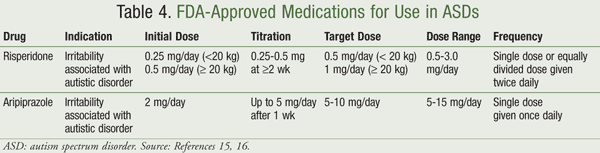
Another atypical antipsychotic, aripiprazole, is also approved for the treatment of irritability associated with autistic disorder in patients aged 6 to 17 years (TABLE 4). Efficacy was determined by two 8-week trials in patients with autistic disorder with significantly improved scores on the ABC and the Clinical Global Impression-Improvement (CGI-I) scale.18 The CGI-I measures change in clinical status since start of treatment using a 7-point scale. Evaluation is based on symptomatic improvements from baseline and impact of the changes on overall behavior and function.19 While aripiprazole may be a reasonable treatment option, the prescribing information specifies that medication treatment for pediatric patients with autistic disorder should only be initiated as part of a treatment program that includes psychological, educational, and social interventions. Adverse events include sedation, fatigue, vomiting, somnolence, and tremor.18
Selective serotonin reuptake inhibitors (SSRIs), while not FDA approved for ASDs, were studied for the treatment of repetitive behaviors, obsessive-compulsive symptoms, aggression, anxiety, and depressive disorder, with conflicting evidence.11,20 In a 12-week trial in adults with ASDs, fluoxetine started at 10 mg/day and titrated to 80 mg/day as tolerated showed significantly greater improvement in repetitive behaviors on both the Yale-Brown compulsion subscale and the CGI-I scale.20 The Yale-Brown compulsion subscale is a component of the larger Yale-Brown Obsessive Compulsive Scale (Y-BOCS). When applied to ASD patients, the subscale measures the severity and type of symptoms associated with compulsions, including washing/cleaning, checking, repeating, counting, arranging, collecting, and miscellaneous.21 The study found 1.4 side effects per patient in the fluoxetine group compared to 0.6 in the placebo group. The most common side effects experienced in the treatment group were vivid dreams, dry mouth, insomnia, depressed mood, and nausea.20
In contrast, a 2009 trial targeting repetitive behaviors in ASD patients aged 5 to 17 years compared citalopram to placebo and found no significant difference in the response rate on the CGI-I or the Yale-Brown compulsion scale.20,22 Citalopram was also associated with a higher incidence of adverse events (impulsiveness, hyperactivity, diarrhea, insomnia).22 It is important to note that the use of SSRIs require caution and careful monitoring, as all carry a black box warning for increased risk of suicidal thinking and behavior in individuals up to age 24 years.23
The use of complementary and alternative medicine (CAM) is common for patients with ASDs.11,24 CAM use in ASDs is classified as nonbiological or biological. Nonbiological methods consist of music therapy and facilitated communication, whereas biological interventions range from dietary restrictions to GI treatments such as probiotics, vitamins, minerals, or other natural products.11 There is currently insufficient evidence to support the majority of CAM interventions for the treatment of ASDs, as many CAM therapies have been inadequately evaluated or have been shown to be ineffective.11
The Pharmacist’s Role
Pharmacists must ensure that the patient and/or parent or caregiver understands each medication, its use, monitoring parameters, and potential side effects and/or interactions, intervening as necessary. Pharmacists should inquire about CAM use due to potential interactions with prescription medications. Pharmacists can also serve as a drug information resource, researching and evaluating available evidence to assist patients in informed decision making.
Pharmacists should discuss not only medication use but also ASDs and their management, and recommend resources for parents and/or caregivers (TABLE 5). Taking the time to speak with the parent and/or caregiver of a child with an ASD helps to develop a strong relationship, which is necessary for the pharmacist in understanding each patient’s needs and providing optimal care.

Finally, pharmacists should work as a member of the patient’s health care team. With frequent patient contact, pharmacists can assess response to therapy, communicate with medical providers, and identify and explore medication nonadherence. Pharmacists may also work with patients to develop goals to better manage the ASD and its associated symptoms.
REFERENCES
1. CDC. Prevalence of autism spectrum disorders—autism and
developmental disabilities monitoring network, 14 sites, United States,
2008. MMWR Surveill Summ. 2012;61:1-19.
2. A parent’s guide to autism spectrum disorder. National
Institute of Mental Health.
www.nimh.nih.gov/health/publications/a-parents-guide-to-autism-spectrum-disorder/what-is-autism-spectrum-disorder-asd.shtml.
Accessed August 2, 2012.
3. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR). Washington, DC: American Psychiatric Publishing, Inc; 2000.
4. Levy SE, Giarelli E, Lee LC, et al. Autism spectrum
disorder and co-occurring developmental, psychiatric, and medical
conditions among children in multiple populations of the United States. J Dev Behav Pediatr. 2010;31:267-275.
5. Peacock C, Amendah D, Ouyang L, Grosse SD. Autism
spectrum disorders and health care expenditures: the effects of
co-occurring conditions. J Dev Behav Pediatr. 2012;33:2-8.
6. Zafeiriou DI, Ververi A, Vargiami E. Childhood autism and associated comorbidities. Brain Dev. 2007;29:257-272.
7. Arking DE, Cutler DJ, Brune CW, et al. A common genetic
variant in the neurexin superfamily member CNTNAP2 increases familial
risk of autism. Am J Hum Genet. 2008;82:160-164.
8. Campbell DB, Sutcliffe JS, Ebert PJ, et al. A genetic variant that disrupts MET transcription is associated with autism. Proc Natl Acad Sci U S A. 2006;103:16834-16839.
9. American Academy of Pediatrics. Prevalence of autism
spectrum disorders.
www.aap.org/en-us/about-the-aap/aap-press-room/Pages/Prevalence-of-Autism-Spectrum-Disorders.aspx.
Accessed August 2, 2012.
10. Autism spectrum disorders (ASDs). CDC. www.cdc.gov/ncbddd/autism/index.html. Accessed August 2, 2012.
11. Myers SM, Johnson CP; American Academy of Pediatrics
Council on Children with Disabilities. Management of children with
autism spectrum disorders. Pediatrics. 2007;120:1162-1182.
12. Johnson CP, Myers SM; American Academy of Pediatrics
Council on Children with Disabilities. Identification and evaluation of
children with autism spectrum disorders. Pediatrics. 2007;120:1183-1215.
13. Filipek PA, Accardo PJ, Ashwal S, et al. Practice
parameter: screening and diagnosis of autism—report of the Quality
Standards Subcommittee of the American Academy of Neurology and the
Child Neurology Society. Neurology. 2000;55:468-479.
14. DSM-V: What changes may mean. Autism Research
Institute. www.autism.com/index.php/news_dsmV. Updated 2012. Accessed
December 8, 2012.
15. Phillips L, Appleton RE. Systematic review of
melatonin treatment in children with neurodevelopmental disabilities and
sleep impairment. Dev Med Child Neurol. 2004;46:771-775.
16. Risperdal (risperidone) package insert. Titusville, NJ: Janssen Pharmaceuticals, Inc; 2007.
17. Aman M, Singh N, Stewart A, Field C. The aberrant
behavior checklist: a behavior rating scale for the assessment of
treatment effects. Am J Ment Defic. 1985;89:485-491.
18. Abilify (aripiprazole) package insert. Tokyo, Japan: Otsuka Pharmaceutical Co; February 2012.19. Busner J, Targum S. The clinical global impressions scale: applying a research tool in clinical practice. Psychiatry. 2007;4:28-37.
20. Hollander E, Soorya L, Chaplin W, et al. A
double-blind placebo-controlled trial of fluoxetine for repetitive
behaviors and global severity in adult autism spectrum disorders. Am J Psychiatry. 2012;169:292-299.
21. Yale-Brown obsessive compulsive scale (Y-BOCS).
www.stlocd.org/handouts/YBOC-Symptom-Checklist.pdf. Accessed December 8,
2012.
22. King BH, Hollander E, Sikich L, et al; STAART
Psychopharmacology Network. Lack of efficacy of citalopram in children
with autism spectrum disorders and high levels of repetitive behaviors:
citalopram ineffective in children with autism. Arch Gen Psychiatry. 2009;66:583-590.
23. Antidepressant use in children, adolescents, and adults. FDA.
www.fda.gov/Drugs/DrugSafety/InformationbyDrugClass/UCM096273. Updated 2010. Accessed August 2, 2012.
24. National Center for Complementary and Alternative Medicine. Expanding Horizons of Healthcare: Five-Year Strategic Plan 2001–2005. Washington, DC: U.S. Department of Health and Human Services; 2000.
25. Autism Research Institute. Structured Teaching—The
TEACCH Method. www.autism.com/index.php/treating_TEACCH. Accessed July
26, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





